IB DP Chemistry Mock Exam SL Paper 1A Set 3 - 2025 Syllabus
IB DP Chemistry Mock Exam SL Paper 1A Set 3
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam SL Paper 1A Set 3. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
\(C_{2}H_{6}(g)+Cl_{2}(g)\rightarrow C_{2}H_{5}Cl(g)+HCl(g)\)
\((C_{2}H_{6}=30.08, Cl_{2}=70.90, C_{2}H_{5}Cl=64.52, HCl=36.46 \text{ g mol}^{-1})\)
What is the atom economy for this reaction?
(B) \(46.6\%\)
(C) \(63.9\%\)
(D) \(99.9\%\)
▶️ Answer/Explanation
1. Atom Economy Formula:
\(\text{Atom Economy} = \frac{\text{Molar Mass of Desired Product}}{\text{Total Molar Mass of All Reactants}} \times 100\%\)
2. Identify Masses:
- Desired Product: Chloroethane (\(C_2H_5Cl\)). Mass = \(64.52\) g/mol.
- Total Reactants: \(C_2H_6 + Cl_2\). Mass = \(30.08 + 70.90 = 100.98\) g/mol. (Alternatively, mass of all products = \(64.52 + 36.46 = 100.98\) g/mol).
3. Calculate:
\(\text{Atom Economy} = \frac{64.52}{100.98} \times 100\%\)
\(\approx 0.6389 \times 100\% = 63.9\%\)
✅ Answer: (C)
Question
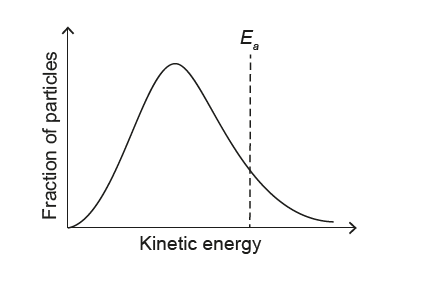
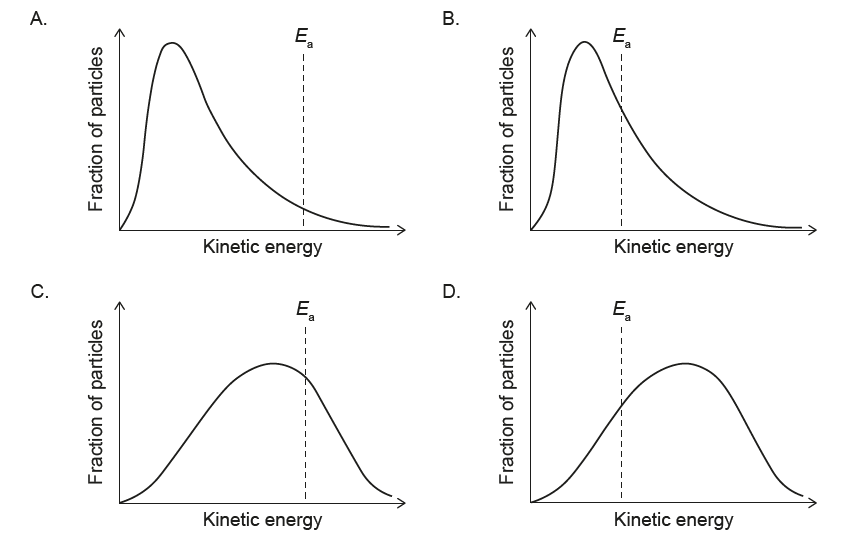
▶️ Answer/Explanation
1. Effect of lowering temperature on the Maxwell–Boltzmann distribution:
When the temperature decreases, the average kinetic energy of the particles decreases.
The Maxwell–Boltzmann curve becomes:
– shifted to the left (towards lower kinetic energy), and
– higher and narrower (more particles with energies close to the new, lower mean).
2. Effect on the activation energy \(E_{a}\):
The activation energy \(E_{a}\) is a fixed property of the reaction and does not change with temperature. Therefore, the vertical line representing \(E_{a}\) must stay at the same kinetic energy value on the horizontal axis.
The only option that shows a narrower, taller curve shifted to lower kinetic energy while keeping the \(E_{a}\) line in the same position is diagram A.
✅ Answer: (A)
Question
(B) \(1.0 \times 10^{-3}\)
(C) \(1.0 \times 10^{3}\)
(D) \(3.0 \times 10^{3}\)
▶️ Answer/Explanation
1. Use Formula:
\([H^+] = 10^{-\text{pH}}\)
2. Substitute:
\([H^+] = 10^{-3.0}\)
\([H^+] = 1.0 \times 10^{-3}\) mol dm\(^{-3}\).
✅ Answer: (B)
