IB DP Chemistry Mock Exam HL Paper 2 Set 5 - 2025 Syllabus
IB DP Chemistry Mock Exam HL Paper 2 Set 5
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam HL Paper 2 Set 5. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
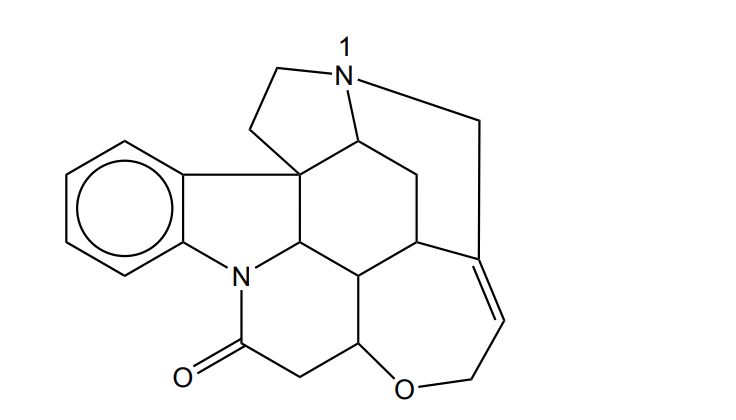
Use section \(7\) of the data booklet (molar masses: \(\mathrm{H}=1.01\), \(\mathrm{C}=12.01\), \(\mathrm{N}=14.01\), \(\mathrm{O}=16.00\), \(\mathrm{S}=32.07\)).
▶️ Answer/Explanation
(a)(i)
The functional group containing \(\mathrm{N_1}\) is a tertiary amine.
(a)(ii)
The amine nitrogen has a lone pair and can accept a proton from water. As a result, strychnine acts as a base in aqueous solution and increases the pH.
(a)(iii)
The alkene \(\mathrm{C{=}C}\) functional group reacts with bromine in the dark by electrophilic addition.
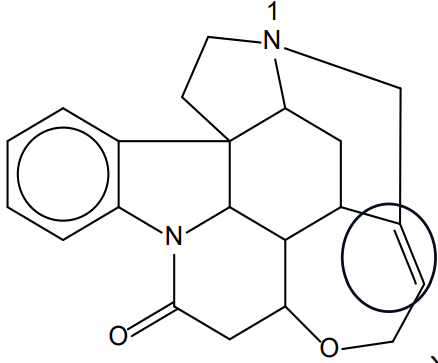
(a)(iv)
The structure of strychnine contains \(\boxed{7}\) rings.
(a)(v)
A chiral carbon atom is bonded to four different atoms or groups, leading to non-superimposable mirror images.
(b)
Moles of strychnine: \[ n = \frac{48.73}{334.4} = 0.1457\,\text{mol} \] Reaction ratio: \(2\) mol strychnine produce \(1\) mol sulfate product. \[ n(\text{product}) = \frac{0.1457}{2} = 0.07286\,\text{mol} \] Using section \(7\) of the data booklet: \[ M_r(\mathrm{H_2SO_4}) = 2(1.01) + 32.07 + 4(16.00) = 98.08 \] Molar mass of sulfate product: \[ M_r = 2(334.4) + 98.08 = 766.88\,\text{g mol}^{-1} \] Theoretical mass of product: \[ m = 0.07286 \times 766.88 = 55.88\,\text{g} \] Percentage yield: \[ \%\,\text{yield} = \frac{51.41}{55.88} \times 100 = \boxed{92.00\%} \]
