IB DP Chemistry Mock Exam SL Paper 2 Set 2 - 2025 Syllabus
IB DP Chemistry Mock Exam SL Paper 2 Set 2
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam SL Paper 2 Set 2. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
(i) A mass of \(0.108 \text{ g}\) of the gaseous compound occupied a volume of \(55.7 \text{ cm}^3\) at \(100^{\circ}\text{C}\) and a pressure of \(1.00 \times 10^5 \text{ Pa}\). Calculate the amount of the compound, in moles.
(ii) Hence, determine the molar mass of the organic compound.
▶️ Answer/Explanation
(a)
• \(C\): \(9.49 / 44.01 = 0.216 \text{ mol}\). Mass \(C = 2.59 \text{ g}\).
• \(H\): \(2 \times (5.18 / 18.02) = 0.575 \text{ mol}\). Mass \(H = 0.581 \text{ g}\).
• \(O\): \(4.32 – (2.59 + 0.581) = 1.15 \text{ g}\). Moles \(O = 1.15 / 16.00 = 0.0719 \text{ mol}\).
• Ratio \(C:H:O = 0.216 : 0.575 : 0.0719\). Divide by \(0.0719\) \(\rightarrow\) \(3 : 8 : 1\).
• Empirical formula: \(\text{C}_3\text{H}_8\text{O}\).
(b)
(i) \(n = \frac{PV}{RT} = \frac{1.00 \times 10^5 \times 55.7 \times 10^{-6}}{8.31 \times 373} = \mathbf{0.00180 \text{ mol}}\).
(ii) \(M = \frac{m}{n} = \frac{0.108}{0.00180} = \mathbf{60.0 \text{ g mol}^{-1}}\).
Question
▶️ Answer/Explanation
(a)
\[ \text{CaCO}_3(s) + 2\text{HCl}(aq) \rightarrow \text{CaCl}_2(aq) + \text{CO}_2(g) + \text{H}_2\text{O}(l) \]
(b)
• \(n(\text{HCl}) = 4.00 \times 0.020 = 0.0800 \text{ mol}\)
• \(n(\text{CaCO}_3) = \frac{3.162}{100.09} = 0.0316 \text{ mol}\)
• Stoichiometric ratio is \(1:2\). The amount of \(\text{HCl}\) required for \(0.0316 \text{ mol}\) of \(\text{CaCO}_3\) is \(0.0632 \text{ mol}\).
• Since \(0.0800 > 0.0632\), \(\text{HCl}\) is in excess and \(\text{CaCO}_3\) is the limiting reactant.
(c)
\(n(\text{CO}_2) = n(\text{CaCO}_3) = 0.0316 \text{ mol}\)
\(V = n \times 22.7 \text{ dm}^3 \text{ mol}^{-1} = 0.0316 \times 22.7 = \mathbf{0.717 \text{ dm}^3}\).
(d) 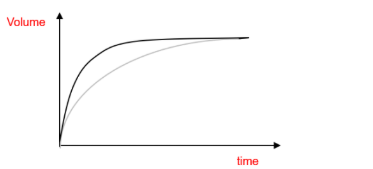
The curve should begin at the origin, show a steeper initial gradient (indicating a faster rate), and level off at the same final volume since the amount of limiting reactant is unchanged.
