Edexcel iGCSE Chemistry -1.1–1.2 Particle Model of Solids, Liquids and Gases; Changes of State- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.1–1.2 Particle Model of Solids, Liquids and Gases; Changes of State- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.1–1.2 Particle Model of Solids, Liquids and Gases; Changes of State- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.1 understand the three states of matter in terms of the arrangement, movement and energy of the particles
1.2 understand the interconversions between the three states of matter in terms of:
• the names of the interconversions
• how they are achieved
• the changes in arrangement, movement and energy of the particles.
1.1 The Three States of Matter
All matter is made up of tiny particles (atoms or molecules). The particle model explains the properties of solids, liquids and gases by describing the arrangement, movement, and energy of their particles.
Solid
In a solid, particles are arranged in a regular, closely packed pattern. They are held together by strong forces of attraction.
Particles in a solid vibrate about fixed positions but cannot move freely.
Solids have the lowest kinetic energy of the three states.
Because of this:
• Solids have a fixed shape.
• Solids have a fixed volume.
• Solids are not easily compressed.
Liquid
In a liquid, particles are still close together but arranged randomly. The forces between particles are weaker than in solids.
Particles can move past one another, allowing liquids to flow.
Liquids have more kinetic energy than solids but less than gases.
Because of this:
• Liquids have a fixed volume.
• Liquids take the shape of their container.
• Liquids are not easily compressed.
Gas
In a gas, particles are far apart and arranged randomly. The forces between particles are very weak.
Particles move rapidly and freely in all directions.
Gases have the highest kinetic energy.
Because of this:
• Gases have no fixed shape.
• Gases have no fixed volume.
• Gases are easily compressed.
Comparison of Solids, Liquids and Gases
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Arrangement | Closely packed, regular | Close together, random | Far apart, random |
| Movement | Vibrate in fixed position | Slide past each other | Move freely and rapidly |
| Energy | Lowest | Medium | Highest |
Example 1 (Conceptual):
Explain why liquids can flow but solids cannot, using particle theory.
▶️ Answer/Explanation
In liquids, particles are close together but not fixed in position. They can move past one another.
In solids, particles are held in fixed positions and can only vibrate, so they cannot flow.
Example 2 (Numerical):
A gas sample has a volume of \( 120 \, \mathrm{cm^3} \). It is compressed to \( 75 \, \mathrm{cm^3} \).
Which state of matter shows this behaviour and why?
▶️ Answer/Explanation
This behaviour shows a gas.
The volume decreases from \( 120 \, \mathrm{cm^3} \) to \( 75 \, \mathrm{cm^3} \), showing it is compressible.
Gas particles are far apart with large spaces between them, allowing compression.
Example 3 (Hard):
When a solid is heated strongly, it becomes a gas. Describe fully the changes in arrangement, movement and energy of the particles.
▶️ Answer/Explanation
As the solid is heated, particles gain kinetic energy and vibrate more strongly.
They overcome some attractive forces and become a liquid, where particles can move past each other.
With further heating, particles gain even more energy, overcome all attractive forces, move far apart and form a gas.
1.2 Interconversions Between the Three States of Matter
Matter can change from one state to another. These changes are called interconversions. Interconversions occur when energy is either transferred to or removed from a substance, usually by heating or cooling.
During a change of state, the substance itself does not change chemically. Only the arrangement, movement and energy of the particles change.
Names of Interconversions
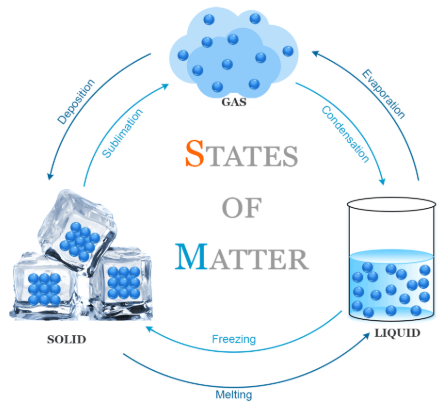
| Change of State | Process Name | Energy Transfer |
|---|---|---|
| Solid → Liquid | Melting | Heating |
| Liquid → Solid | Freezing | Cooling |
| Liquid → Gas | Boiling / Evaporation | Heating |
| Gas → Liquid | Condensation | Cooling |
| Solid → Gas | Sublimation | Heating |
| Gas → Solid | Deposition | Cooling |
How Interconversions Are Achieved
When a substance is heated, particles gain kinetic energy.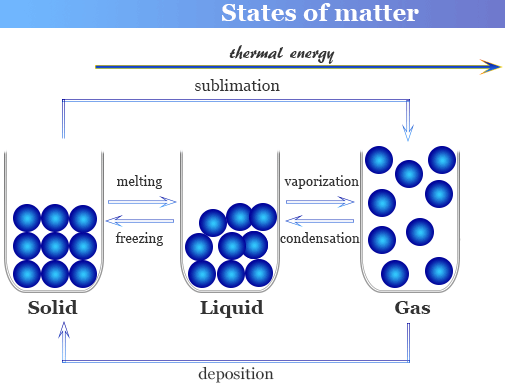
• They move faster.
• Attractive forces weaken.
• Particles move further apart.
When a substance is cooled, particles lose kinetic energy.
• They move more slowly.
• Attractive forces become stronger.
• Particles move closer together.
Changes in Arrangement, Movement and Energy
| Process | Arrangement Change | Movement Change | Energy Change |
|---|---|---|---|
| Melting | Regular → Irregular but close | Vibrate → Slide | Increase |
| Boiling | Close → Far apart | Sliding → Rapid random motion | Increase |
| Freezing | Irregular → Regular | Slide → Vibrate | Decrease |
| Condensation | Far apart → Close | Rapid → Slower sliding | Decrease |
Example 1 (Conceptual):
Explain why condensation happens when warm air touches a cold surface.
▶️ Answer/Explanation
When warm gas particles touch a cold surface, they lose kinetic energy.
They move more slowly and attractive forces pull them closer together, forming a liquid.
Example 2 (Numerical):
A sample of ice at \( 0^\circ \mathrm{C} \) is heated until it becomes water at \( 0^\circ \mathrm{C} \).
Explain why the temperature does not increase during melting.
▶️ Answer/Explanation
The energy supplied is used to overcome the attractive forces between particles rather than increase kinetic energy.
Since kinetic energy does not increase, the temperature stays constant.
Example 3 (Hard):
Describe fully the particle changes when dry ice (solid carbon dioxide) turns directly into a gas.
▶️ Answer/Explanation
This process is sublimation.
Particles gain kinetic energy and overcome the strong forces holding them in fixed positions.
They move from a regular, closely packed arrangement to being far apart and moving rapidly in random directions.
Energy increases and particle movement changes from vibration to free motion.
