IB DP Chemistry Mock Exam SL Paper 2 Set 4 - 2025 Syllabus
IB DP Chemistry Mock Exam SL Paper 2Set 4
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam SL Paper 2 Set 4. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
(i) Write an equation for the initiation step of this reaction.
(ii) Identify the type of bond fission involved.
(iii) Calculate the enthalpy change for the reaction between carbon monoxide and chlorine using average bond enthalpies. Refer to section 12 of the data booklet.
(iv) Draw an energy profile diagram for the reaction and clearly label the reactants, products and \(\Delta H\).
(i) Suggest a method for monitoring the rate of this reaction at constant temperature.
(ii) Explain why not all collisions between reacting particles lead to reaction.
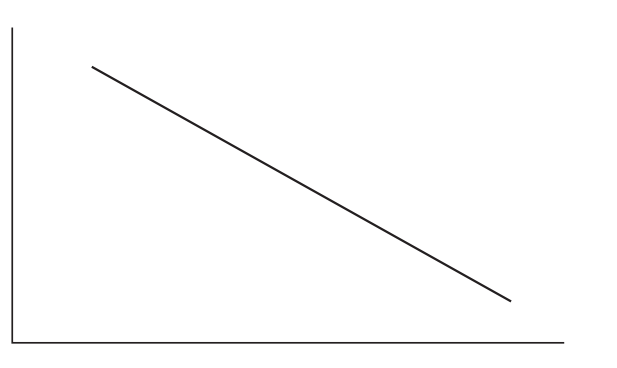
(iii) Sketch a Maxwell–Boltzmann energy distribution curve at a temperature higher than that shown.
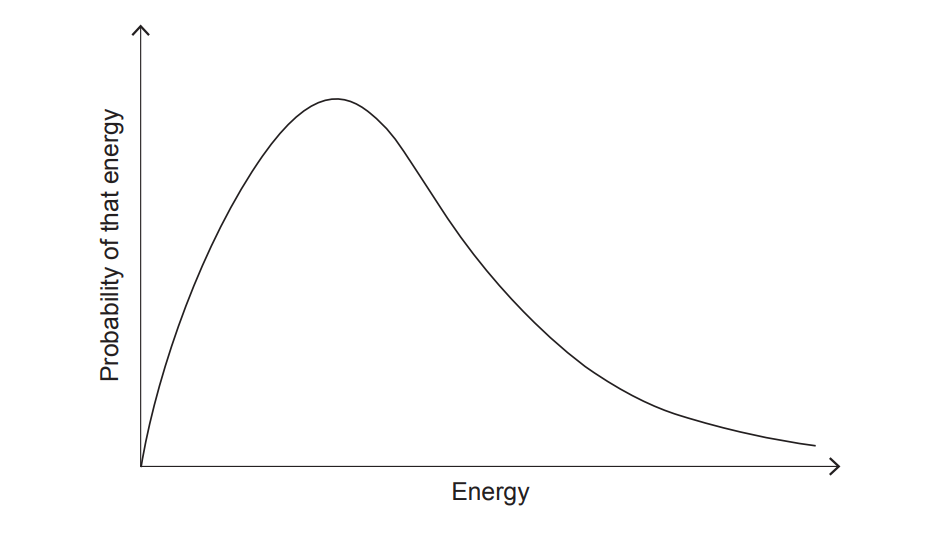
(iv) Use the diagram in (b)(iii) to explain why increasing the temperature increases the reaction rate.
(v) State two observations that would confirm that a solid added to the reaction mixture is acting as a catalyst.
▶️ Answer/Explanation
(a)
(i)
\(\text{Cl}_2(g) \xrightarrow{\text{UV}} 2\text{Cl}^\bullet(g)\).
(ii)
The chlorine–chlorine bond undergoes homolytic fission.
(iii)
Bonds broken: \(\text{C}\equiv\text{O} = 1077\), \(\text{Cl}–\text{Cl} = 242\). Total = \(1319 \text{ kJ mol}^{-1}\).
Bonds formed: \(\text{C}=\text{O} = 804\), \(2 \times \text{C}–\text{Cl} = 648\). Total = \(1452 \text{ kJ mol}^{-1}\).
\(\Delta H = 1319 – 1452 = \mathbf{-133 \text{ kJ mol}^{-1}}\).
(iv)
The energy profile shows reactants at a higher energy than products, indicating an exothermic reaction, with \(\Delta H\) shown as a downward arrow. 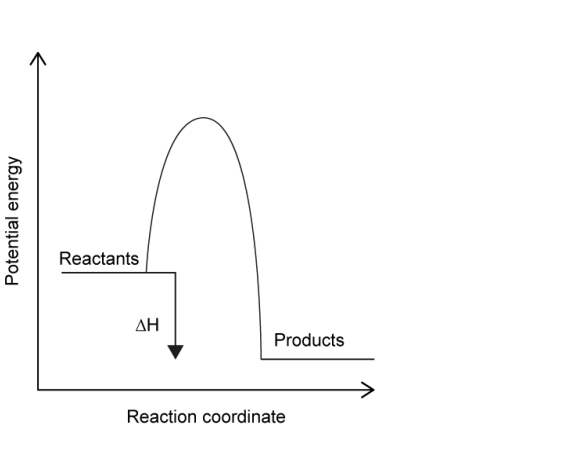
(b)
(i)
The rate may be followed by measuring the change in gas pressure at constant volume or the change in volume at constant pressure.
(ii)
Some collisions are unsuccessful because the particles collide with incorrect orientation or with energy less than the activation energy.
(iii)
At higher temperature, the Maxwell–Boltzmann curve is broader, lower in height, and shifted to the right.
(iv)
Increasing temperature increases the proportion of particles with energy greater than or equal to the activation energy, resulting in more successful collisions per unit time.
(v)
The solid remains chemically unchanged and the reaction proceeds at a faster rate.
Question
(i) Deduce the nuclear symbol of the thallium isotope that contains \(122\) neutrons. Use section 7 of the data booklet.
(From section 7: thallium has atomic number \(81\) and relative atomic mass \(204.38\).)
(ii) Calculate the relative atomic mass of thallium, giving your answer to two decimal places.
(iii) Explain why the first ionization energy of thallium is lower than that of lead, with reference to nuclear charge and shielding of the outer electrons.
(i) This compound contains both ionic and covalent bonding. Identify which particles are connected by covalent bonds and which are connected by ionic bonds.
(ii) Compare covalent and ionic bonding in terms of interactions involving valence electrons.
(iii) Write a balanced equation for the preparation of aqueous thallium(I) sulfate by reacting solid thallium(I) hydroxide with sulfuric acid.
(iv) Calculate the volume of \(2.00\ \text{mol dm}^{-3}\) sulfuric acid needed to completely react with \(10.0\ \text{g}\) of thallium(I) hydroxide.
(v) Using the position of thallium in the periodic table, predict whether thallium(I) hydroxide is amphoteric.
(vi) Describe how an experiment using the metals copper and thallium, together with aqueous solutions of their sulfates, could be used to compare their relative reactivities.
▶️ Answer/Explanation
(a)
(i)
From section 7, thallium has atomic number \(81\).
Isotope has \(122\) neutrons, so mass number \(A = 81 + 122 = 203\).
Nuclear symbol: \(\boxed{{}^{203}\text{Tl}}\)
(ii)
Two isotopes: \({}^{203}\text{Tl}\) (30%) and \({}^{205}\text{Tl}\) (70%) since \(81+124=205\).
\[ A_r = (0.30 \times 203) + (0.70 \times 205) \] \[ A_r = 60.9 + 143.5 = \boxed{204.40} \]
(iii)
Lead has a higher nuclear charge (\(Z=82\)) than thallium (\(Z=81\)).
The shielding of the outer electron is similar (both are period 6 with many inner shells), so the effective nuclear attraction is slightly lower in thallium.
Therefore, the outer electron in thallium is held less strongly, so its first ionization energy is lower than that of lead.
(b)
(i)
• Covalent bonds: within the sulfate ion, between \(\text{S}\) and \(\text{O}\) atoms in \(\text{SO}_4^{2-}\).
• Ionic bonds: between \(\text{Tl}^+\) ions and \(\text{SO}_4^{2-}\) ions.
(ii)
• Covalent bonding: atoms share pairs of valence electrons.
• Ionic bonding: electrons are transferred (forming cations and anions), then ions are held by electrostatic attraction.
(iii)
\[ 2\text{TlOH}(s) + \text{H}_2\text{SO}_4(aq) \rightarrow \text{Tl}_2\text{SO}_4(aq) + 2\text{H}_2\text{O}(l) \]
(iv)
From section 7: \(\text{Tl} = 204.38\), \(\text{O} = 16.00\), \(\text{H} = 1.01\).
\[ M_r(\text{TlOH}) = 204.38 + 16.00 + 1.01 = 221.39\ \text{g mol}^{-1} \] \[ n(\text{TlOH}) = \frac{10.0}{221.39} = 0.0452\ \text{mol} \] From the equation, \(2\ \text{mol TlOH}\) react with \(1\ \text{mol H}_2\text{SO}_4\):
\[ n(\text{H}_2\text{SO}_4) = \frac{0.0452}{2} = 0.0226\ \text{mol} \] \[ V = \frac{n}{c} = \frac{0.0226}{2.00} = 0.0113\ \text{dm}^3 = \boxed{11.3\ \text{cm}^3} \]
(v)
No. Thallium is a metal (group 13, lower down the group shows strong metallic character), so \(\text{TlOH}\) is expected to be mainly basic rather than amphoteric.
(vi)
Set up two displacement tests:
• Place thallium metal in aqueous copper(II) sulfate and observe whether copper is displaced (copper deposits / solution colour decreases).
• Place copper metal in aqueous thallium(I) sulfate and observe whether thallium is displaced.
The metal that displaces the other metal from its sulfate solution is the more reactive metal.
