Edexcel iGCSE Chemistry -1.10 Separation Techniques- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.10 Separation Techniques- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.10 Separation Techniques- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.10 describe these experimental techniques for the separation of mixtures:
• simple distillation
• fractional distillation
• filtration
• crystallisation
• paper chromatography
1.10 Separation Techniques for Mixtures
Mixtures can be separated using physical methods. These methods do not involve chemical reactions — they separate substances based on differences in physical properties such as boiling point or solubility.
Simple Distillation
Simple distillation is used to separate:
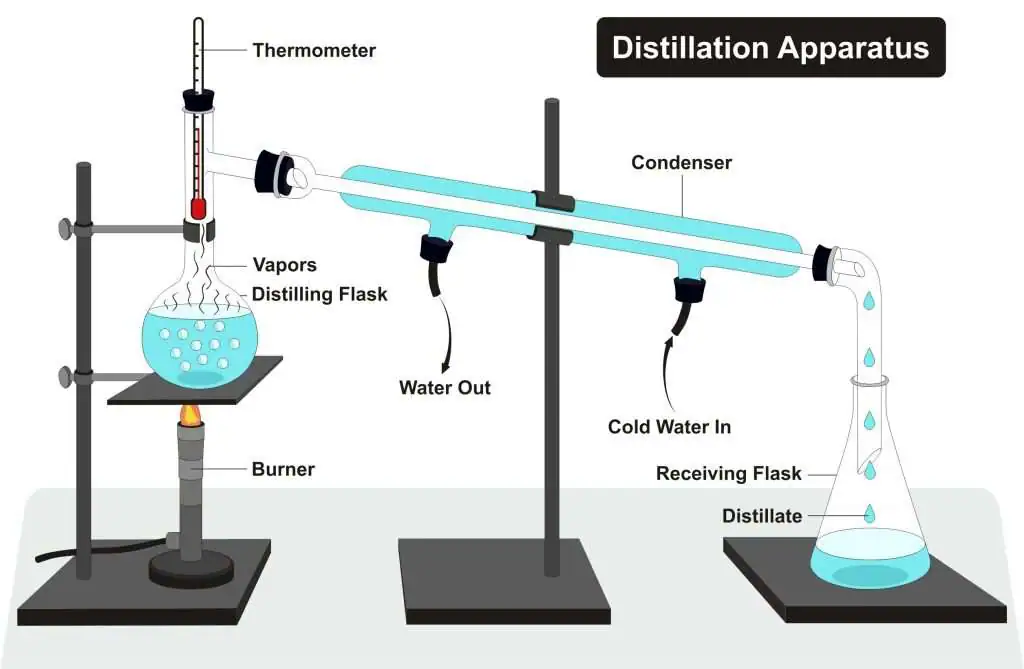
• A solvent from a solution
• A liquid from a dissolved solid
Principle: Different boiling points.
Process:
1. The mixture is heated.
2. The liquid with the lowest boiling point evaporates first.
3. The vapour passes into a condenser.
4. It cools and condenses back into a liquid (distillate).
Used to obtain pure water from salt water.
Fractional Distillation
Used to separate a mixture of two or more liquids with different boiling points.
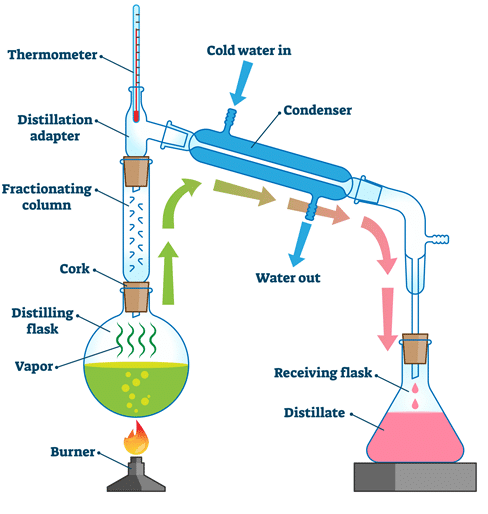
Principle: Different boiling points (closer than in simple distillation).
A fractionating column is used. It contains glass beads which allow repeated condensation and evaporation.
The liquid with the lowest boiling point reaches the top first and is collected.
Used to separate ethanol and water or fractions in crude oil.
Filtration
Filtration separates an insoluble solid from a liquid.
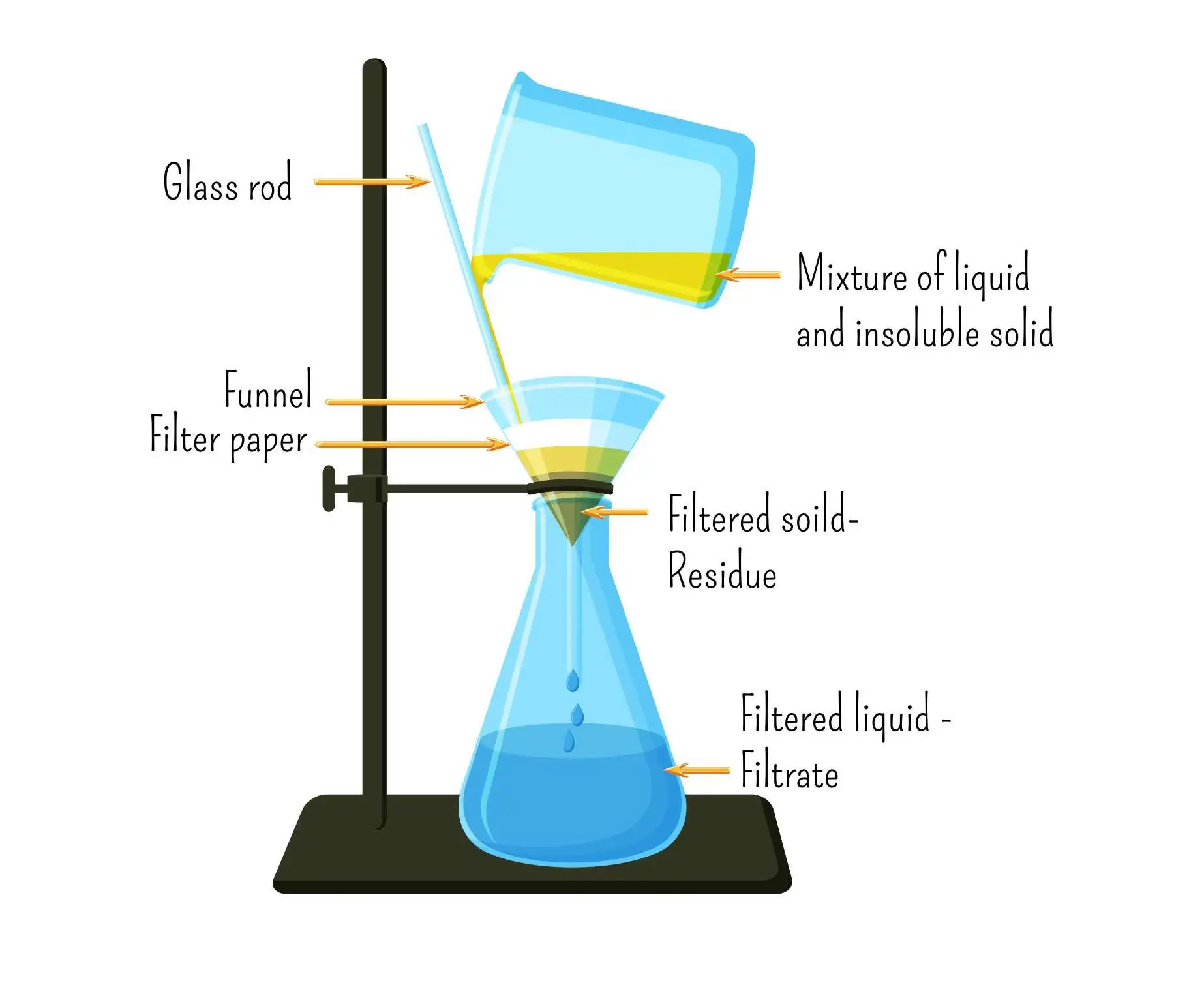
Process:
1. The mixture is poured through filter paper.
2. The liquid passes through (filtrate).
3. The solid remains on the paper (residue).
Used to separate sand from water.
Crystallisation
Used to obtain a pure solid from a solution.
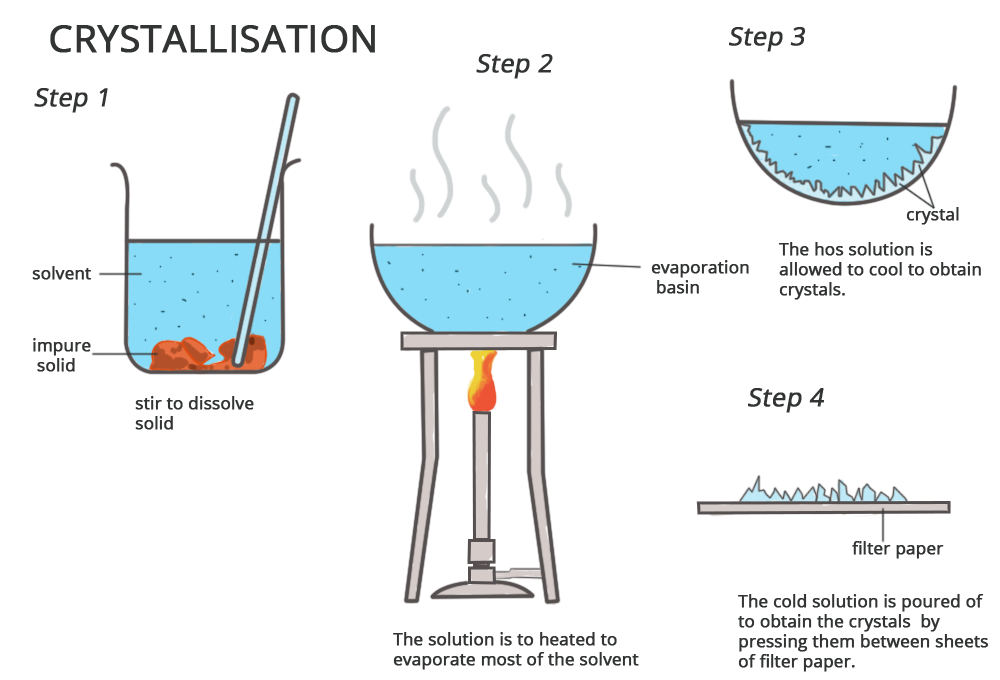
Process:
1. The solution is heated to evaporate some solvent.
2. When nearly saturated, heating stops.
3. The solution is left to cool.
4. Crystals form as solubility decreases.
5. Crystals are filtered and dried.
Used to purify copper sulfate crystals.
Paper Chromatography
Used to separate substances that are dissolved in a solution.
Principle: Different substances travel at different speeds in a solvent.
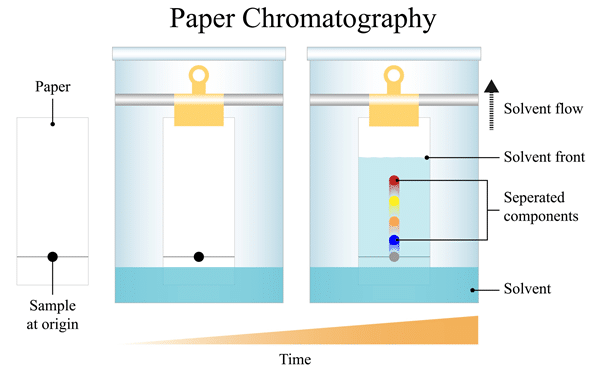
Process:
1. A small spot of solution is placed on chromatography paper.
2. The paper is placed in solvent.
3. The solvent moves up the paper.
4. Substances move different distances.
Rf value:
\( R_f = \dfrac{\text{distance travelled by substance}}{\text{distance travelled by solvent}} \)
Rf values are between 0 and 1.
Comparison of Techniques
| Technique | Used For | Property Used |
|---|---|---|
| Simple distillation | Liquid + dissolved solid | Boiling point |
| Fractional distillation | Two or more liquids | Boiling point (close values) |
| Filtration | Insoluble solid + liquid | Particle size |
| Crystallisation | Solid from solution | Solubility |
| Chromatography | Dissolved substances | Solubility & attraction |
Example 1 (Conceptual):
Which method would you use to separate sand from salt solution?
▶️ Answer/Explanation
Filtration first to remove sand (insoluble solid).
Then simple distillation or crystallisation to obtain salt.
Example 2 (Numerical):
A substance travels 4.5 cm on a chromatography paper. The solvent front travels 6.0 cm. Calculate the \( R_f \) value.
▶️ Answer/Explanation
\( R_f = \dfrac{4.5}{6.0} \)
\( R_f = 0.75 \)
Example 3 (Hard):
Explain fully why fractional distillation is more effective than simple distillation for separating ethanol and water.
▶️ Answer/Explanation
Ethanol and water have relatively close boiling points.
A fractionating column allows repeated evaporation and condensation.
The liquid with the lower boiling point (ethanol) rises further up the column.
The higher boiling liquid (water) condenses and falls back down.
This repeated process improves separation efficiency.
