IB DP Chemistry Mock Exam HL Paper 2 Set 4 - 2025 Syllabus
IB DP Chemistry Mock Exam HL Paper 2 Set 4
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam HL Paper 2 Set 4. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
(iii) Determine the enthalpy change for \(\text{CO} + \text{Cl}_2 \rightarrow \text{Cl}_2\text{CO}\) using average bond enthalpies.
Use section \(12\) of the data booklet (average bond enthalpies), and also the values below:
• \(\text{Cl}-\text{Cl}\): \(\;242\ \text{kJ mol}^{-1}\)
• \(\text{C}=\text{O}\): \(\;804\ \text{kJ mol}^{-1}\)
• \(\text{C}-\text{Cl}\): \(\;324\ \text{kJ mol}^{-1}\)
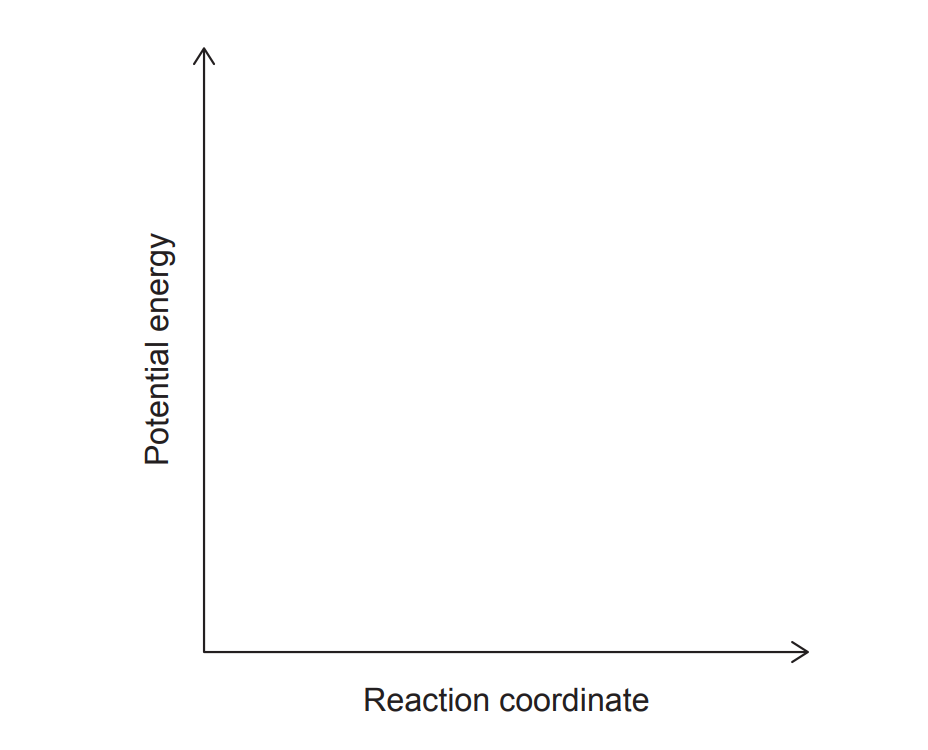
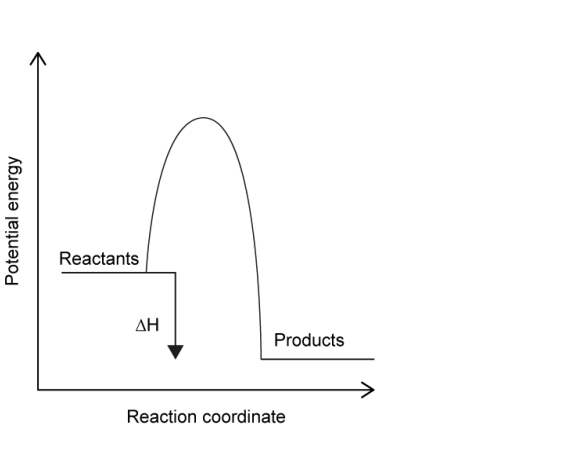
(iv) Sketch an energy profile for this reaction and label “Reactants”, “Products” and \(\Delta H\).
(v) Calculate the standard entropy change, \(\Delta S^{\ominus}\), for the reaction.
Use section \(13\) of the data booklet and the data below .
• \(\text{S}^{\ominus}(\text{Cl}_2) = 223\ \text{J mol}^{-1}\text{K}^{-1}\) (given)
• \(\text{S}^{\ominus}(\text{phosgene}) = 284\ \text{J mol}^{-1}\text{K}^{-1}\) (given)
(vi) Deduce whether the reaction will go to completion or establish equilibrium at \(298\ \text{K}\).
Use sections \(1\) and \(2\) of the data booklet along with your answers to (a)(iii) and (a)(v), and also the values below:
• \(\Delta G^{\ominus} = \Delta H^{\ominus} – T\Delta S^{\ominus}\) (sections \(1\)–\(2\))
• \(\Delta G^{\ominus} = -RT\ln K\) (sections \(1\)–\(2\))
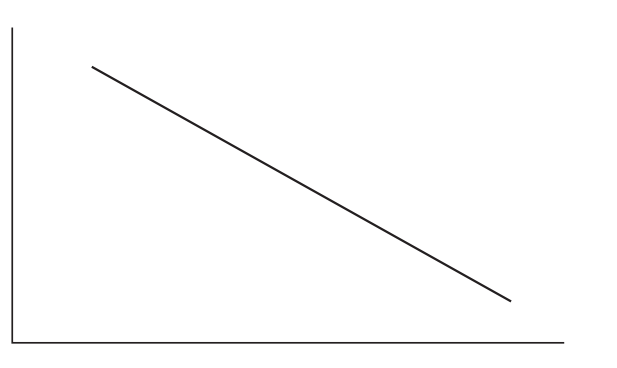
(vi) Calculate the gradient of the line in (b)(v) if \(E_a = 12\ \text{kJ mol}^{-1}\).
Use sections \(1\) and \(2\) of the data booklet , and also:
• Gradient of \(\ln k\) vs \(1/T\) is \(-E_a/R\) (sections \(1\)–\(2\))
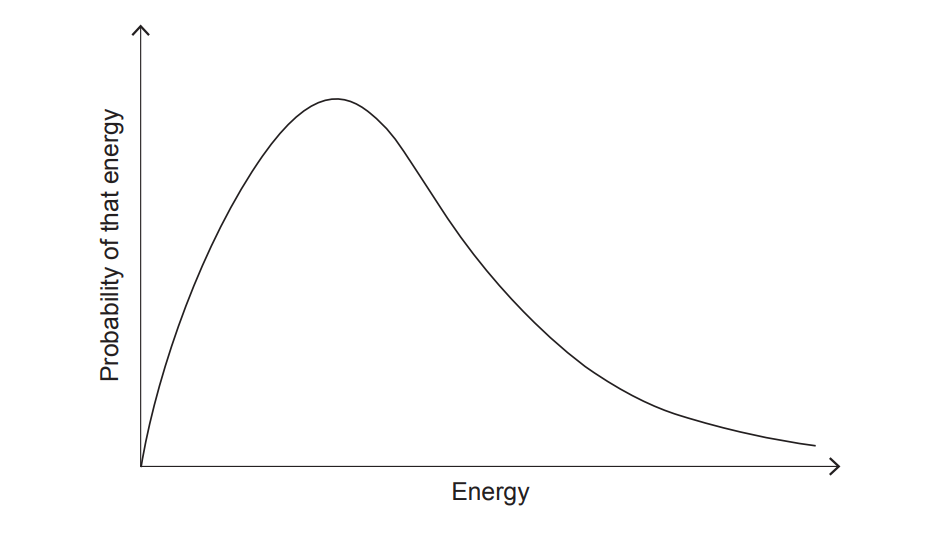
(vii) Sketch a Maxwell–Boltzmann energy distribution at a higher temperature than the one shown.
(iii) Predict the main features of the IR spectrum and the \(^1\text{H}\) NMR spectrum of phosgene.
Use section \(20\) of the data booklet , and also the typical values below:
• \(\text{C}-\text{Cl}\) absorption: \(\;600\text{–}800\ \text{cm}^{-1}\)
• \(^1\text{H}\) NMR: phosgene contains no hydrogen atoms, so no \(^1\text{H}\) signals are expected.
▶️ Answer/Explanation
\(\text{Cl}_2 \xrightarrow{\text{UV}} 2\text{Cl}^\bullet\)
Homolytic fission.
Bonds broken:
\(\text{C}\equiv\text{O} + \text{Cl}-\text{Cl} = 1077 + 242 = 1319\ \text{kJ mol}^{-1}\)
Bonds formed (in \(\text{Cl}_2\text{C}=\text{O}\)):
\(\text{C}=\text{O} + 2(\text{C}-\text{Cl}) = 804 + 2(324) = 1452\ \text{kJ mol}^{-1}\)
\(\Delta H = 1319 – 1452 = \mathbf{-133\ \text{kJ mol}^{-1}}\)
(iv)
Exothermic profile: reactants higher than products; \(\Delta H\) shown as a downward energy difference.
\(\Delta S^\ominus = S^\ominus_{\text{products}} – S^\ominus_{\text{reactants}}\)
\(\Delta S^\ominus = 284 – (198 + 223) = 284 – 421 = \mathbf{-137\ \text{J mol}^{-1}\text{K}^{-1}}\)
Convert \(\Delta S^\ominus\) to \(\text{J mol}^{-1}\text{K}^{-1}\) form inside the calculation and keep units consistent:
\(\Delta G^\ominus = \Delta H^\ominus – T\Delta S^\ominus\)
\(\Delta G^\ominus = -133000 – \big(298 \times (-137)\big) = -133000 + 40826 = -92174\ \text{J mol}^{-1}\)
\(\Delta G^\ominus = -RT\ln K \Rightarrow \ln K = -\Delta G^\ominus/(RT)\)
\(\ln K = \dfrac{92174}{8.31 \times 298} = 37.2\)
\(K = e^{37.2} \approx \mathbf{1.4 \times 10^{16}}\)
Since \(K \gg 1\), the reaction proceeds essentially to completion at \(298\ \text{K}\).
Follow the rate by monitoring gas pressure (or gas volume) at constant temperature, since total moles of gas increase from \(2\) to \(3\).
Some collisions fail because particles may collide with the wrong orientation and/or with kinetic energy less than \(E_a\).
If first order in each reactant:
\(\text{Rate} = k[\text{Cl}_2\text{CO}][\text{H}_2\text{O}]\)
In dilute aqueous solution, \(\text{H}_2\text{O}\) is the solvent and its concentration is effectively constant.
So \(\text{Rate} = k[\text{Cl}_2\text{CO}][\text{H}_2\text{O}] = k'[\text{Cl}_2\text{CO}]\), giving pseudo-first-order behaviour overall.
Arrhenius plot (linear form):
\(y\)-axis: \(\ln k\)
\(x\)-axis: \(1/T\)
Gradient of \(\ln k\) vs \(1/T\) is \(-E_a/R\).
\(E_a = 12\ \text{kJ mol}^{-1} = 12000\ \text{J mol}^{-1}\)
Gradient \(= -\dfrac{12000}{8.31} = \mathbf{-1444\ \text{K}}\)
At higher temperature, the curve becomes lower and broader, with the peak shifted to the right (higher energy). The area under the curve stays the same.
Increasing temperature increases the fraction of molecules with energy \(\ge E_a\).
On the Maxwell–Boltzmann plot, the shaded area to the right of \(E_a\) is larger at higher temperature, so there are more successful collisions per second.
Two confirming observations (any two):
• The reaction is faster when the solid is present.
• The solid is not used up (mass/appearance essentially unchanged after the reaction).
• The catalyst can be recovered at the end of the reaction.
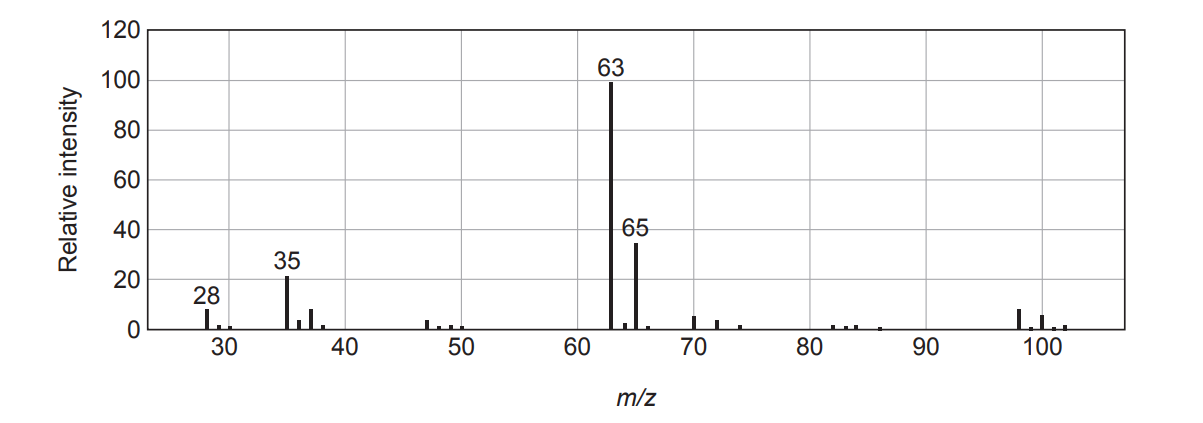
Peaks at lower \(m/z\) occur because the molecular ion fragments into smaller ions in the mass spectrometer.
Approximate molecular mass of phosgene: \(\approx 98\) (using \(\text{Cl}\approx 35\)).
\(98 – 35 = 63\), so this is consistent with loss of \(\text{Cl}\cdot\).
Fragment at \(m/z=63\): \(\mathbf{\text{COCl}^+}\).
IR:
Strong \(\text{C}=\text{O}\) absorption around \(\mathbf{1700\text{–}1750\ \text{cm}^{-1}}\); \(\text{C}-\text{Cl}\) absorptions in the \(\mathbf{600\text{–}800\ \text{cm}^{-1}}\) region.
\(^1\text{H}\) NMR:
No \(^1\text{H}\) signals, because \(\text{Cl}_2\text{CO}\) contains no hydrogen atoms.
