Use the Periodic Table to help you answer this question.
(a) Identify the element with atomic number 7.
(b) Identify a solid non-metallic element in Period 3.
(c) Name an element in Group 7 that is a liquid at room temperature.
(d) State the relative atomic mass of the element that is in Group 4 and Period 4.
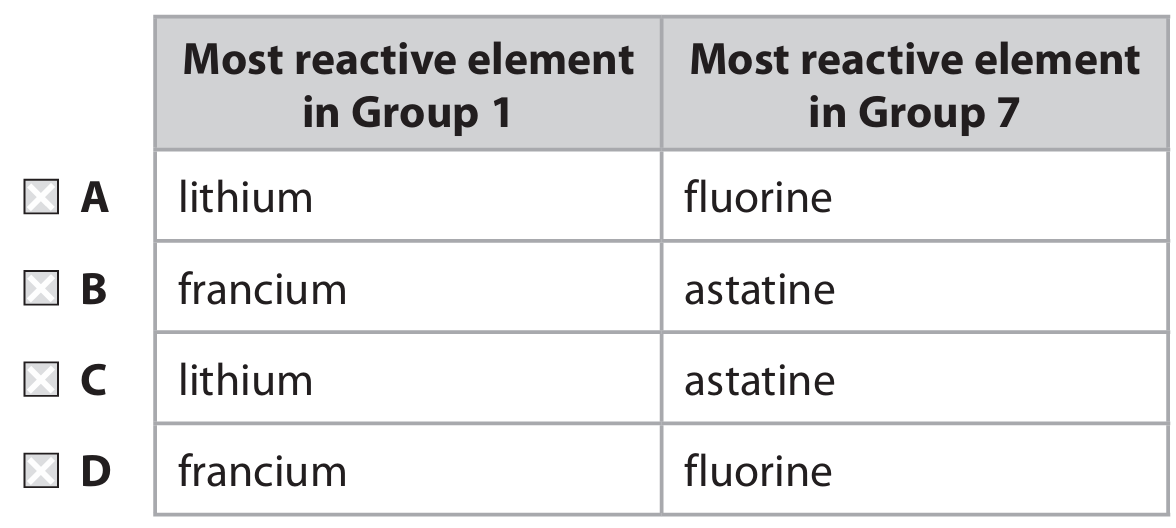
(e) Which row shows the most reactive element in Group 1 and Group 7?
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(a): Group 1 (alkali metals) — part (e) – reactivity trend in Group 1
• 2(b): Group 7 (halogens) — parts (c), (e) – physical states and reactivity trend in Group 7
▶️ Answer/Explanation
(a) nitrogen (ALLOW N or \( \text{N}_2 \))
Atomic number 7 corresponds to nitrogen on the Periodic Table.
(b) silicon/Si OR phosphorus/P OR sulfur/S
Period 3 contains the elements Na, Mg, Al, Si, P, S, Cl, Ar. The solid non-metals in this period are silicon, phosphorus, and sulfur.
(c) bromine
In Group 7 (halogens), fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid at room temperature.
(d) 73
The element in Group 4 and Period 4 is germanium (Ge). Its relative atomic mass, rounded to the nearest whole number as typically used in IGCSE, is 73.
(e) The only correct answer is D (francium / fluorine).
• A is not correct because lithium is not the most reactive element in Group 1.
• B is not correct because astatine is not the most reactive element in Group 7.
• C is not correct because lithium is not the most reactive element in Group 1 and astatine is not the most reactive element in Group 7.
Explanation: Reactivity increases down Group 1 (alkali metals), making francium the most reactive. Reactivity decreases down Group 7 (halogens), making fluorine the most reactive.
(a) The box lists words that may be used to explain the term saturated solution.

Explain, using all the words in the box, the term saturated solution.
(b) The diagram shows the apparatus a student uses to make a saturated solution.
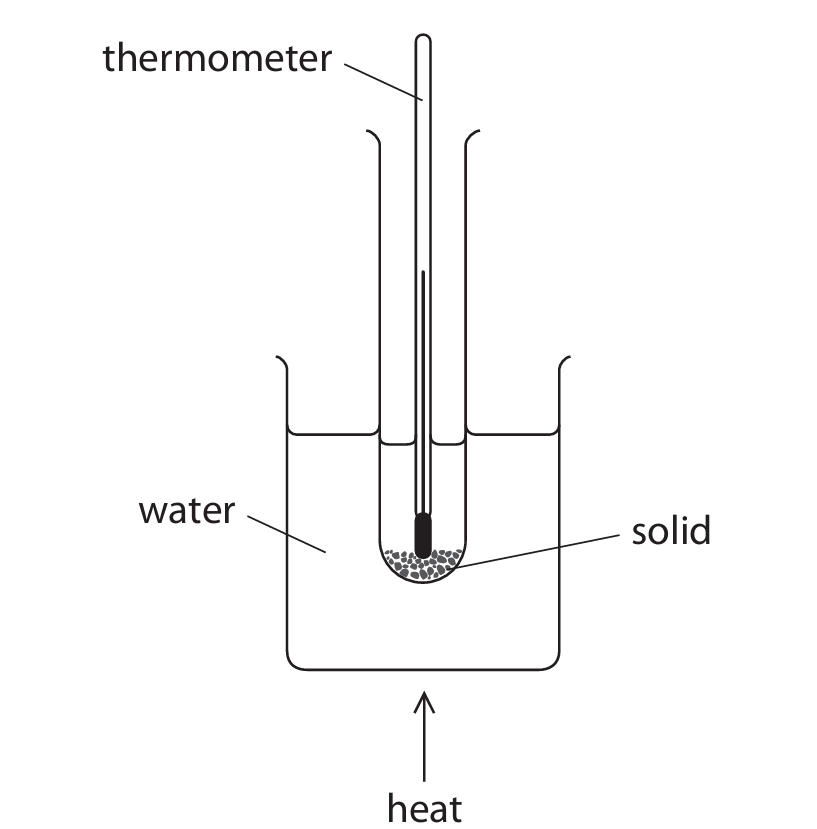
This is the student’s method.
- Step 1 add 4.5 g of solid to a boiling tube
- Step 2 measure exactly 10.0 cm³ of pure water and pour into the boiling tube
- Step 3 place the boiling tube in the beaker of water and heat gently, stirring the mixture continuously until all the solid dissolves
- Step 4 remove the boiling tube from the beaker and allow it to cool
- Step 5 record the temperature when crystals start to form in the boiling tube
The recorded temperature shows when the solution becomes saturated.
(i) Name the piece of apparatus that the student should use in Step 2 to measure exactly 10.0 cm³ of pure water.
(ii) Suggest why the boiling tube is not heated directly using a Bunsen burner in Step 3.
(iii) Suggest how the student could improve the reliability of her recorded temperature in Step 5.
(iv) In Step 5, crystals start to form at 26°C.
Calculate the solubility of the solid, in g per 100 g of water, at 26°C.
\( [1.0 \, \text{cm}^3 \text{ of pure water has a mass of } 1.0 \, \text{g}] \)
(c) The solubility curves for two solids, A and B, are shown on the grid.
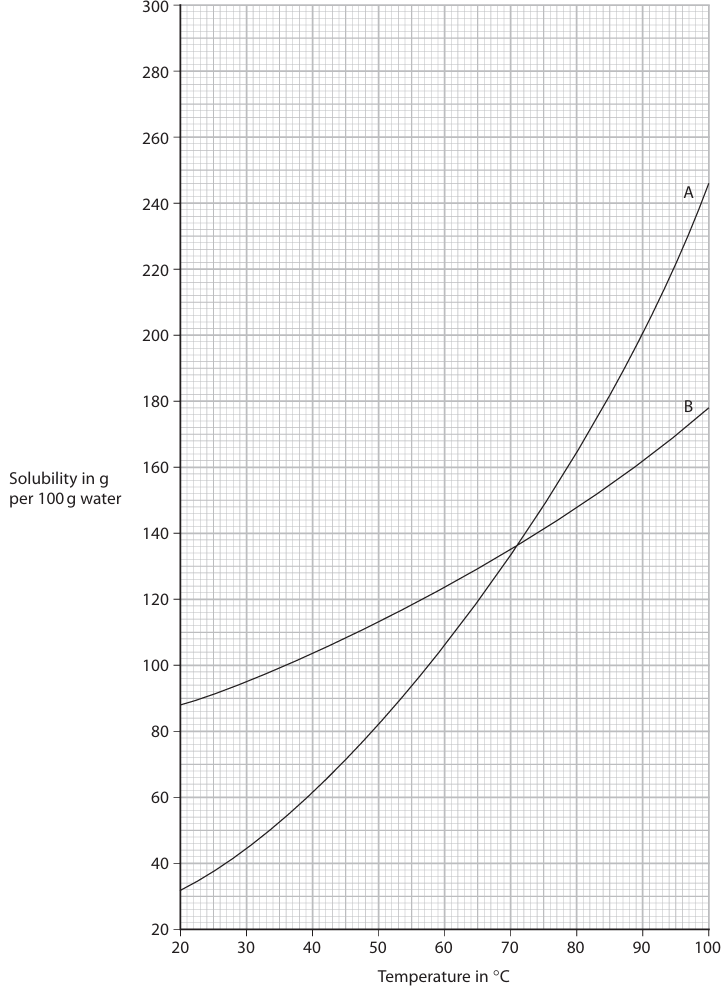
(i) State the temperature when A and B have the same solubility.
(ii) Calculate the mass of B that will dissolve in 250 g of water at 60 °C. Show your working.
(iii) Suggest why the values for the solubility of A and B may be less accurate at 95 °C than at lower temperatures.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1.4, 1.5C, 1.7C: Solubility, saturated solutions, practical investigation — parts (a), (b), (c)
• 1.6C: Plotting and interpreting solubility curves — part (c)
• 3.8 (practical): Investigate temperature changes — part (b) practical context
• Experimental Skills: Apparatus selection, method improvement, reliability — parts (b)(i), (ii), (iii)
▶️ Answer/Explanation
2 (a) A saturated solution contains the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. No more solute can dissolve at that temperature without changing conditions.
2 (b)
(i) (Volumetric) pipette OR burette.
Reason: These pieces of apparatus are designed to deliver a precise, fixed volume of liquid.
(ii) To avoid heating the solution too quickly / unevenly, which could cause boiling, violent spitting, loss of solution through evaporation, or breakage of the boiling tube. Gentle heating via a water bath provides more controlled, uniform heating.
(iii) Repeat the experiment (several times) and calculate the mean (average) temperature at which crystals form. This reduces the effect of random errors and improves reliability.
(iv)
• Mass of solid = 4.5 g
• Volume of water = 10.0 cm³ → Mass of water = 10.0 g (since 1.0 cm³ = 1.0 g)
• Solubility in g per 100 g water = \( \frac{4.5 \, \text{g}}{10.0 \, \text{g}} \times 100 = 45 \, \text{g per 100 g water} \).
2 (c)
(i) 71 °C (accept 70–72 °C). This is the intersection point of the two curves on the graph.
(ii)
• From the graph, at 60 °C, solubility of B ≈ 124 g per 100 g water.
• For 250 g water: \( \text{mass of B} = \frac{124}{100} \times 250 = 310 \, \text{g} \).
Working: \( 124 \times 2.5 = 310 \).
(iii) At high temperatures (like 95 °C), significant evaporation of the solvent (water) can occur during the experiment. This loss of water makes it seem like more solute has dissolved per 100 g of solvent than actually has, reducing the accuracy of the measured solubility value.
Sulfur dioxide (SO2) and hydrogen sulfide (H2S) are both gases. The two gases react together to form solid sulfur and water.
(a) (i) Complete the chemical equation for the reaction.
![]()
(a) (ii) State why the sulfur dioxide is reduced in the reaction.
(b) The diagram shows apparatus used to compare the speed at which particles of the two gases diffuse.
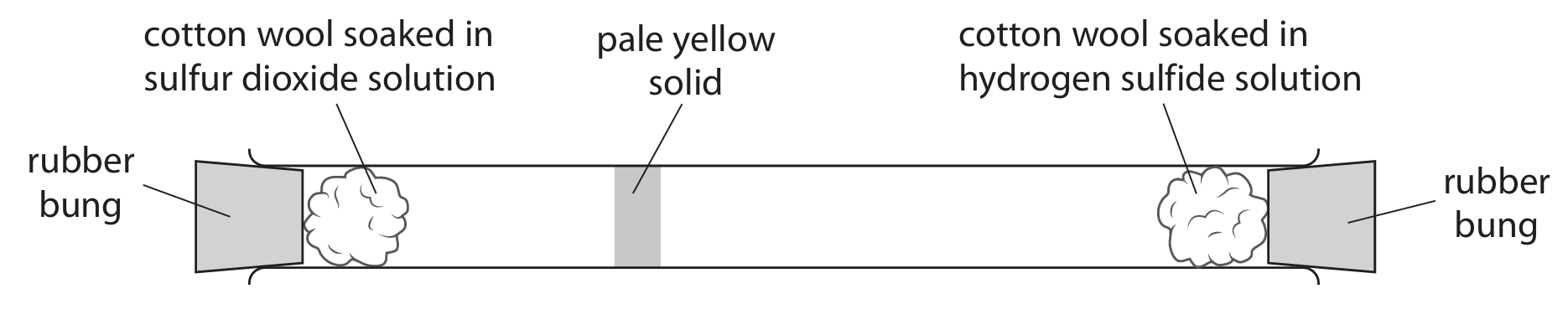
(b) (i) Explain how the diagram shows that hydrogen sulfide gas diffuses more quickly than sulfur dioxide gas.
(b) (ii) Deduce a relationship between the relative formula mass (\(M_r\)) of a gas and the speed at which a gas diffuses. Use the \(A_r\) values to help you.
\([A_r \text{ values: } H = 1 \quad S = 32 \quad O = 16]\)
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(e) / 2(d): Redox reactions — part (a)(ii)
• 1(a): States of matter & diffusion — parts (b)(i), (b)(ii)
• 1(e): Relative formula mass calculations — part (b)(ii)
▶️ Answer/Explanation
(a)(i) \(2H_2S(g) + SO_2(g) \rightarrow 3S(s) + 2H_2O(l)\)
Details: The equation must be balanced with correct state symbols. The reaction produces sulfur (solid) and water (liquid).
(a)(ii) Sulfur dioxide is reduced because it loses oxygen.
Details: In this redox reaction, SO2 loses oxygen to form sulfur (S), which is reduction. Alternatively, one could note it gains electrons, but the mark scheme specifies oxygen loss.
(b)(i) The pale yellow solid (sulfur) forms closer to the sulfur dioxide side / left side. This shows that hydrogen sulfide particles diffused further in the same time, meaning they move faster.
Details: The solid forms where the gases meet. Since it’s nearer the SO2 side, H2S traveled a greater distance in the same time, indicating higher diffusion speed.
(b)(ii)
1. Calculate \(M_r\) of H2S = \(2 \times 1 + 32 = 34\)
2. Calculate \(M_r\) of SO2 = \(32 + 2 \times 16 = 64\)
3. Relationship: The higher the relative formula mass (\(M_r\)), the slower the gas diffuses.
Details: This follows Graham’s law of diffusion—lighter gases diffuse faster. H2S (\(M_r = 34\)) diffuses faster than SO2 (\(M_r = 64\)).
This question is about ionic compounds.
(a) State the formula of the cation and the anion in magnesium sulfate.
Cation: ……………………. Anion: …………………….
(b) The diagram shows the electronic configuration of a potassium atom and an oxygen atom.
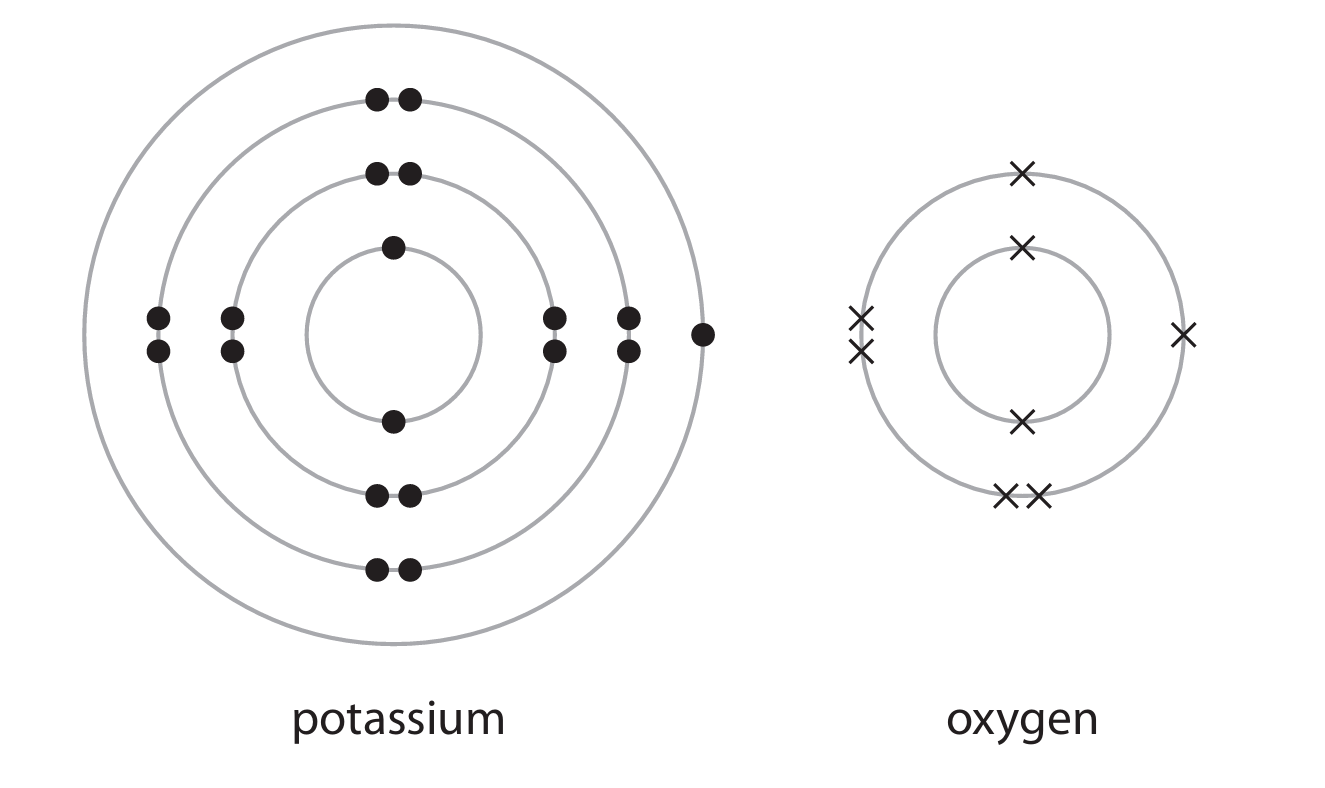
Potassium oxide (K2O) is an ionic compound.
Draw the electronic configuration of a potassium ion and an oxide ion.
Show the charge on each ion.

(c) A sample of solid potassium oxide is added to water. A reaction occurs and a colourless solution forms. When a few drops of phenolphthalein indicator are added to the solution it turns pink.
(i) Identify the ion responsible for the colour change.
(ii) Give a chemical equation for the reaction between potassium oxide and water.
(d) Explain why ionic compounds conduct electricity when molten or in aqueous solution, but not when in the solid state.
(e) The diagram shows the apparatus a teacher uses to demonstrate the electrolysis of a concentrated aqueous solution of sodium chloride.
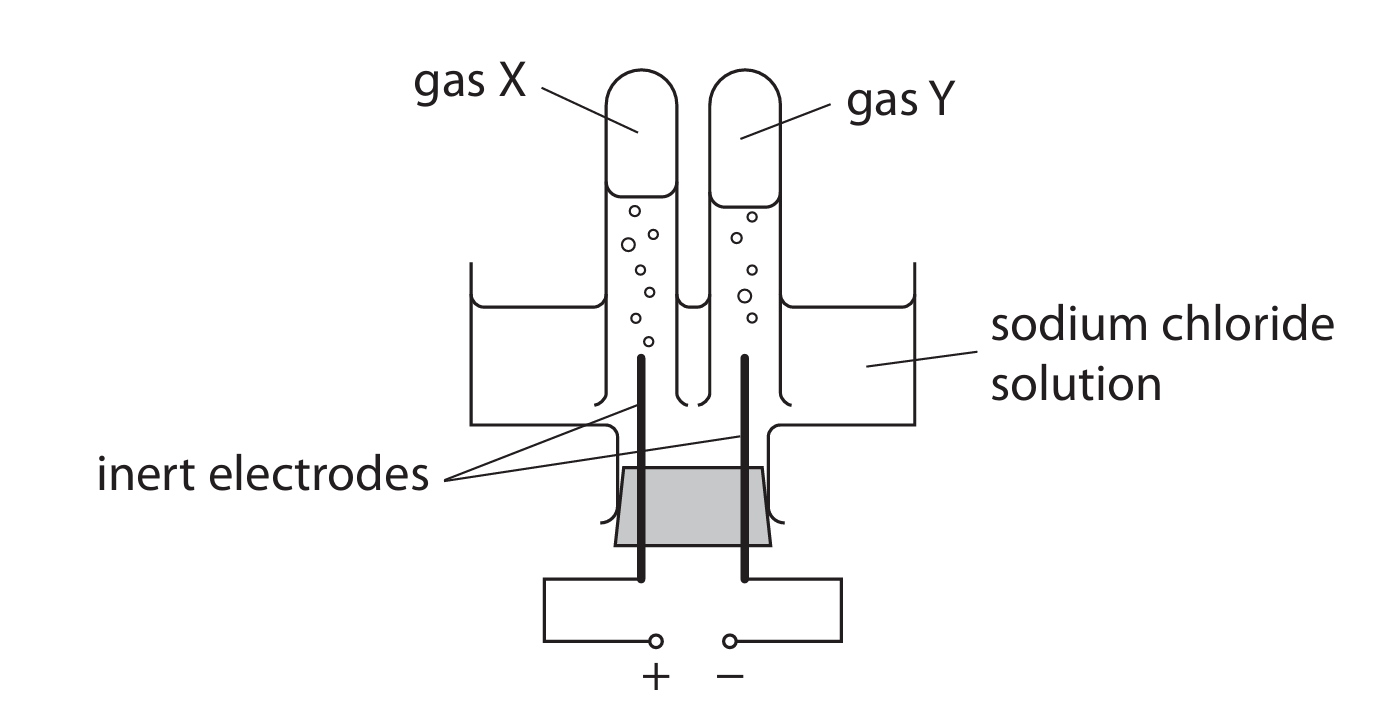
During the electrolysis two gases, X and Y, are formed. One of the gases produces a squeaky pop when tested with a lighted splint.
Use ionic half-equations to identify X and Y.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(f) & 2(g): Acids, alkalis and titrations / Acids, bases and salt preparations — part (c)
• 1(f) & 1(i): Ionic bonding & Electrolysis — part (d)
• 1(i): Electrolysis — part (e)
▶️ Answer/Explanation
(a)
Cation: Mg2+
Anion: SO42-
Explanation: Magnesium sulfate (MgSO4) is an ionic compound. Magnesium, a Group 2 metal, loses two electrons to form Mg2+. The sulfate ion (SO42-) is a polyatomic anion.
(b)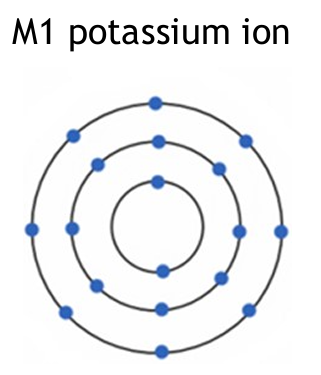
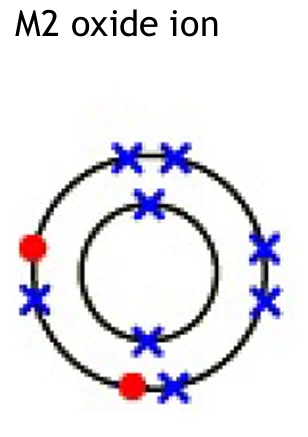
Potassium ion: K+ (loses one electron from its outer shell).
Oxide ion: O2- (gains two electrons to fill its outer shell).
Explanation: In ionic bonding, potassium (Group 1) donates one electron to achieve a full outer shell, becoming a positive ion. Oxygen (Group 6) accepts two electrons to complete its outer shell, becoming a negative ion. The charges must be clearly shown.
(c)
(i) Hydroxide ion / OH– (allow HO–)
Explanation: Potassium oxide reacts with water to form potassium hydroxide (KOH), an alkali. Phenolphthalein turns pink in alkaline solutions due to the presence of OH– ions.
(ii) \(\text{K}_2\text{O} + \text{H}_2\text{O} \rightarrow 2\text{KOH}\) (allow multiples)
Explanation: This is a metal oxide + water → metal hydroxide reaction. Potassium oxide dissolves exothermically in water to form a colourless, alkaline solution of potassium hydroxide.
(d)
M1: When molten or in aqueous solution, the ions are free to move / are mobile.
M2: In the solid state, the ions are fixed in position / held in a lattice and cannot move.
Explanation: Ionic compounds conduct electricity by the movement of charged particles (ions). In a solid, the ions are locked in a rigid giant ionic lattice. When molten or dissolved, the lattice breaks down, allowing ions to move and carry charge.
(e)
M1: X is chlorine / Cl2
M2: Y is hydrogen / H2
M3: \(2\text{Cl}^- \rightarrow \text{Cl}_2 + 2\text{e}^-\) (half-equation at anode)
M4: \(2\text{H}^+ + 2\text{e}^- \rightarrow \text{H}_2\) (half-equation at cathode)
Explanation: In the electrolysis of concentrated aqueous sodium chloride, chloride ions are discharged at the anode (positive electrode) to form chlorine gas. Hydrogen ions from water are preferentially discharged at the cathode (negative electrode) to form hydrogen gas, which gives a squeaky pop with a lighted splint. Sodium ions remain in solution.
Metals are found in the Earth’s crust either as uncombined elements or in metal compounds in rocks.
The method of extraction of a metal is related to its position in the reactivity series.
The table shows the positions of some metals and carbon in the reactivity series.
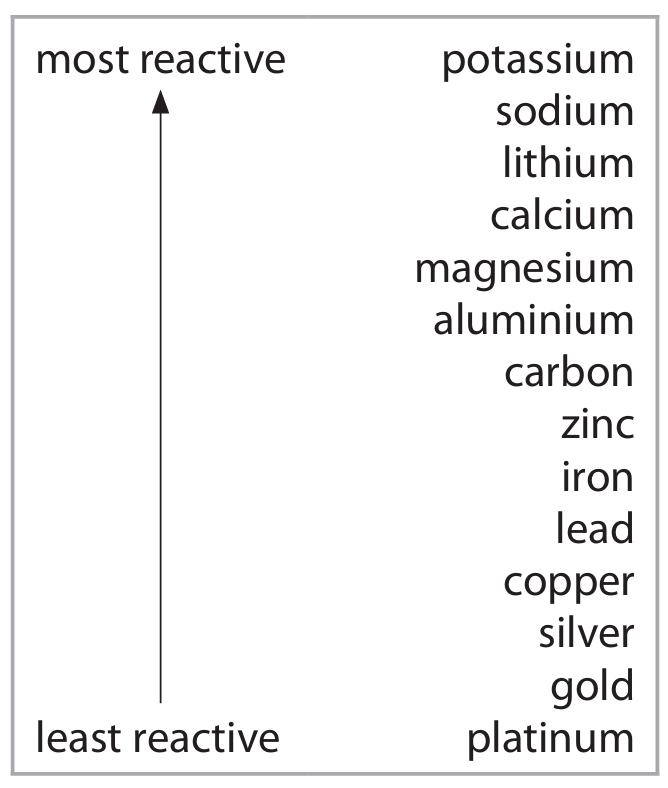
(a) (i) State the name given to rocks that contain metal compounds used in the extraction of metals.
(a) (ii) Name a metal that is found as an uncombined element in the Earth’s crust.
(b) Carbon extraction and electrolysis are two methods of obtaining a metal from a compound.
(i) Explain, without giving practical details, which method is most suitable to obtain calcium from calcium chloride.
(ii) Explain, without giving practical details, which method is most suitable to obtain lead from lead oxide.
(c) Explain, using a labelled diagram, why lead metal is malleable.
(d) Aluminium is extracted from aluminium oxide.
The overall equation for the process is:
\( 2Al_2O_3 \rightarrow 4Al + 3O_2 \)
Calculate the maximum mass, in grams, of aluminium that could be obtained from \( 1.275 \, \text{kg} \) of aluminium oxide.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(h) Metallic bonding — part (c)
• 1(e) Chemical formulae, equations and calculations — part (d)
• 2(d) Reactivity series — link to part (b)
▶️ Answer/Explanation
(a) (i) ore(s)
(a) (ii) gold / platinum / silver / copper
(b) (i) Electrolysis is the most suitable method. Calcium is more reactive than carbon, so carbon cannot displace calcium from its compounds. Electrolysis uses electrical energy to decompose the molten calcium chloride into calcium and chlorine.
(b) (ii) Carbon extraction is the most suitable method. Carbon is more reactive than lead, so it can displace lead from lead oxide in a reduction reaction (e.g., heating with coke).
(c) A labelled diagram showing at least two layers of metal atoms/positive ions in a regular lattice. Explanation: In metallic bonding, the lattice consists of layers of positive ions in a ‘sea’ of delocalised electrons. These layers can slide over each other without breaking the metallic bonds, which are non-directional. This allows the metal to be hammered or bent into shape — a property known as malleability.
(d) Calculation:
1. Calculate \( M_r \) of \( Al_2O_3 \): \( (2 \times 27) + (3 \times 16) = 102 \).
2. Convert mass to moles: \( \text{moles of } Al_2O_3 = \frac{1275 \, \text{g}}{102} = 12.5 \, \text{mol} \).
3. From the equation \( 2Al_2O_3 \rightarrow 4Al \), mole ratio is \( 2:4 \) or \( 1:2 \).
So, moles of \( Al = 12.5 \times 2 = 25.0 \, \text{mol} \).
4. Mass of aluminium: \( 25.0 \times 27 = 675 \, \text{g} \).
Final answer: \( 675 \, \text{g} \) of aluminium.
This question is about alcohols, carboxylic acids and esters.
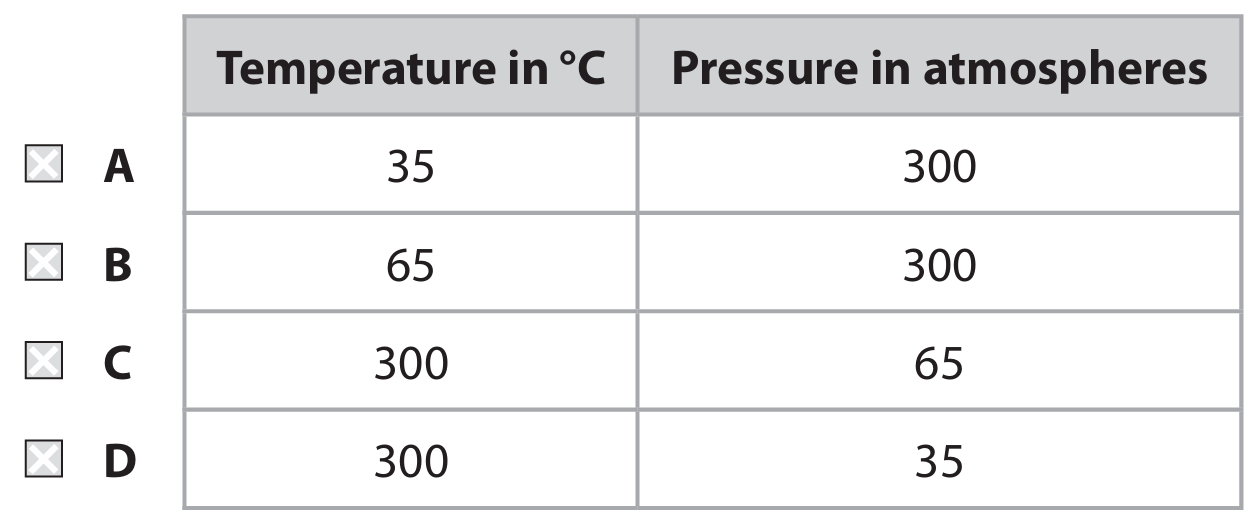
(a) Ethanol can be manufactured by reacting ethene with steam in the presence of a phosphoric acid catalyst.
Which row gives the correct conditions of temperature and pressure for this reaction?
(b) Give the displayed formula of butanol.
(c) Ethanoic acid (CH3COOH) is a carboxylic acid present in vinegar.
(c)(i) The concentration of CH3COOH in vinegar can be found by titration with aqueous potassium hydroxide (KOH). The equation for the reaction is:
\[ \text{CH}_3\text{COOH} + \text{KOH} \rightarrow \text{CH}_3\text{COOK} + \text{H}_2\text{O} \]
In a titration, a 25.0 cm3 sample of vinegar is neutralised by 45.00 cm3 of KOH solution of concentration 0.400 mol/dm3.
Calculate the concentration, in mol/dm3, of CH3COOH in this sample of vinegar.
(c)(ii) A sample of vinegar containing 0.0030 mol of CH3COOH is poured into a flask. Calculate the maximum volume, in cm3, of carbon dioxide gas formed at rtp when excess sodium carbonate is added to the flask. The equation for the reaction is:
\[ 2\text{CH}_3\text{COOH} + \text{Na}_2\text{CO}_3 \rightarrow 2\text{CH}_3\text{COONa} + \text{H}_2\text{O} + \text{CO}_2 \]
[Assume that the molar volume of carbon dioxide at rtp is 24 000 cm3]
(d) Alcohols react with carboxylic acids to form esters. Which alcohol could react to form the ester ethyl propanoate?
- A) \(\text{CH}_3\text{OH}\)
- B) \(\text{C}_2\text{H}_5\text{OH}\)
- C) \(\text{C}_3\text{H}_7\text{OH}\)
- D) \(\text{C}_4\text{H}_9\text{OH}\)
(e) Polyesters are formed in condensation polymerisation reactions between dicarboxylic acids and diols.
(e)(i) State one difference between condensation polymerisation and addition polymerisation.
(e)(ii) The repeat unit of a polyester is:
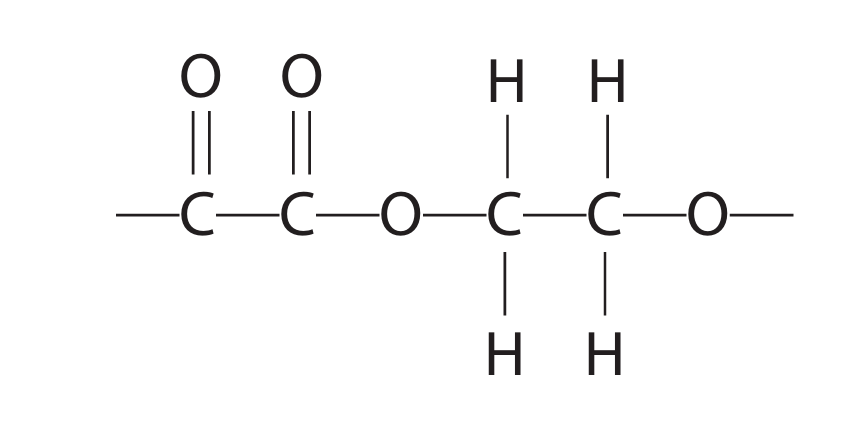
Give the displayed formula of each of the two monomers needed to form this polyester.
(e)(iii) Give one advantage of biopolyssters.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(f): Carboxylic acids — parts (c)(i), (c)(ii)
• 1(e): Chemical formulae, equations and calculations — parts (c)(i), (c)(ii)
• 4(g): Esters — part (d)
• 4(h): Synthetic polymers — parts (e)(i), (e)(ii), (e)(iii)
▶️ Answer/Explanation
(a) C (300°C and 65 atm)
A is incorrect because 35°C and 300 atm are not the correct conditions.
B is incorrect because 65°C and 300 atm are not the correct conditions.
D is incorrect because 300°C and 35 atm are not the correct conditions.
The industrial hydration of ethene to ethanol requires a high temperature (around 300°C) and a moderately high pressure (60–70 atmospheres) to achieve a reasonable reaction rate and equilibrium yield in the presence of a phosphoric acid catalyst.
(b) Displayed formula of butanol (butan-1-ol):
H H H H
| | | |
H—C—C—C—C—O—H
| | | |
H H H H
This is the displayed formula for butan-1-ol, showing all atoms and bonds. Butanol has the molecular formula C4H9OH.
(c)(i)
Step 1: Calculate moles of KOH used.
\[ \text{Moles of KOH} = \frac{45.00}{1000} \times 0.400 = 0.0180 \text{ mol} \]
Step 2: From the 1:1 stoichiometry of the reaction, moles of CH3COOH = 0.0180 mol.
This is present in 25.0 cm3 of vinegar.
\[ \text{Concentration of CH}_3\text{COOH} = \frac{0.0180}{25.0/1000} = 0.720 \text{ mol/dm}^3 \]
Answer: 0.720 mol/dm3
(c)(ii)
Step 1: From the equation, 2 mol CH3COOH produce 1 mol CO2.
Moles of CH3COOH given = 0.0030 mol.
\[ \text{Moles of CO}_2 = \frac{0.0030}{2} = 0.0015 \text{ mol} \]
Step 2: Volume of CO2 at rtp:
\[ \text{Volume} = 0.0015 \times 24000 = 36 \text{ cm}^3 \]
Answer: 36 cm3
(d) B (C2H5OH)
A is not correct because CH3OH would form methyl propanoate.
C is not correct because C3H7OH would form propyl propanoate.
D is not correct because C4H9OH would form butyl propanoate.
Ethyl propanoate is formed from ethanol (C2H5OH) and propanoic acid.
(e)(i) In condensation polymerisation, a small molecule (e.g., water) is lost when monomers join, whereas in addition polymerisation, monomers add together without the loss of any small molecules.
(e)(ii) The two monomers are a dicarboxylic acid (ethanedioic acid) and a diol (ethane-1,2-diol).
Displayed formulae:
Ethanedioic acid (oxalic acid):
O O
|| ||
HO—C—C—OH
Ethane-1,2-diol:
H H
| |
HO—C—C—OH
| |
H H
(e)(iii) Biopolyssters are biodegradable / can be broken down by microorganisms, reducing environmental pollution compared to non-biodegradable plastics.
Hydrogen gas and iodine gas react together to form hydrogen iodide gas.
\[ H_2(g) + I_2(g) \rightleftharpoons 2HI(g) \]
(a) (i) The pressure of an equilibrium mixture of the three gases is increased. Predict the effect of this change on the yield of hydrogen iodide at equilibrium, giving a reason for your answer.
(a) (ii) A catalyst is added to an equilibrium mixture of the three gases. Predict the effect of the catalyst on the yield of hydrogen iodide at equilibrium, giving a reason for your answer.
(b) Hydrogen gas reacts with fluorine gas to form hydrogen fluoride gas.
\[ H_2(g) + F_2(g) \rightarrow 2HF(g) \]
The table gives some bond energies.
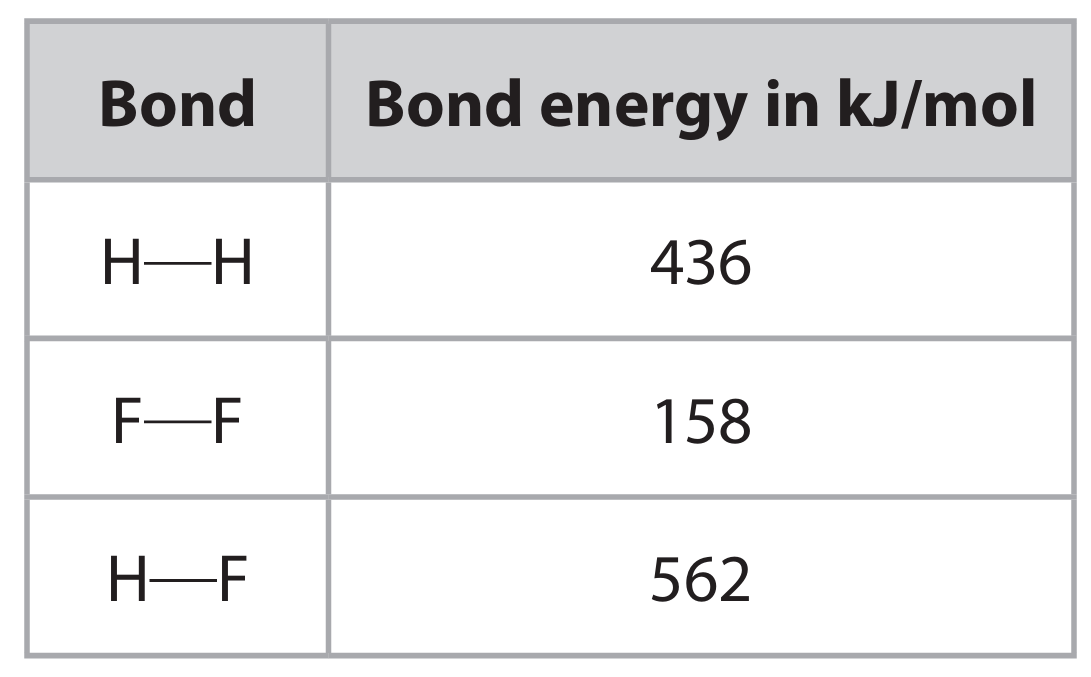
Use the equation and the data in the table to calculate the enthalpy change \( \Delta H \) in kJ/mol, for the reaction. Include a sign in your answer.
(c) Draw an energy level diagram for the reaction between hydrogen and fluorine.
Label the enthalpy change, \( \Delta H \).
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
• 3(a): Energetics — parts (b), (c)
▶️ Answer/Explanation
(a) (i)
• Effect: No effect on the yield of HI.
• Reason: Because there are the same number of moles of gas on both sides of the equation. According to Le Chatelier’s principle (or equilibrium position arguments), changing pressure only affects the equilibrium if there is a change in the number of gaseous moles. Here, 2 moles of gas become 2 moles of gas, so the position of equilibrium is unaffected.
(a) (ii)
• Effect: No effect on the yield of HI at equilibrium.
• Reason: Because a catalyst increases the rates of both the forward and reverse reactions by the same amount. It provides an alternative reaction pathway with a lower activation energy, which helps the reaction reach equilibrium faster, but does not change the position of the equilibrium itself.
(b)
The enthalpy change \( \Delta H \) is calculated using bond energies:
Bonds Broken (Endothermic): 1 H—H + 1 F—F = 436 + 158 = 594 kJ/mol
Bonds Made (Exothermic): 2 H—F = 2 × 562 = 1124 kJ/mol
\[ \Delta H = \text{(Energy to break bonds)} – \text{(Energy released making bonds)} \]
\[ \Delta H = 594 – 1124 = -530 \text{ kJ/mol} \]
The reaction is exothermic, so \( \Delta H = \mathbf{-530 \text{ kJ/mol}} \).
(c)
The energy level diagram should show:
1. The energy level of the reactants \( H_2 + F_2 \) on the left.
2. The energy level of the products \( 2HF \) on the right, drawn below the reactants because the reaction is exothermic (\( \Delta H \) is negative).
3. A downward arrow connecting the reactant and product energy levels, labelled \( \Delta H \) or “–530 kJ/mol”.
Note: An accurate diagram would also show a ‘hump’ for the activation energy, but the question primarily focuses on showing the overall enthalpy change.
