IB DP Chemistry Mock Exam SL Paper 2 Set 5 - 2025 Syllabus
IB DP Chemistry Mock Exam SL Paper 2 Set 5
Prepare for the IB DP Chemistry Exam with our comprehensive IB DP Chemistry Exam Mock Exam SL Paper 2 Set 5. Test your knowledge and understanding of key concepts with challenging questions covering all essential topics. Identify areas for improvement and boost your confidence for the real exam
Question
(a) The structural formula of strychnine is shown below.
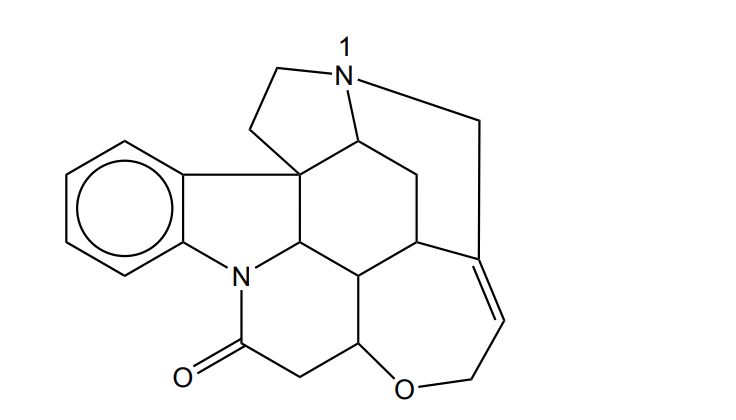
(i) Identify the type of structural formula displayed.
(ii) Name the functional group that contains the nitrogen atom labelled “1”.
(iii) Explain how the functional group containing \(\text{N}_1\) influences the pH when strychnine dissolves in water.
(iv) On the diagram in (a), circle a functional group that would react with bromine in the absence of light.
(v) State how many rings are present in the structure of strychnine.
\(2\text{C}_{21}\text{H}_{22}\text{N}_2\text{O}_2(aq) + \text{H}_2\text{SO}_4(aq) \rightarrow (\text{C}_{21}\text{H}_{23}\text{N}_2\text{O}_2)_2\text{SO}_4(aq)\)
Calculate the percentage yield if \(51.41 \text{ g}\) of the product was formed. Use section 7 of the data booklet.
▶️ Answer/Explanation
(a)
(i)
Skeletal formula.
(ii)
(Tertiary) amino / amine.
(iii)
• Direction: Increase in pH.
• Reason: The nitrogen atom can accept a proton from water, so the group behaves as a base.
(iv)
The alkene (\(\text{C}=\text{C}\)) double bond should be circled. 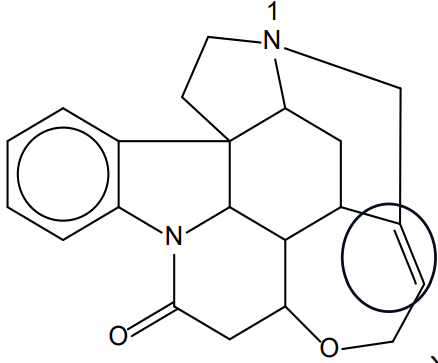
(v)
\(7\).
(b)
• Moles of strychnine \(= 48.73 / 334.4 = 0.1457 \text{ mol}\).
• Moles of product \(= 0.1457 / 2 = 0.07286 \text{ mol}\).
• Molar mass of product \(= (2 \times 334.4) + 98.1 = 766.9 \text{ g mol}^{-1}\).
• Theoretical yield \(= 0.07286 \times 766.9 = 55.87 \text{ g}\).
• Percentage yield \(= (51.41 / 55.87) \times 100 = \mathbf{92.00\%}\).
