(a) The box gives some methods used in the separation of mixtures.

Use words from the box to answer these questions.
(i) Identify the method used to obtain pure water from sea water.
(ii) Identify the method used to separate the dyes in a food colouring.
(iii) Identify the method used to obtain ethanol from a mixture of ethanol and water.
(b) Complete the sentences by writing a suitable word in each blank space.
When salt is added to water and stirred until no more will , a saturated solution forms.
The salt is the .
The water is the .
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(a): States of matter — part (b) – covers the terms solvent, solute, solution, and saturated solution.
▶️ Answer/Explanation
(a)(i) simple distillation
Explanation: Simple distillation is used to separate a pure solvent (like water) from a solution (like sea water) by boiling the solution and then condensing the vapor. It works because water has a much lower boiling point than the dissolved salts.
(a)(ii) chromatography
Explanation: Chromatography, specifically paper chromatography, is used to separate mixtures of dyes or pigments based on their different solubilities and attractions to the paper and solvent. Each dye travels a different distance, allowing them to be identified.
(a)(iii) fractional distillation
Explanation: Fractional distillation is used to separate two or more miscible liquids with different boiling points, such as ethanol (boiling point ~78°C) and water (boiling point 100°C). A fractionating column allows for more efficient separation than simple distillation.
(b)
M1: dissolve
M2: solute
M3: solvent
Explanation: A saturated solution is one in which no more solute can dissolve at a given temperature. The substance that dissolves (salt) is the solute, and the substance it dissolves in (water) is the solvent.
The diagram shows the positions of some elements in the Periodic Table.
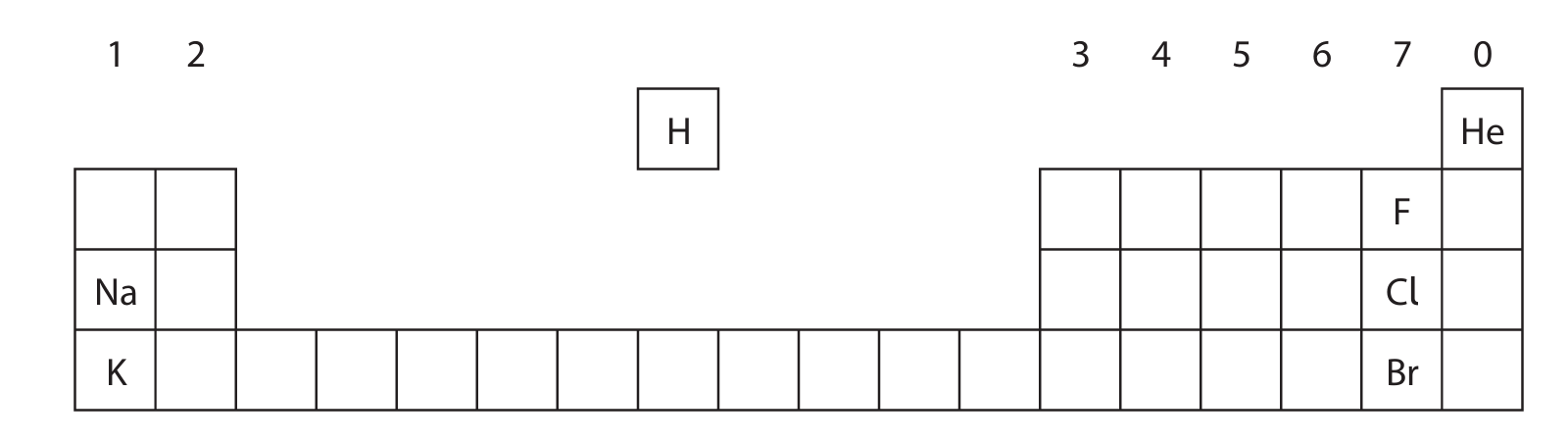
(a) Use symbols from this table to answer these questions. Each symbol may be used once, more than once or not at all.
(i) Give the symbol of a metal.
(ii) Give the symbol of a noble gas.
(iii) Give the symbol of a liquid at room temperature.
(iv) Give the symbols of the two elements in Period 3.
(b) Deduce the electronic configuration of Na.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(d): The Periodic Table (Periods and Groups) — part (a)(iv), identifying Period 3
• 1(c): Atomic structure — part (b), electronic configuration
• 2(b): Group 7 (Halogens) — part (a)(iii), physical states of halogens
• 1.21: Identify metal/non-metal from position in Periodic Table — part (a)(i)
• 1.19: Deduce electronic configurations of first 20 elements — part (b)
▶️ Answer/Explanation
(a)(i) Na or K (Accept sodium/potassium)
Explanation: Metals are found on the left side and centre of the Periodic Table. Sodium (Na) and Potassium (K) are both Group 1 alkali metals.
(a)(ii) He (Accept helium)
Explanation: Noble gases are in Group 0 (Group 18). Helium (He) is the only noble gas shown in the table snippet.
(a)(iii) Br (Accept bromine)
Explanation: At room temperature, bromine (Br) is the only element shown that exists as a liquid. Chlorine (Cl) is a gas, and the others are solids.
(a)(iv) Na and Cl (Accept sodium and chlorine)
Note: Reject “bromide” or “chloride” as these refer to ions/compounds, not elements.
Explanation: Period 3 refers to the third row of the Periodic Table. From the given table, sodium (Na) and chlorine (Cl) are in Period 3.
(b) 2.8.1 (Accept 2,8,1 or 2 8 1; also allow a diagram showing the electron configuration)
Explanation: Sodium (Na) has atomic number 11. Its electron configuration is: 2 electrons in the first shell, 8 in the second, and 1 in the third, represented as \(2.8.1\).
This question is about alkenes and alkanes.
(a) Complete the table by giving the missing information about the alkene with the molecular formula \( C_3H_6 \)
| Molecular formula | \( C_3H_6 \) |
|---|---|
| Name | |
| Empirical formula | |
| General formula | |
| Displayed formula |
(b) Alkenes are unsaturated compounds.
(i) State what is meant by the term unsaturated.
(ii) Describe a test to show that a compound is unsaturated.
(c) When the alkane methane reacts with chlorine, the products are chloromethane (\( CH_3Cl \)) and hydrogen chloride gas.
(i) Give a chemical equation for this reaction.
(ii) What is the name of this type of reaction?
- A) addition
- B) decomposition
- C) neutralisation
- D) substitution
(iii) State the condition needed for this reaction to occur.
(d) When ethane reacts with chlorine, one of the products of the reaction has the formula \( C_2H_4Cl_2 \).
There are two isomers with this formula.
(i) State what is meant by the term isomers.
(ii) Draw the displayed formulae of the two isomers with the formula \( C_2H_4Cl_2 \).
| Isomer 1 | Isomer 2 |
|---|---|
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4.3 & 4.4: Homologous series, functional groups, isomerism, IUPAC nomenclature — parts (a), (d)(i)
• 4.23 & 4.25: Alkenes – functional group, unsaturation — part (b)
• 4.28: Test for unsaturation (bromine water) — part (b)(ii)
• 4.6 & 4.22: Reaction types (substitution) – halogenation of alkanes — part (c)
• 4.5 & 4.21: Drawing structural/displayed formulae — parts (a), (d)(ii)
▶️ Answer/Explanation
(a) Completing the Table:
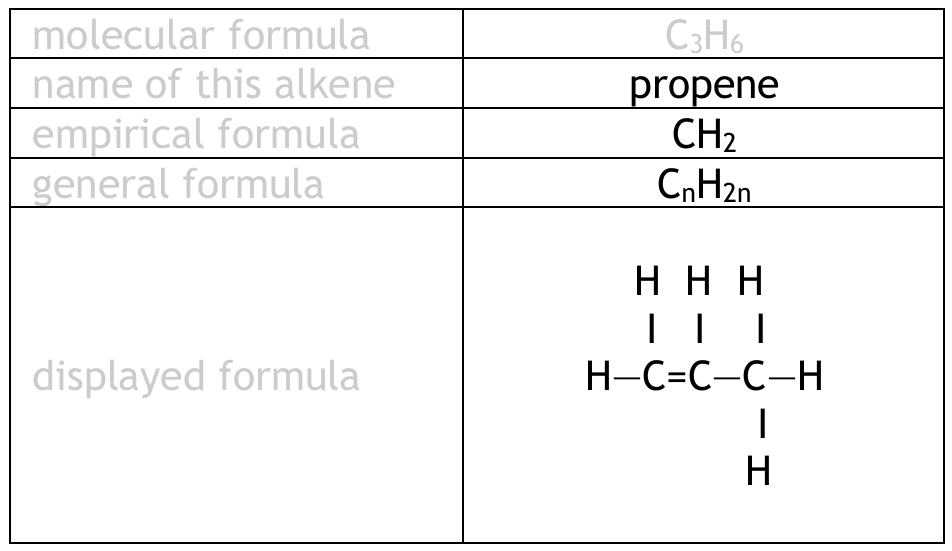
Explanation: Propene is the alkene with three carbons. The empirical formula is the simplest whole-number ratio (\(CH_2\)). The general formula for alkenes is \(C_nH_{2n}\). The displayed formula shows the carbon-carbon double bond, which is characteristic of alkenes.
(b)(i) Unsaturated:
• (A compound) containing a carbon-carbon double bond.
Explanation: Unsaturated hydrocarbons, like alkenes, have at least one C=C bond, meaning they can undergo addition reactions.
(b)(ii) Test for Unsaturation:
• Add bromine water (orange/brown) to the compound.
• If unsaturated, the bromine water decolourises (turns colourless).
Explanation: The double bond in alkenes undergoes an addition reaction with bromine, forming a colourless dibromoalkane. This is a standard qualitative test.
(c)(i) Equation:
• \( CH_4 + Cl_2 \rightarrow CH_3Cl + HCl \)
Explanation: This shows the substitution of one hydrogen atom in methane with a chlorine atom.
(c)(ii) Reaction Type:
• D (substitution)
Explanation: A hydrogen atom is replaced (substituted) by a chlorine atom. This is characteristic of the reaction between alkanes and halogens.
(c)(iii) Condition:
• Ultraviolet (UV) light / sunlight.
Explanation: The reaction requires UV light to provide the energy to break the Cl–Cl bond, initiating the free-radical chain reaction.
(d)(i) Isomers:
• Molecules with the same molecular formula
• but different structural/displayed formulae (or different arrangement of atoms)
Explanation: Isomerism is a key concept in organic chemistry. Structural isomers have the same atoms connected in different ways.
(d)(ii) Displayed Formulae of \( C_2H_4Cl_2 \) Isomers:
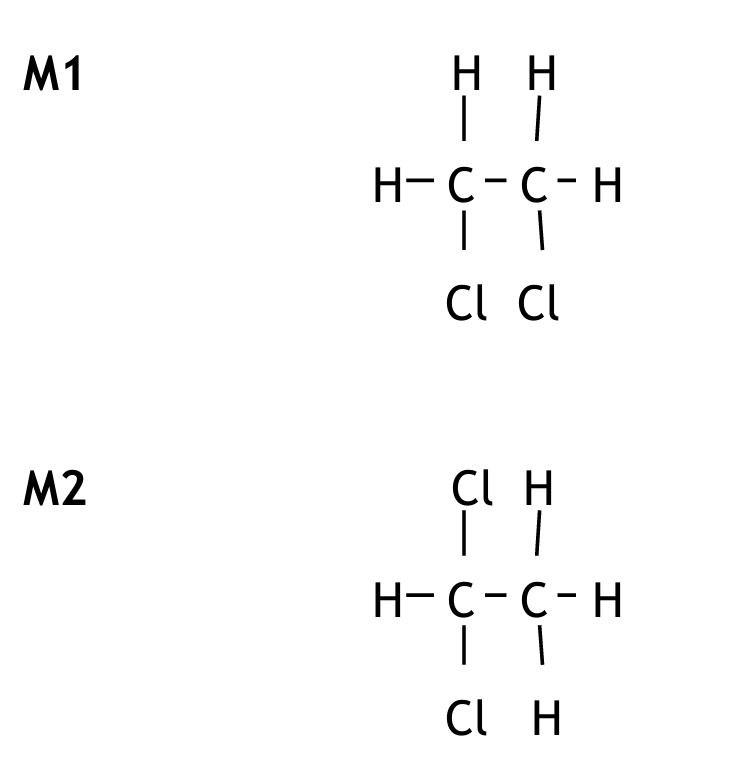
Explanation: Isomer 1 has both chlorine atoms on the same carbon. Isomer 2 has one chlorine on each carbon. These are structural isomers.
A solution of hydrogen peroxide decomposes when a catalyst of manganese(IV) oxide is added. The products of the reaction are water and oxygen.
(a) Complete the chemical equation for this reaction.
…… \( \text{H}_2\text{O}_2 \) → …… \( \text{H}_2\text{O} \) + …… \( \text{O}_2 \)
(b) Give a test for oxygen.
(c) State the reason for adding a catalyst.
(d) A student investigates how changing the concentration of the hydrogen peroxide solution affects the rate of this reaction. She uses the apparatus shown.
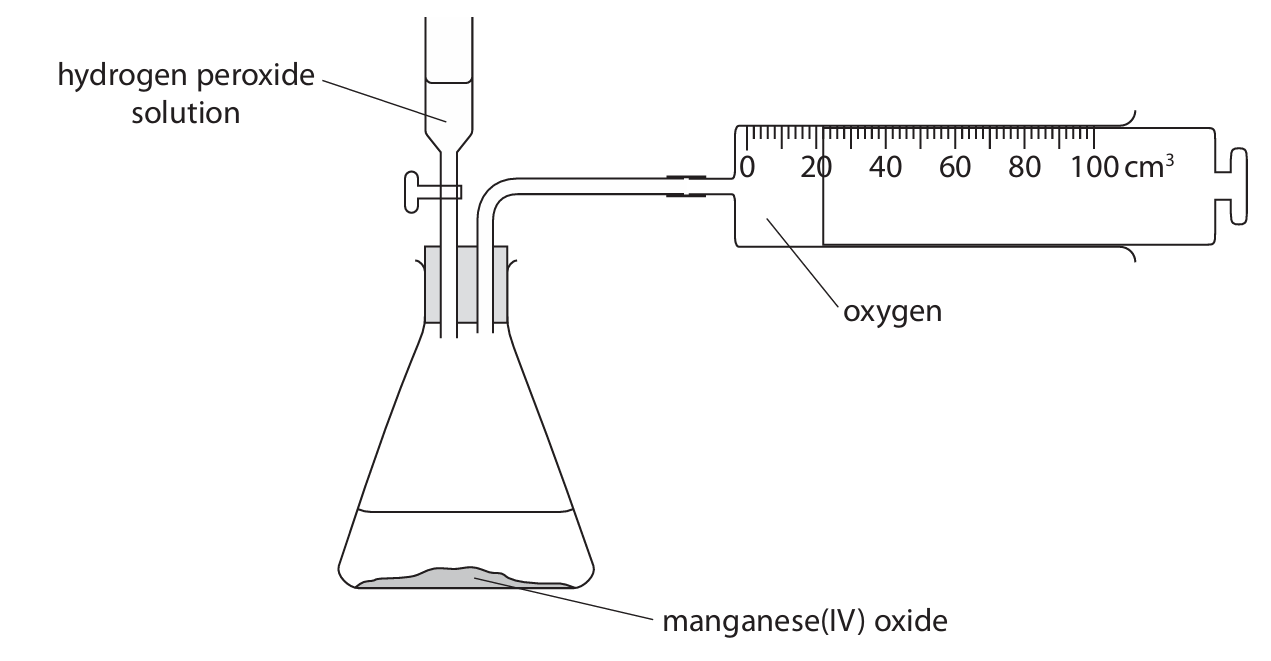
The student records the volume of oxygen that collects every 2 minutes for 16 minutes. The table shows her results.
| Time in minutes | 0 | 2 | 4 | 6 | 8 | 10 | 12 | 14 | 16 |
| Volume of oxygen in cm³ | 0 | 22 | 38 | 50 | 55 | 69 | 76 | 80 | 80 |
(i) Plot the student’s results on the grid.
(ii) Draw a circle on the grid around the anomalous result.
(iii) Draw a curve of best fit through the points, ignoring the anomalous result.
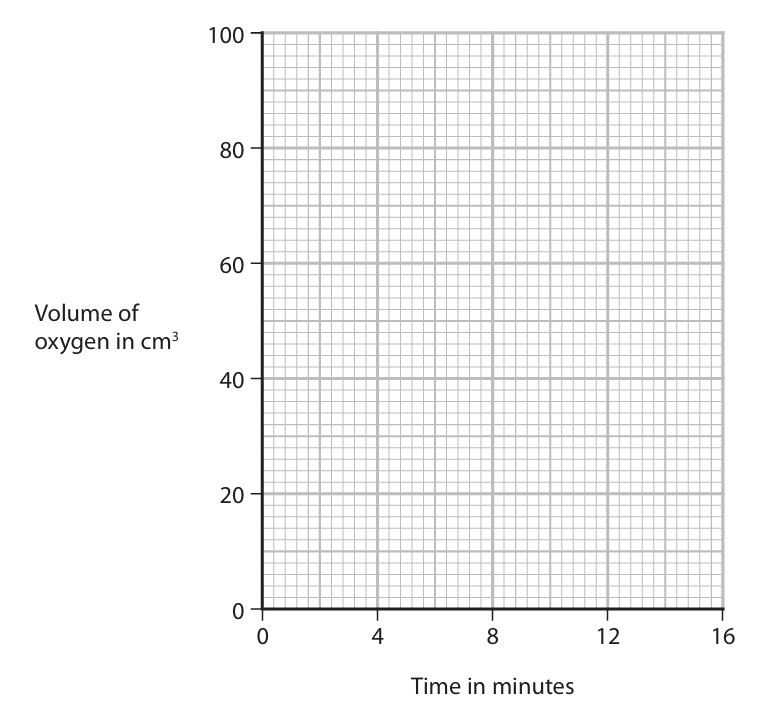
(iv) Suggest a mistake that the student might have made to cause the anomalous result.
(v) Determine the volume of oxygen collected during the first 3 minutes. Show on your graph how you obtain your answer.
(e) The student repeats the experiment using hydrogen peroxide solution of half the concentration of the original solution. She keeps the volume of the hydrogen peroxide solution and all other conditions the same.
(i) Draw on the grid the curve you would expect the student to obtain.
(ii) Explain how using hydrogen peroxide solution of half the concentration affects the rate of the reaction. Refer to particle collision theory in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(h): Chemical tests — part (b)
• 3(b): Rates of reaction — parts (c), (d), (e)
• 3(b): Catalysts — part (c)
• 3(b): Effect of concentration on rate — parts (d), (e)
• 3(b): Particle collision theory — part (e)(ii)
• Appendix 4: Mathematical skills (Graphs) — parts (d)(i, ii, iii, v), (e)(i)
▶️ Answer/Explanation
(a) \( 2\ \text{H}_2\text{O}_2 \rightarrow 2\ \text{H}_2\text{O} + \text{O}_2 \)
Balance the equation. Two molecules of hydrogen peroxide decompose to produce two molecules of water and one molecule of oxygen. Accept multiples or fractions (e.g., \( \text{H}_2\text{O}_2 \rightarrow \text{H}_2\text{O} + \frac{1}{2}\text{O}_2 \)).
(b) Relights a glowing splint/spill
Oxygen supports combustion. A glowing wooden splint will burst into flames when inserted into oxygen.
(c) Speeds up/increases rate of the reaction
A catalyst provides an alternative reaction pathway with a lower activation energy, increasing the frequency of successful collisions without being consumed. IGNORE references to lowering activation energy in this answer.
(d)(i)-(iii)
Graph plotted from the table, with a circle around the point at 8 minutes (55 cm³) as it is anomalous. A smooth curve of best fit is drawn through the other points. The curve should show an increasing gradient at first (fast reaction) that gradually levels off as the reaction finishes (oxygen volume becomes constant at 80 cm³).
(d)(iv) Took the reading too soon/before 8 minutes (OR misread the volume of oxygen)
The volume at 8 minutes (55 cm³) is lower than the trend suggests. This could be because the student recorded the volume before the 8-minute mark, or misread the measuring cylinder.
(d)(v) 29 or 30 cm³
A vertical line should be drawn from 3 minutes on the time axis up to the curve of best fit. A horizontal line from this intersection to the volume axis gives the volume. The expected value from a correctly drawn graph is 29 or 30 cm³. The student must show the construction lines on their graph.
(e)(i)
A curve should be drawn on the same grid that is less steep than the original curve, showing a slower initial rate. The curve should level off at 40 cm³ (half the final volume of the original experiment, as the concentration of hydrogen peroxide is halved, but the volume of solution is the same, so half the amount of reactant is present).
(e)(ii) An explanation that links the following three points:
1. The reaction is slower.
2. There are fewer hydrogen peroxide particles/molecules in the same volume (OR particles are further apart/less crowded).
3. This leads to fewer collisions per unit time between reactant particles (OR less frequent collisions).
Halving the concentration reduces the number of reactant particles in a given volume. According to collision theory, the rate of reaction depends on the frequency of successful collisions. Fewer particles mean fewer collisions, hence a slower rate. IGNORE “less chance of a collision”.
(a) The diagram shows the displayed formula of the organic compound methanol, CH\(_3\)OH
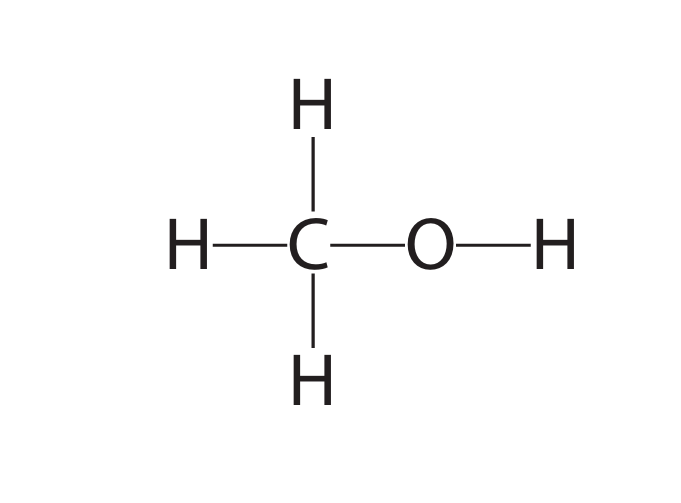
(a) (i) Determine the number of atoms in one molecule of methanol.
(a) (ii) State why methanol is not a hydrocarbon.
(b) The atoms in methanol are held together by covalent bonds.
(b) (i) State what is meant by the term covalent bond.
(b) (ii) Draw a dot-and-cross diagram to show the bonding in a molecule of methanol. Show only the outer electrons of each atom.
(c) Another organic compound has the percentage composition by mass:
\[ C = 38.7\% \quad H = 9.7\% \quad O = 51.6\% \]
(c) (i) Calculate the empirical formula of this compound.
(c) (ii) The relative molecular mass (\( M_r \)) of the compound is 62. Determine the molecular formula of the compound.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(g): Covalent Bonding — parts (b)(i), (b)(ii)
• 1(e): Chemical Formulae, Equations and Calculations — parts (c)(i), (c)(ii)
• 1.32 & 1.33: Empirical and Molecular Formulae — part (c)
• 4(e): Alcohols — context for methanol
▶️ Answer/Explanation
(a)(i) 6 (six)
Explanation: The molecular formula is CH\(_3\)OH. This represents 1 Carbon, 4 Hydrogens (3 from CH\(_3\) and 1 from OH), and 1 Oxygen atom. Total = 1 + 4 + 1 = 6 atoms.
(a)(ii) One of the following:
• Methanol does not contain only carbon and hydrogen.
• Methanol contains (an atom of) oxygen.
Explanation: A hydrocarbon is defined as a compound containing only hydrogen and carbon atoms. Methanol contains oxygen, so it is not a hydrocarbon; it is an alcohol.
(b)(i) A covalent bond is a shared pair of electrons between two atoms. Alternatively: It is an electrostatic attraction between two nuclei and the shared pair(s) of electrons between them.
Explanation: This definition is from specification point 1.44: “know that a covalent bond is formed between atoms by the sharing of a pair of electrons.”
(b)(ii) A correct dot-and-cross diagram for methanol (CH\(_3\)OH). Key features:
- Central carbon atom with four outer electrons (e.g., crosses).
- Carbon shares one pair (single bond) with oxygen (oxygen can use dots for its outer electrons).
- Carbon shares one pair with each of the three hydrogen atoms.
- Oxygen shares its remaining pair with the fourth hydrogen atom.
- Oxygen also has two lone pairs (non-bonding electrons).
Note: The diagram should show only outer-shell electrons. A typical representation has the carbon atom in the center bonded to three H atoms and one O atom. The O atom is also bonded to an H atom and has two lone pairs.
(c)(i) Step-by-step calculation for empirical formula:
- Assume 100 g of compound, so masses are: C = 38.7 g, H = 9.7 g, O = 51.6 g.
- Convert masses to moles:
Moles of C = \( \frac{38.7}{12.0} = 3.225 \)
Moles of H = \( \frac{9.7}{1.0} = 9.7 \)
Moles of O = \( \frac{51.6}{16.0} = 3.225 \) - Divide by the smallest number of moles (3.225):
C : \( \frac{3.225}{3.225} = 1 \)
H : \( \frac{9.7}{3.225} \approx 3 \)
O : \( \frac{3.225}{3.225} = 1 \) - Ratio C : H : O = 1 : 3 : 1
Empirical formula = CH\(_3\)O
(c)(ii) Step to find molecular formula:
- Calculate empirical formula mass of CH\(_3\)O:
C: 12.0, H\(_3\): 3.0, O: 16.0 → Total = 12 + 3 + 16 = 31.0 - Divide relative molecular mass by empirical formula mass:
\( \frac{62}{31} = 2 \) - Multiply empirical formula by this integer:
(CH\(_3\)O) × 2 = C\(_2\)H\(_6\)O\(_2\)
Molecular formula = C\(_2\)H\(_6\)O\(_2\)
Explanation: This compound could be ethylene glycol, a common organic compound.
This question is about elements in Group 7 of the Periodic Table and their compounds.
(a) (i) Give the name of this group of elements.
(a) (ii) State the colour of chlorine gas.
(a) (iii) Give a test for chlorine gas.
(b) Give a test to show that a solution contains iodide ions.
(c) A student compares the reactivity of the elements bromine, chlorine and iodine. He mixes these pairs of solutions and observes the reactions that occur.
- chlorine solution and potassium bromide solution
- bromine solution and potassium iodide solution
Explain how the reactions can be used to show the order of reactivity of the three elements. Include the colour change that the student would observe in each reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(h): Inorganic Chemistry – Chemical tests – parts (a)(iii), (b)
▶️ Answer/Explanation
(a)(i) halogens
Detail: This is the collective name for Group 7 elements (e.g., fluorine, chlorine, bromine, iodine, astatine).
(a)(ii) pale green
Detail: Chlorine gas has a distinctive pale yellow-green colour.
(a)(iii)
• Test: Place damp (or moist) blue litmus paper into the gas.
• Result: The litmus paper is bleached (turns white).
Detail: Chlorine is a powerful bleaching agent. It reacts with water to form hypochlorous acid, which bleaches the dye in litmus paper. The paper may initially turn red (as chlorine forms an acidic solution) before being bleached.
(b)
• Test:
- Add dilute nitric acid to the test solution.
- Then add silver nitrate solution.
• Result: A pale yellow precipitate forms.
Detail: Nitric acid is added first to remove any carbonate or hydroxide ions that might interfere by forming precipitates with silver ions. Silver nitrate reacts with iodide ions \( (I^-) \) to form insoluble silver iodide \( (AgI) \), which is pale yellow. For chloride ions \( (Cl^-) \) the precipitate is white \( (AgCl) \), and for bromide ions \( (Br^-) \) it is cream \( (AgBr) \).
(c)
The order of reactivity is: chlorine > bromine > iodine.
• First reaction: Chlorine solution + potassium bromide solution.
– Observation: The solution turns orange (or yellow-brown).
– Explanation: Chlorine \( (Cl_2) \) is more reactive than bromine. It displaces bromine from potassium bromide \( (KBr) \).
– Chemical Equation: \( Cl_2(aq) + 2KBr(aq) \rightarrow 2KCl(aq) + Br_2(aq) \)
– The orange/yellow-brown colour is due to the formation of dissolved bromine \( (Br_2) \). This shows chlorine is more reactive than bromine.
• Second reaction: Bromine solution + potassium iodide solution.
– Observation: The solution turns brown.
– Explanation: Bromine \( (Br_2) \) is more reactive than iodine. It displaces iodine from potassium iodide \( (KI) \).
– Chemical Equation: \( Br_2(aq) + 2KI(aq) \rightarrow 2KBr(aq) + I_2(aq) \)
– The brown colour is due to the formation of dissolved iodine \( (I_2) \). This shows bromine is more reactive than iodine.
• Conclusion: Chlorine displaces bromine, and bromine displaces iodine. Therefore, the reactivity order is chlorine > bromine > iodine.
Detail: This is an example of a displacement (or redox) reaction. A more reactive halogen will displace a less reactive halogen from its halide salt in solution. Reactivity decreases down Group 7 due to increasing atomic size and decreasing ability to gain an electron (electronegativity).
A student uses the reaction between zinc and dilute sulfuric acid to prepare some zinc sulfate crystals.
(a) (i) Complete the equation for this reaction by giving the correct state symbols.
\[ \text{Zn} + \text{H}_2\text{SO}_4 \rightarrow \text{ZnSO}_4 + \text{H}_2 \]
(a) (ii) State what would be observed during this reaction.
(b) (i) The student adds excess zinc to a beaker of dilute sulfuric acid. Explain why it is necessary to add excess zinc.
(b) (ii) Draw a diagram of the apparatus the student should use to remove the unreacted zinc and collect the zinc sulfate solution.
(c) The student obtains a pure, dry sample of zinc sulfate crystals. The formula of zinc sulfate crystals is \(\text{ZnSO}_4\cdot7\text{H}_2\text{O}\).
(c) (i) Calculate the relative molecular mass (\(M_r\)) of zinc sulfate crystals.
(c) (ii) The student uses 0.0200 mol of dilute sulfuric acid in her preparation. Show that the maximum mass of zinc sulfate crystals that the student could obtain is about 6 g.
(c) (iii) The student obtains a mass of 4.28 g of zinc sulfate crystals. Calculate the percentage yield of the zinc sulfate crystals. Give your answer to three significant figures.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(g): Acids, bases and salt preparations — parts (a), (b)(i), (b)(ii) practical procedure
• 1(e): Chemical formulae, equations and calculations — part (c)(i) relative formula mass, (c)(ii) reacting masses, (c)(iii) percentage yield
• 1.30: Calculate percentage yield — part (c)(iii)
• 2.39: Describe an experiment to prepare a pure, dry sample of a soluble salt — parts (b)(i), (b)(ii) experimental method
▶️ Answer/Explanation
(a)(i) \(\text{Zn (s)} + \text{H}_2\text{SO}_4 \text{ (aq)} \rightarrow \text{ZnSO}_4 \text{ (aq)} + \text{H}_2 \text{ (g)}\)
Details: Zinc is a solid (s), sulfuric acid is aqueous (aq), zinc sulfate forms an aqueous solution (aq), and hydrogen is a gas (g).
(a)(ii) Effervescence / bubbles / fizzing.
Details: The reaction produces hydrogen gas, which is seen as bubbles forming on the surface of the zinc and rising through the solution.
(b)(i) An explanation that links the following two points:
• To make sure all of the acid reacts.
• So that a pure zinc sulfate solution is obtained / pure zinc sulfate crystals are obtained.
Details: Adding excess zinc ensures that the sulfuric acid is the limiting reactant. This prevents any residual acid from contaminating the product and ensures all sulfate ions are used to form zinc sulfate.
(b)(ii) Diagram should show:
• A filter funnel containing filter paper.
• A suitable container to collect the filtrate (e.g., beaker, conical flask, evaporating basin).
Details: The mixture of zinc sulfate solution and excess solid zinc is poured through the filter paper. The solution (filtrate) passes through, while the unreacted zinc solid (residue) is left on the filter paper.
(c)(i)
Calculation:
\(M_r = 65 + 32 + (4 \times 16) + (7 \times 18)\)
\(= 65 + 32 + 64 + 126\)
\(= 287\)
Therefore, \(M_r = 287\).
Details: Atomic masses: Zn = 65, S = 32, O = 16, H = 1. For \(\text{ZnSO}_4\cdot7\text{H}_2\text{O}\): ZnSO₄ part = 65+32+64=161; 7H₂O part = 7×(2+16)=126; Total = 161+126=287.
(c)(ii)
From the balanced equation: \(\text{Zn} + \text{H}_2\text{SO}_4 \rightarrow \text{ZnSO}_4 + \text{H}_2\)
1 mole of \(\text{H}_2\text{SO}_4\) produces 1 mole of \(\text{ZnSO}_4\cdot7\text{H}_2\text{O}\).
Moles of acid used = 0.0200 mol.
Therefore, maximum moles of crystals = 0.0200 mol.
Mass = moles × \(M_r\) = \(0.0200 \times 287 = 5.74 \, \text{g}\).
This is approximately 6 g.
Details: The calculation shows the theoretical maximum yield based on the limiting reactant (sulfuric acid). Using the \(M_r\) from part (i), the mass is 5.74 g, which rounds to about 6 g as stated.
(c)(iii)
Percentage yield = \(\frac{\text{actual mass}}{\text{theoretical mass}} \times 100\%\)
Theoretical mass from (ii) = 5.74 g.
Actual mass = 4.28 g.
Percentage yield = \(\frac{4.28}{5.74} \times 100\%\)
\(= 0.7456 \times 100\% = 74.6\%\) (to three significant figures).
Details: The yield is less than 100% due to practical losses during steps like filtration, transfer, and crystallization. 74.6% is a reasonable yield for a school laboratory preparation.
(a) A piece of magnesium ribbon is ignited and placed in a gas jar of oxygen. The equation for the reaction is:
\[ 2Mg + O_2 \rightarrow 2MgO \]
(i) Give two observations that would be made in this reaction.
(ii) State why this is an oxidation reaction.
(b) A second piece of magnesium ribbon is ignited and placed in a gas jar of carbon dioxide. A very exothermic reaction occurs, forming magnesium oxide and carbon.
(i) State what is meant by the term exothermic.
(ii) Give the chemical equation for this reaction.
(iii) A fire starts in a warehouse where magnesium is stored. Suggest why it would not be suitable to use a carbon dioxide fire extinguisher to put out this fire.
(c) A student uses this apparatus to find the mass of magnesium oxide that forms when a known mass of magnesium is heated.
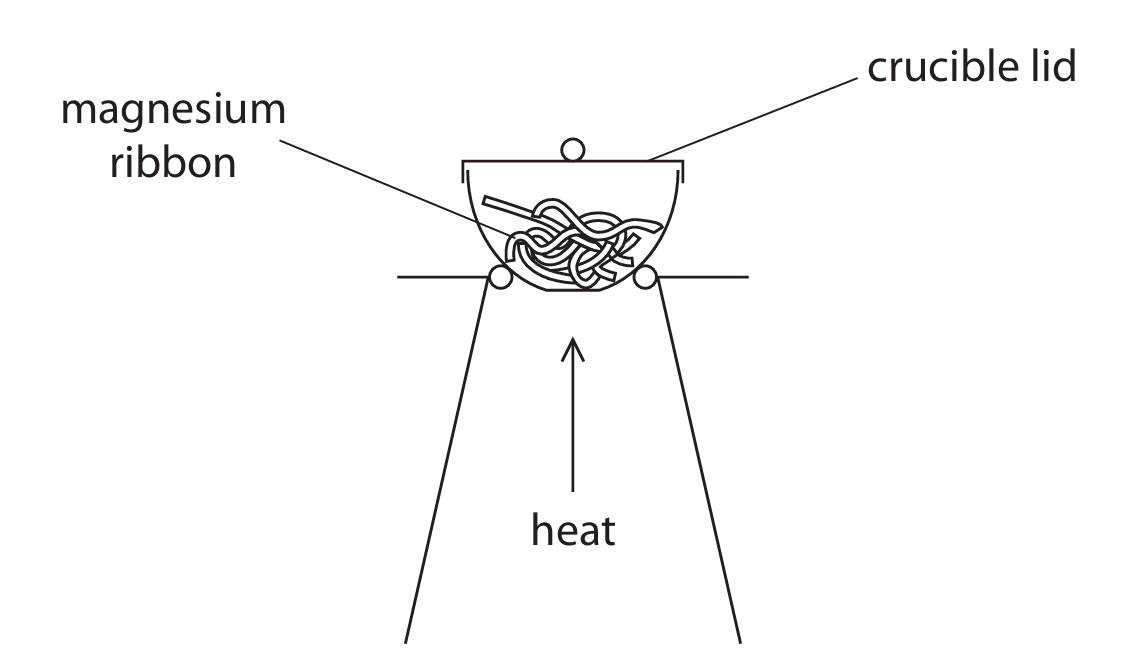
This is his method.
• find the mass of the crucible and lid
• place some magnesium ribbon in the crucible
• find the mass of the crucible, lid and magnesium
• heat the crucible with the lid on for a few minutes
• find the mass of the crucible, lid and magnesium oxide
Using this method, the mass of magnesium oxide formed is less than expected.
Explain two changes that the student should make to his method to obtain a mass of magnesium oxide closer to the expected mass.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(a): Energetics — Definition of exothermic
• 2(c): Gases in the atmosphere — Combustion of elements in oxygen, unusual reaction with CO₂
• 1(e): Chemical formulae, equations and calculations — Writing equations, determining formulae by experiment
• 1.36 Practical: Determine the formula of a metal oxide by combustion — Part (c)
▶️ Answer/Explanation
(a)(i)
1. (Burns with a) bright white flame / intense white light.
2. Formation of a white powder / white solid / white ash.
Detailed Answer: Magnesium burns vigorously in oxygen with a characteristic brilliant white flame, producing magnesium oxide as a fine white solid (ash).
(a)(ii) Magnesium gains oxygen.
Alternative answers: Magnesium loses electrons / The oxidation state (of Mg) increases from 0 to +2.
Detailed Answer: Oxidation is defined as the gain of oxygen or loss of electrons. In this reaction, each magnesium atom gains an oxygen atom to form MgO.
(b)(i) A reaction that gives out / releases heat (energy) to the surroundings.
Detailed Answer: In an exothermic reaction, the energy released from forming new bonds is greater than the energy needed to break the old bonds, resulting in a net release of thermal energy.
(b)(ii) \[ 2Mg + CO_2 \rightarrow 2MgO + C \]
Detailed Answer: The magnesium is so reactive that it can reduce carbon dioxide, removing the oxygen to form magnesium oxide and leaving behind carbon (soot).
(b)(iii) The magnesium would continue to burn in the carbon dioxide / The reaction is highly exothermic, releasing a large amount of heat.
Detailed Answer: Carbon dioxide extinguishers work by smothering fires, but magnesium can strip oxygen from CO₂ itself. Using a CO₂ extinguisher on a magnesium fire would therefore be ineffective and could even provide more reactant for the vigorous, heat-producing reaction.
(c) An explanation giving two linked changes. The student should:
First Change:
• M1: Lift and replace the lid repeatedly during heating.
• M2: (To allow) oxygen/air to enter the crucible (to ensure all magnesium reacts completely).
Second Change:
• M3: Reheat and reweigh / heat to constant mass.
• M4: (To make sure) that all the magnesium has reacted / that the reaction is complete.
Detailed Answer: The initial low yield is likely because the lid was kept on, limiting the oxygen supply and causing incomplete combustion (some unreacted Mg may remain). Lifting the lid allows oxygen in. Furthermore, a single heating may not be enough; reheating until the mass no longer increases ensures the conversion of all Mg to MgO.
This question is about some compounds of the elements in Group 4 of the Periodic Table.
(a) When carbon dioxide dissolves in water, a weak acid forms.
(a) (i) Which of these could be the pH of this weak acid?
- A) 1
- B) 5
- C) 7
- D) 9
(a) (ii) Which of these is a correct statement about acids?
- A) acids contain OH⁻ ions
- B) acids are electron donors
- C) acids are proton acceptors
- D) acids are proton donors
(b) When lead(II) carbonate is heated, lead(II) oxide and carbon dioxide form.
(b) (i) Give the name of this type of reaction.
(b) (ii) Complete the equation for this reaction.
\[ \text{PbCO}_3 \rightarrow \text{……………} + \text{……………} \]
(c) Silicon dioxide, SiO₂, and silicon(IV) chloride, SiCl₄, are both covalently bonded compounds.
The table shows the melting and boiling points of these two compounds, and the physical state of silicon dioxide at room temperature.
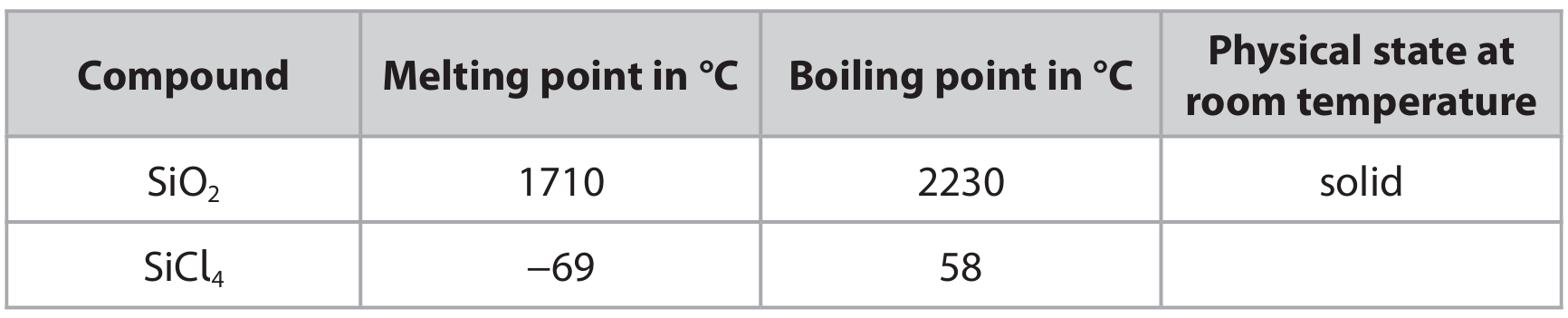
(c) (i) Complete the table by giving the physical state of silicon(IV) chloride at room temperature.
(c) (ii) Explain, in terms of structure and bonding, why silicon dioxide has a much higher melting point than silicon(IV) chloride.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(g): Acids, bases and salt preparations — part (a)(ii) acid definition
• 1(g): Covalent bonding — part (c)(ii) structure and bonding
• 1(e): Chemical formulae, equations and calculations — part (b)(ii) completing an equation
• 2(d): Reactivity series — part (b)(i) thermal decomposition
▶️ Answer/Explanation
(a)(i) B (5)
- A (pH 1) is incorrect as it is the pH of a strong acid.
- C (pH 7) is incorrect as it is the pH of a neutral solution.
- D (pH 9) is incorrect as it is the pH of a weak alkali.
- Weak acids (like carbonic acid formed from CO₂) typically have a pH between 4 and 6.
(a)(ii) D (acids are proton donors)
- A is incorrect because alkalis (bases) contain OH⁻ ions, not acids.
- B is incorrect because acids are not typically described as electron donors in the Brønsted-Lowry definition used at IGCSE.
- C is incorrect because bases are proton acceptors, not acids.
- This is a key definition from the Brønsted-Lowry theory of acids and bases (Specification point 2.36).
(b)(i) (Thermal) decomposition
This is a reaction where a single compound breaks down into two or more simpler substances when heated (Specification point 2.12).
(b)(ii) \[ \text{PbCO}_3 \rightarrow \text{PbO} + \text{CO}_2 \]
This is a balanced thermal decomposition equation. Metal carbonates decompose to form a metal oxide and carbon dioxide gas.
(c)(i) Liquid
Since the boiling point of SiCl₄ (58 °C) is above room temperature (approx. 20-25 °C) and its melting point (-69 °C) is far below, it exists as a liquid at room temperature.
(c)(ii)
An explanation linking structure and bonding:
- Silicon dioxide (SiO₂) has a giant covalent (macromolecular) structure (Specification point 1.49).
- In this structure, many strong covalent bonds hold all the atoms together in a rigid, continuous network.
- To melt SiO₂, a large amount of energy is required to break these numerous strong covalent bonds.
- Silicon(IV) chloride (SiCl₄) has a simple molecular structure (Specification point 1.47).
- The atoms within each SiCl₄ molecule are held by strong covalent bonds, but the forces between the molecules (intermolecular forces) are weak.
- To melt SiCl₄, only a small amount of energy is needed to overcome these weak intermolecular forces, not to break the covalent bonds within the molecules.
- Therefore, SiO₂ has a much higher melting point than SiCl₄ because breaking the strong covalent bonds in a giant structure requires significantly more energy than overcoming the weak forces between simple molecules.
This directly assesses understanding of Specification points 1.47, 1.49, and 1.50, comparing simple vs. giant covalent structures.
A student uses this apparatus to investigate the reaction between potassium hydroxide solution and dilute hydrochloric acid.
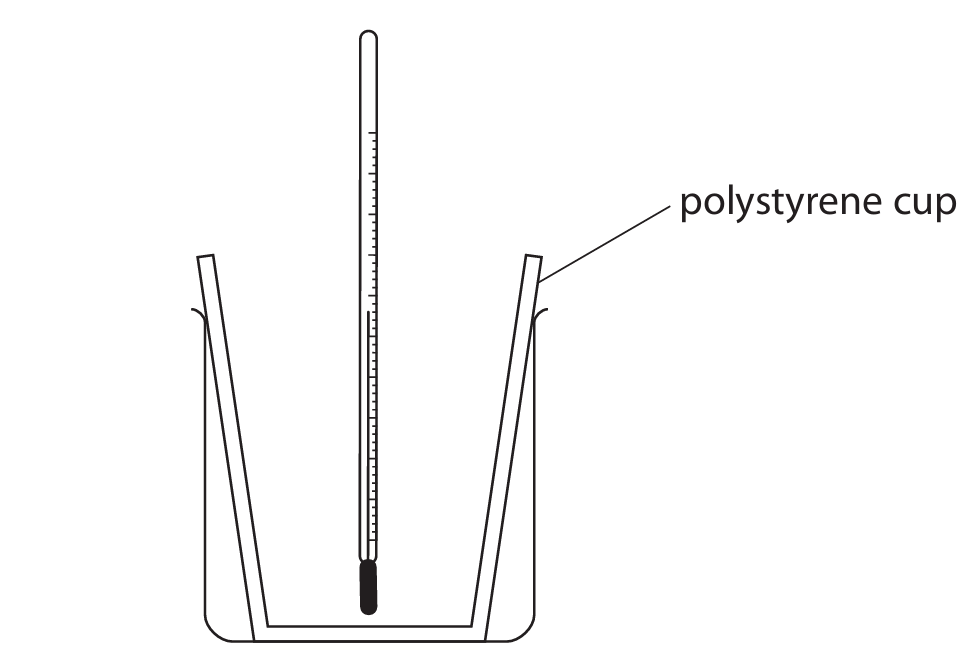
This is her method.
- pour 25 cm³ of potassium hydroxide solution into a polystyrene cup and record the temperature of the solution
- pour 25 cm³ of dilute hydrochloric acid into a measuring cylinder and record the temperature of the acid
- add the acid to the polystyrene cup and stir the mixture
- record the highest temperature reached
(a) (i) Give a word equation for the reaction between potassium hydroxide and hydrochloric acid.
(a) (ii) Explain why the student needs to stir the mixture.
(b) The table gives the temperatures of the solutions before the student mixes them.

Calculate the mean (average) temperature of the two solutions.
(c) The student repeats the experiment on a different day, using 25 cm³ of potassium hydroxide solution and 25 cm³ of dilute hydrochloric acid.
The thermometer shows the highest temperature reached at the end of the experiment.
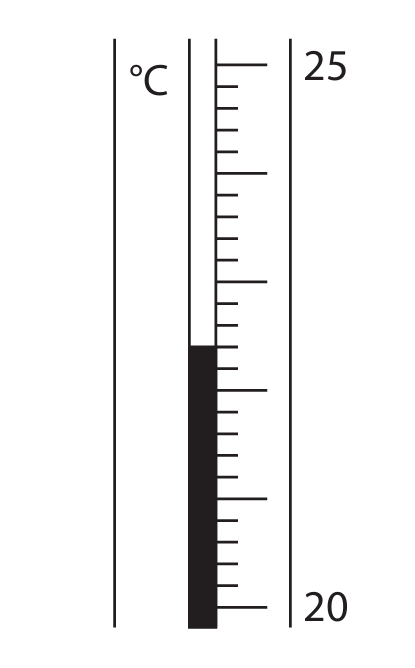
(i) Complete the table by giving the missing information. Give both temperatures to the nearest 0.1 °C.

(ii) Show that the heat energy change, \( Q \), in the student’s experiment is about 1100 J.
(for the mixture, \( c = 4.2 \, \text{J g}^{-1} °C^{-1} \) )
(mass of 1.0 cm³ of mixture = 1.0 g)
(iii) The student uses 0.020 mol of potassium hydroxide in his experiment.
Calculate the enthalpy change (\( \Delta H \)) in kJ/mol, for 1.0 mol of potassium hydroxide.
Include a sign in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(a): Energetics — parts (a)(ii), (b), (c)(i), (c)(ii), (c)(iii)
• 3.3: Calculate the heat energy change from a measured temperature change using the expression \( Q = mc\Delta T \) — parts (b), (c)(ii)
• 3.4: Calculate the molar enthalpy change (\( \Delta H \)) from the heat energy change, \( Q \) — part (c)(iii)
▶️ Answer/Explanation
(a)(i)
potassium hydroxide + hydrochloric acid → potassium chloride + water
This is a neutralisation reaction, forming a salt (potassium chloride) and water.
(a)(ii)
• To mix the two solutions thoroughly so that all the reactant particles come into contact.
• To ensure the mixture has a uniform temperature throughout.
• This allows the heat energy from the exothermic reaction to be released quickly and uniformly, ensuring an accurate measurement of the maximum temperature change.
(b)
Mean temperature = \( \frac{17.8 + 18.4}{2} = \frac{36.2}{2} = 18.1 \)
mean temperature = 18.1 °C
The average of the two initial temperatures is used as the starting temperature for calculating the temperature rise.
(c)(i)
From the given temperature rise of 5.2 °C and the provided answer key:
mean temperature at start in °C = 17.2
temperature at end in °C = 22.4
These values are consistent: \( 22.4 – 17.2 = 5.2 \).
(c)(ii)
Volume of mixture = \( 25 + 25 = 50 \) cm³
Since mass of 1 cm³ = 1 g, total mass, \( m = 50 \) g
Specific heat capacity, \( c = 4.2 \, \text{J g}^{-1} °C^{-1} \)
Temperature rise, \( \Delta T = 5.2 \) °C
\( Q = mc\Delta T \)
\( Q = 50 \times 4.2 \times 5.2 \)
\( Q = 1092 \, \text{J} \approx 1100 \, \text{J} \) (to about 1100 J)
The calculation shows the heat energy change is approximately 1100 J.
(c)(iii)
Heat change for 0.020 mol KOH = 1092 J (from (c)(ii))
Heat change for 1 mol KOH = \( \frac{1092}{0.020} = 54600 \, \text{J} \)
Convert to kJ: \( 54600 \, \text{J} = 54.6 \, \text{kJ} \)
The reaction is exothermic (temperature increased), so \( \Delta H \) is negative.
\( \Delta H = \mathbf{-54.6 \, \text{kJ mol}^{-1}} \)
Enthalpy change is negative because heat is released to the surroundings.
