The diagram shows some pieces of apparatus.
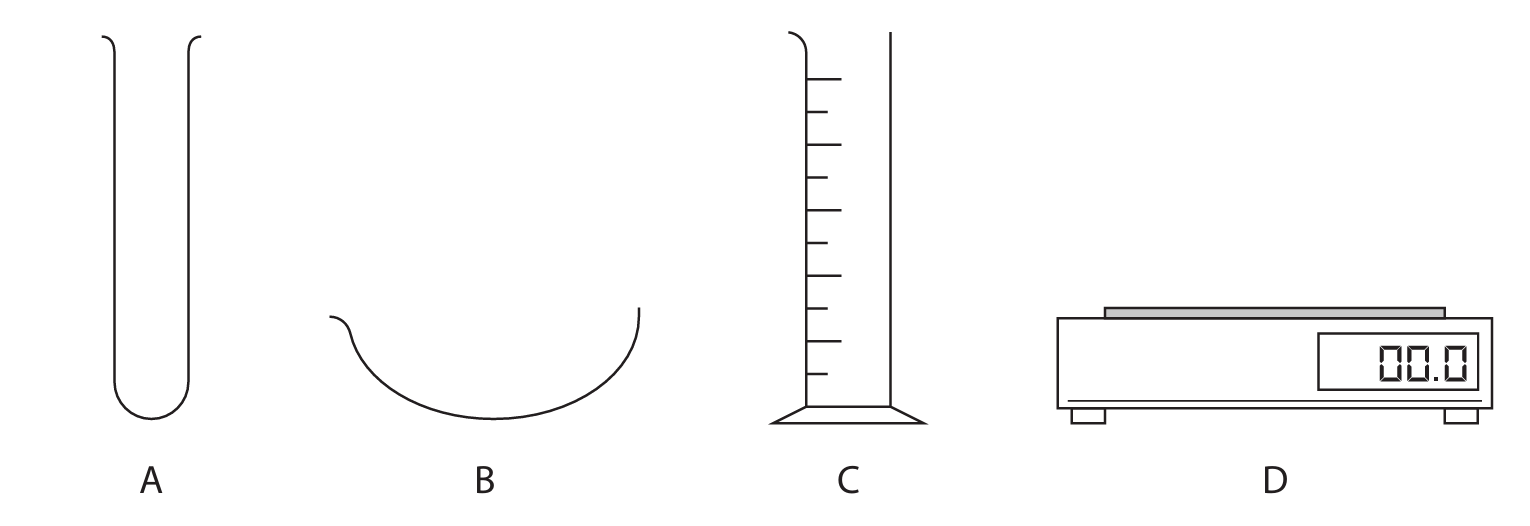
(a) Complete the table by giving the name of each piece of apparatus.
(b) Which piece of apparatus can be used to measure the volume of a liquid?
- A) A
- B) B
- C) C
- D) D
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(g): Acids, bases and salt preparations – Preparation of pure, dry samples of salts
• Experimental Skills (General): Selection and use of appropriate laboratory apparatus.
▶️ Answer/Explanation
(a)
| Letter | Name |
|---|---|
| A | test tube / boiling tube |
| B | evaporating basin / dish |
| C | measuring cylinder |
| D | (top pan) balance / scale(s) |
Details: This tests knowledge of standard laboratory glassware and equipment. A test tube is for holding small quantities of liquids or solids. An evaporating basin is used for heating solutions to evaporate solvent, often during crystallization. A measuring cylinder is used to measure volumes of liquid, though with less precision than a volumetric pipette or burette. A balance is used to measure mass.
(b) C (measuring cylinder)
Explanation:
• A is incorrect because a test tube cannot accurately measure a specific volume of liquid; it has no volume scale.
• B is incorrect because an evaporating basin is used for heating and evaporation, not for measuring volume.
• D is incorrect because a balance measures mass (in grams), not volume (in cm³ or dm³).
• C is correct because a measuring cylinder is specifically designed and graduated to measure approximate volumes of liquids.
Thallium, Tl, is an element in Group 3 and Period 6 of the Periodic Table. The atomic number of thallium is 81.
(a) How many electrons are there in the outer shell of an atom of thallium?
- A) 3
- B) 6
- C) 13
- D) 81
(b) A thallium ion has a charge of 3+.
How many electrons are there in this thallium ion?
- A) 3
- B) 78
- C) 81
- D) 84
(c) A sample of thallium contains two isotopes. The table shows the mass number and percentage abundance of each isotope in the sample.
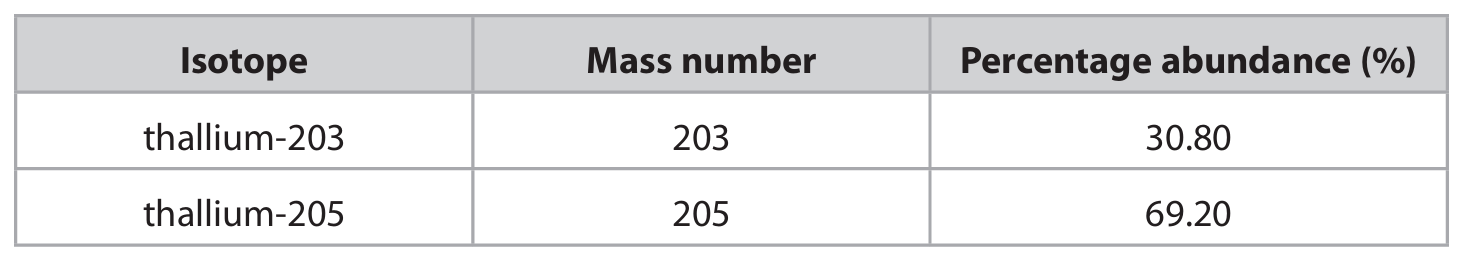
(c) (i) Give the number of protons and the number of neutrons in one atom of the thallium-205 isotope.
(c) (ii) Calculate the relative atomic mass of this sample of thallium. Give your answer to one decimal place.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(c): Atomic Structure — parts (c)(i), (c)(ii)
• 1.16 & 1.17: Isotopes and calculating relative atomic mass (part c(ii))
▶️ Answer/Explanation
(a) A (3)
Explanation: The atomic number is 81, so a neutral atom has 81 electrons. As it is in Group 3, it has 3 electrons in its outer shell.
B is incorrect as there are not 6 electrons in the outer shell of a thallium atom.
C is incorrect as there are not 13 electrons in the outer shell of a thallium atom.
D is incorrect as 81 is the total number of electrons in a thallium atom, not the number in the outer shell.
(b) B (78)
Explanation: A neutral thallium atom has 81 electrons. A \( \text{Tl}^{3+} \) ion has lost 3 electrons, so \( 81 – 3 = 78 \) electrons remain.
A is incorrect as there are not 3 electrons in a thallium ion.
C is incorrect as 81 is the number of electrons in a thallium atom, not a thallium ion.
D is incorrect as there are not 84 electrons in a thallium ion.
(c) (i)
Number of protons: 81
Number of neutrons: 124
Explanation: The atomic number (protons) is 81 for all thallium isotopes. For thallium-205, the mass number is 205. Number of neutrons = mass number – atomic number = \( 205 – 81 = 124 \).
(c) (ii)
The relative atomic mass is calculated as: \[ \text{Ar} = \frac{(203 \times 30.8) + (205 \times 69.2)}{100} \] Step-by-step calculation:
M1: Calculate the sum of (mass number × percentage abundance):
\( (203 \times 30.8) + (205 \times 69.2) = 6252.4 + 14186 = 20438.4 \)
M2: Divide by 100:
\( 20438.4 \div 100 = 204.384 \)
M3: Round to one decimal place:
\( 204.4 \)
Answer: 204.4
The diagram shows a fractionating column used to separate crude oil into fractions.
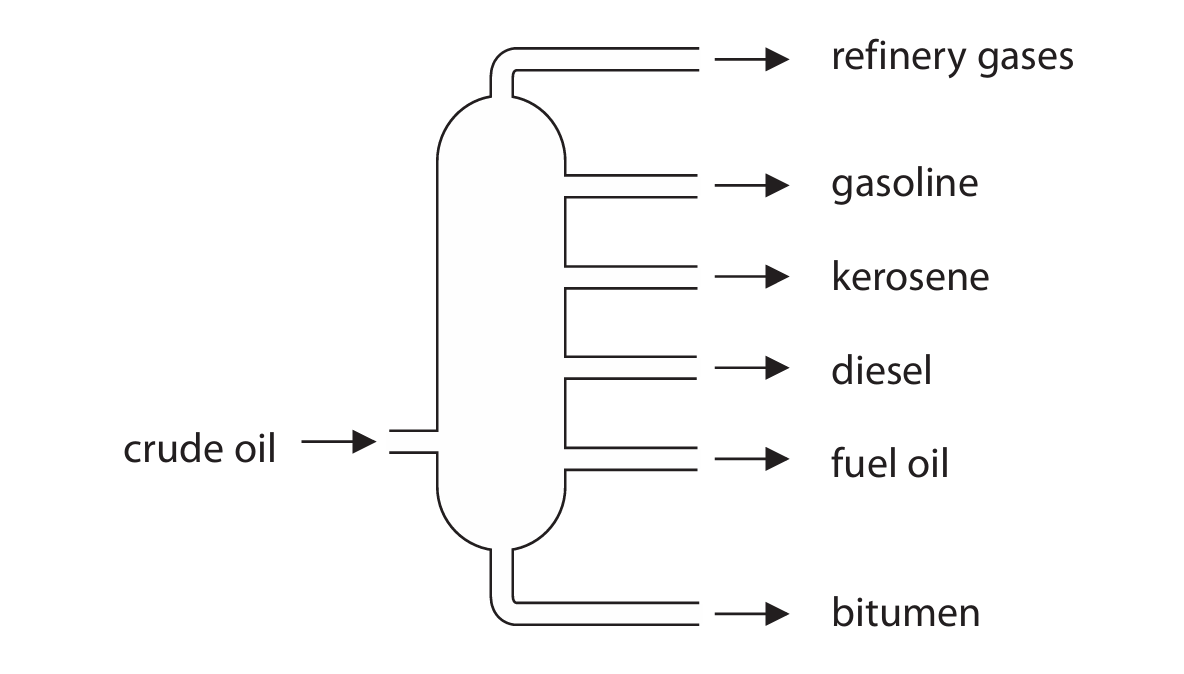
(a) (i) Give a use for bitumen and a use for gasoline.
(a) (ii) Explain why bitumen is collected at the bottom of the fractionating column and gasoline is collected near the top of the fractionating column.
(b) (i) State the conditions needed for cracking.
(b) (ii) Dodecane (\( C_{12}H_{26} \)) can be cracked to produce an alkane and two alkenes. Complete the equation by giving the formulae of the two alkenes.
\[ C_{12}H_{26} \xrightarrow{\text{}} C_7H_{16} + C_3H_6 + C_2H_4 \]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b): Crude oil (cracking) — part (b)
• 4(d): Alkenes — part (b)(ii)
• 4.8–4.10, 4.17–4.18: Fractional distillation, uses of fractions, cracking — whole question
▶️ Answer/Explanation
(a)(i)
• Use for bitumen: (surfacing) roads / (surfacing) roofs / waterproofing.
• Use for gasoline: petrol / fuel for cars or vehicles / fuel for cooking.
(a)(ii)
An explanation that links the following two points:
• The column is cooler near the top than at the bottom (temperature decreases up the column).
• Gasoline has a lower boiling point than bitumen, so it vaporises and rises higher before condensing and being collected near the top. Conversely, bitumen has a high boiling point, so it remains liquid and is collected at the bottom.
(b)(i)
• Catalyst: alumina (\( Al_2O_3 \)) or silica (\( SiO_2 \)) or aluminosilicates / zeolites.
• Temperature: \( 600–700\,^{\circ}\text{C} \).
(b)(ii)
\[ C_{12}H_{26} \rightarrow C_7H_{16} + C_2H_4 + C_3H_6 \]
• The two alkenes are ethene (\( C_2H_4 \)) and propene (\( C_3H_6 \)).
Cracking breaks long-chain hydrocarbons into shorter, more useful molecules, including alkenes which are valuable for making polymers and other chemicals.
This question is about some of the alkali metals and their compounds.
(a) When a teacher drops a small piece of sodium into a trough of cold water, she observes bubbles of gas.
Give two other observations that would be made when sodium reacts with cold water.
(b) Lithium reacts with fluorine to form the compound lithium fluoride.
(i) Give a chemical equation for this reaction.
(ii) Give a test to show that lithium fluoride contains lithium ions.
(iii) Draw diagrams to show the arrangement of the electrons in a lithium ion and in a fluoride ion. Include the charge on each ion.

(c) The table shows the electronic configurations of sodium and potassium.
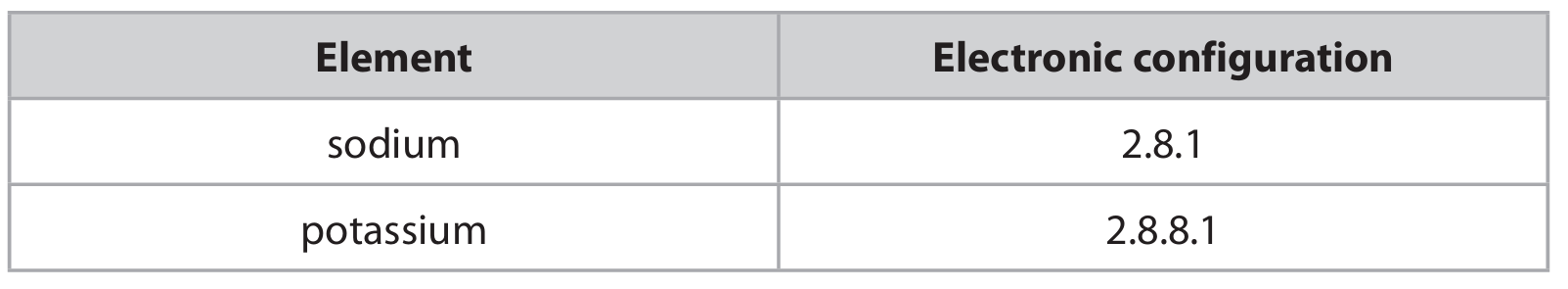
Explain, in terms of their electronic configurations, why potassium is more reactive than sodium.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(h): Chemical tests — part (b)(ii)
• 1(f): Ionic bonding — parts (b)(i) and (b)(iii)
• 1(e): Chemical formulae, equations and calculations — part (b)(i)
▶️ Answer/Explanation
(a) Any two of the following observations:
- (Sodium) floats / moves on the surface (of the water).
- (Sodium) melts / forms a ball.
- (Sodium) gets smaller / disappears.
- A white trail is formed.
Bubbles of gas (effervescence) was already mentioned in the question, so it cannot be given as an answer.
(b)(i) \( 2Li + F_2 \rightarrow 2LiF \)
A balanced equation is required. The state symbols (s) for solid lithium, (g) for fluorine gas, and (s) for solid lithium fluoride are not required for the mark but are chemically correct.
(b)(ii) Perform a flame test. A red/crimson/scarlet flame colour indicates the presence of lithium ions (\( Li^+ \)).
The flame test is a standard test for Group 1 metal ions. Lithium gives a distinctive red colour.
(b)(iii)
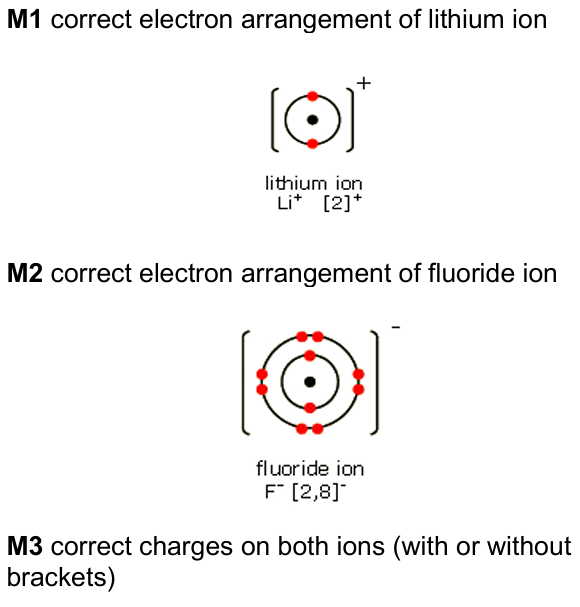
(c) An explanation linking the following points:
- In potassium, the outer electron is in the fourth shell / is further from the nucleus / the atom has a larger atomic radius compared to sodium.
- There are more inner electron shells in potassium, providing greater shielding from the positive nuclear charge.
- This results in a weaker electrostatic attraction between the nucleus and the outer electron in potassium.
- Therefore, the outer electron in potassium is more easily lost in reactions, making it more reactive than sodium.
Reactivity in Group 1 increases down the group because the single outer electron is held less strongly as atomic size and shielding increase, making it easier to lose in redox reactions (e.g., with water).
This question is about the metal aluminium.
(a) (i) Draw a labelled diagram to represent the structure and bonding in a metal.
(a) (ii) Explain why a metal conducts electricity.
(b) Aluminium is used to make cans for drinks.

Give two properties of aluminium that make it suitable for this use.
(c) Aluminium is extracted from aluminium oxide \( \text{(Al}_2\text{O}_3) \) by electrolysis. The electrolyte is aluminium oxide dissolved in molten cryolite.
(c) (i) State why aluminium cannot be extracted by heating aluminium oxide with carbon.
(c) (ii) Aluminium is produced at the negative electrode. The ionic half-equation for the reaction is:
\( \text{Al}^{3+} + 3\text{e}^- \rightarrow \text{Al} \)
State why this is a reduction reaction.
(c) (iii) Complete the ionic half-equation for the reaction at the positive electrode.
\( \_\_\_\_\_\_\_ \rightarrow \_\_\_\_\_\_\_ + \_\_\_\_\_\_\_ \)
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(e): Extraction and uses of metals — parts (b), (c)(i), (c)(ii), (c)(iii)
• 1(i): Electrolysis — parts (c)(i), (c)(ii), (c)(iii)
▶️ Answer/Explanation
(a) (i) A labelled diagram showing:
- At least three rows of at least three cations/atoms in a regular arrangement (grid of ‘+’ signs).
- Surrounded by a ‘sea’ or cloud of delocalised electrons (labelled as such).
Example diagram description: A regular lattice of positive metal ions (labelled) with delocalised electrons (labelled) shown moving freely around them.
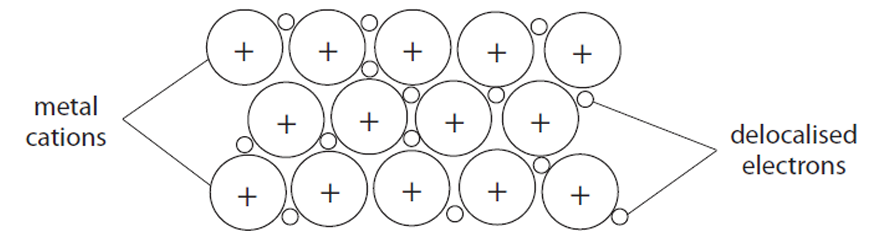
(a) (ii) An explanation that links the following:
- Metals contain delocalised/free/mobile electrons (M1).
- These electrons can move/flow through the structure when a potential difference (voltage) is applied (M2).
Note: The mark scheme specifies that any mention of ions/atoms moving scores 0.
(b) Any two of the following properties:
- Low density / lightweight: Makes cans easy to transport and handle.
- Does not react with the drink / non-toxic / corrosion-resistant: Aluminium forms a protective oxide layer, preventing it from contaminating the drink.
- Malleable / easy to shape: Can be rolled into thin sheets and pressed into the can shape.
Note: High melting point, cost, or conductivity are not relevant for this use.
(c) (i) Aluminium is more reactive than carbon / is higher in the reactivity series than carbon.
Explanation: Carbon can only displace metals less reactive than itself from their oxides. Aluminium’s position in the reactivity series makes reduction by carbon thermodynamically unfavourable.
(c) (ii) This is a reduction reaction because the aluminium ion \( \text{(Al}^{3+}) \) gains electrons.
Linked concept: Reduction is defined as a gain of electrons or a decrease in oxidation state (from +3 to 0).
(c) (iii) The completed ionic half-equation is:
\( 2\text{O}^{2-} \rightarrow \text{O}_2 + 4\text{e}^- \)
Mark scheme:
M1 for correct formulae of products \( \text{(O}_2 \) and \( \text{e}^- \)).
M2 for balancing the equation (which also requires the correct number of oxide ions).
Process: Oxide ions \( \text{(O}^{2-}) \) are oxidised (lose electrons) at the positive electrode (anode) to form oxygen gas.
A student wants to prepare sodium chloride crystals from sodium hydroxide solution and dilute hydrochloric acid. He does a titration to find the volume of dilute hydrochloric acid needed to neutralise the sodium hydroxide solution. This is his method.
- add 25.0 cm\(^3\) of sodium hydroxide solution to a conical flask
- add a few drops of phenolphthalein indicator to the conical flask
- titrate the solution with the hydrochloric acid
(a) Name a suitable piece of apparatus that the student should use to measure 25.0 cm\(^3\) of sodium hydroxide solution.
(b) (i) Give the colour of the phenolphthalein indicator in sodium hydroxide solution and in hydrochloric acid.
(b) (ii) Suggest why universal indicator is never used in a titration.
(c) (i) The student finds that 21.50 cm\(^3\) of hydrochloric acid is needed to neutralise 25.0 cm\(^3\) of sodium hydroxide solution. Describe what the student should do next to prepare a pure solution of sodium chloride.
(c) (ii) Describe how the student could obtain dry crystals of sodium chloride from the pure sodium chloride solution.
(d) The student needs 21.50 cm\(^3\) of hydrochloric acid to neutralise 25.0 cm\(^3\) of sodium hydroxide solution of concentration 0.800 mol/dm\(^3\). The equation for the reaction is: $$\text{NaOH} + \text{HCl} \rightarrow \text{NaCl} + \text{H}_2\text{O}$$ Calculate the concentration, in mol/dm\(^3\), of the hydrochloric acid.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(g): Acids, bases and salt preparations — parts (c)(i), (c)(ii)
• 1(e): Chemical formulae, equations and calculations — part (d)
▶️ Answer/Explanation
(a) Pipette (or volumetric pipette).
Explanation: A pipette is used to accurately measure a fixed volume of liquid (here, 25.0 cm³).
(b)(i) Colour in sodium hydroxide solution: pink / magenta / red. Colour in hydrochloric acid: colourless.
Explanation: Phenolphthalein is an indicator that is colourless in acidic solutions (pH < 8.2) and pink/magenta in alkaline solutions (pH > 10). Sodium hydroxide is a strong alkali, hydrochloric acid is a strong acid.
(b)(ii) Universal indicator has a range of colours / no clear (colour change at the) end point / difficult to determine which shade of green is pH 7.
Explanation: Universal indicator shows a spectrum of colours for different pH values. For a titration, you need a sharp, distinct colour change to identify the endpoint accurately.
(c)(i) Add 21.5 cm\(^3\) of hydrochloric acid to 25 cm\(^3\) of sodium hydroxide solution (without indicator).
Explanation: The titration with indicator determined the exact volume of acid needed for neutralisation. To prepare the pure salt solution, the same volumes are mixed without the indicator to avoid contamination.
(c)(ii)
- Heat the solution to evaporate some of the water / to form a saturated solution / to crystallisation point.
- Leave the solution to cool / for crystals to form.
- Filter off the crystals.
- Dry the crystals (e.g., between filter papers / in a warm oven / leave to dry).
Explanation: This is a standard method for preparing a pure, dry sample of a soluble salt from its solution.
(d)
- Moles of NaOH: \( n = c \times V = 0.800 \, \text{mol/dm}^3 \times 0.0250 \, \text{dm}^3 = 0.0200 \, \text{mol} \).
- From the equation, NaOH and HCl react in a 1:1 ratio, so moles of HCl = 0.0200 mol.
- Volume of HCl = 21.50 cm\(^3\) = 0.02150 dm\(^3\).
- Concentration of HCl: \( c = \frac{n}{V} = \frac{0.0200}{0.02150} = 0.930 \, \text{mol/dm}^3 \) (to 3 significant figures).
Answer: 0.930 mol/dm\(^3\).
(a) Ethanol, \( \text{C}_2\text{H}_5\text{OH} \), can be oxidised to produce ethanoic acid, \( \text{CH}_3\text{COOH} \), by heating it with potassium dichromate(VI).
(i) Name one other reactant needed for this reaction to occur.
(ii) Which colour change occurs during this reaction?
- A) colourless to green
- B) green to orange
- C) orange to colourless
- D) orange to green
(b) When ethanol is burned in air, complete combustion can occur. The equation for this reaction is
\( \text{C}_2\text{H}_5\text{OH} + 3\text{O}_2 \rightarrow 2\text{CO}_2 + 3\text{H}_2\text{O} \)
This equation can also be written using displayed formulae to show all the covalent bonds in the molecules.
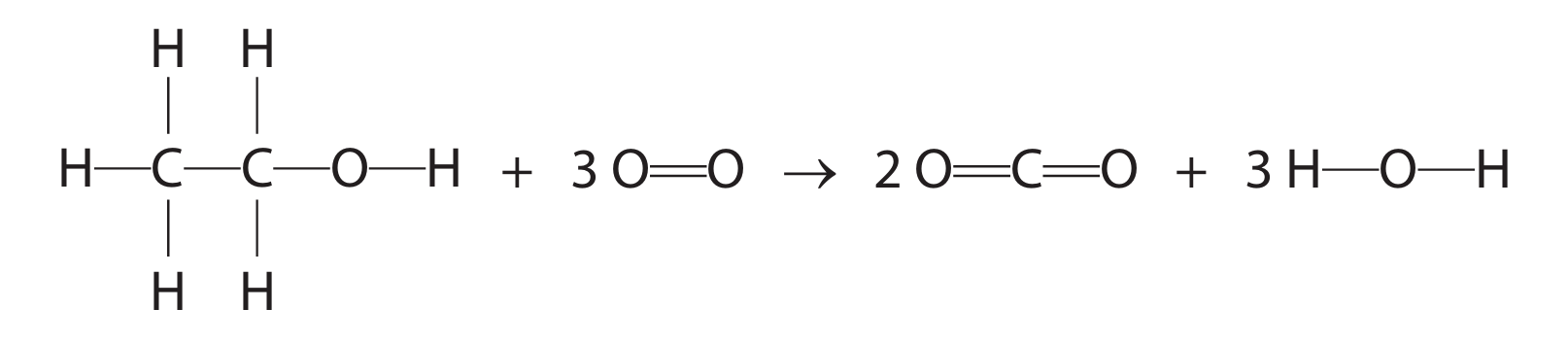
The table gives the bond energies for these bonds.
(i) Use values from the table to calculate the energy needed to break all the bonds in the reactants.
(ii) Use values from the table to calculate the energy released when all the bonds in the products are formed.
(iii) Calculate the molar enthalpy change (\(\Delta H\)) in kJ/mol, for the complete combustion of ethanol. Include a sign in your answer.
(c) Ethanol reacts with methanoic acid, \( \text{HCOOH} \), in the presence of an acid catalyst to form an ester. The equation for the reaction is
\( \text{C}_2\text{H}_5\text{OH} + \text{HCOOH} \rightleftharpoons \text{HCOOC}_2\text{H}_5 + \text{H}_2\text{O} \)
(i) Give the name of the ester that forms.
(ii) Draw the displayed formula for this ester.
(iii) When this reaction takes place in a sealed container, the reaction can reach dynamic equilibrium. Give two characteristics of a reaction at dynamic equilibrium.
(d) Methanoic acid reacts with sodium carbonate to form sodium methanoate, carbon dioxide and water. The equation for the reaction is
\( 2\text{HCOOH} + \text{Na}_2\text{CO}_3 \rightarrow 2\text{HCOONa} + \text{CO}_2 + \text{H}_2\text{O} \)
Calculate the volume, in cm³, of carbon dioxide gas produced when 2.3 g of methanoic acid reacts completely with sodium carbonate.
[\( M_r \) of \( \text{HCOOH} = 46 \); molar volume of carbon dioxide at rtp = 24 dm³]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(a): Energetics — part (b)(i), (ii), (iii)
• 4(g): Esters — part (c)(i), (c)(ii)
• 3(c): Reversible reactions and equilibria — part (c)(iii)
• 4(f): Carboxylic acids — part (d)
• 1(e): Chemical formulae, equations and calculations — part (d)
• 1(g): Covalent bonding — part (b) (bond energies)
• 2(f) & (g): Acids, alkalis, titrations & salt preparations — part (d)
▶️ Answer/Explanation
(a)(i) Sulfuric acid (or \( \text{H}_2\text{SO}_4 \))
Explanation: The oxidation of ethanol to ethanoic acid using potassium dichromate(VI) requires an acid catalyst, typically dilute sulfuric acid. This provides the acidic medium necessary for the oxidation.
(a)(ii) D (orange to green)
Explanation: Potassium dichromate(VI) ions (\( \text{Cr}_2\text{O}_7^{2-} \)) are orange in colour. During the oxidation reaction, they are reduced to chromium(III) ions (\( \text{Cr}^{3+} \)), which are green. Hence, the colour change is from orange to green.
(b)(i) Energy needed to break bonds in reactants:
Ethanol molecule: 1×C-C (346) + 5×C-H (5×412) + 1×C-O (358) + 1×O-H (463)
Oxygen molecules: 3×O=O (3×496)
Total = \( 346 + 2060 + 358 + 463 + 1488 = 4715 \) kJ
(b)(ii) Energy released when bonds in products are formed:
Carbon dioxide molecules: 4×C=O (4×743) = 2972 kJ
Water molecules: 6×O-H (6×463) = 2778 kJ
Total = \( 2972 + 2778 = 5750 \) kJ
(b)(iii) Molar enthalpy change (\(\Delta H\)):
\( \Delta H = \text{Energy absorbed} – \text{Energy released} = 4715 – 5750 = -1035 \) kJ/mol
Explanation: The negative sign indicates that the reaction is exothermic (heat is released).
(c)(i) Ethyl methanoate (or ethyl formate)
(c)(ii) Displayed formula of ethyl methanoate:
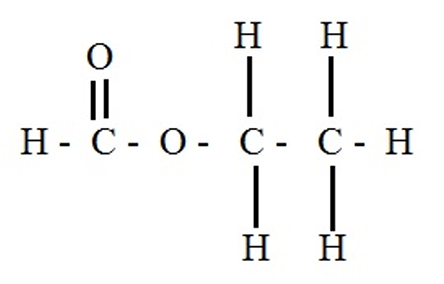
Explanation: The ester functional group (\( -\text{COO}- \)) is shown. The alkyl group from ethanol (\( \text{C}_2\text{H}_5- \)) is attached to the oxygen, and the H from methanoic acid remains on the carbonyl side.
(c)(iii) Two characteristics of a reaction at dynamic equilibrium:
1. The forward and reverse reactions occur at the same rate.
2. The concentrations of reactants and products remain constant (not necessarily equal).
(d) Volume of \( \text{CO}_2 \) produced:
Moles of \( \text{HCOOH} = \frac{2.3}{46} = 0.05 \) mol
From the equation: \( 2\text{HCOOH} \rightarrow 1\text{CO}_2 \)
Moles of \( \text{CO}_2 = \frac{0.05}{2} = 0.025 \) mol
Volume at rtp = \( 0.025 \times 24 = 0.6 \) dm³ = \( 600 \) cm³
Explanation: The molar volume at room temperature and pressure (rtp) is 24 dm³/mol. Converting dm³ to cm³ requires multiplying by 1000.
