Edexcel iGCSE Chemistry -1.11–1.12 Chromatograms and Rf Values- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.11–1.12 Chromatograms and Rf Values- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.11–1.12 Chromatograms and Rf Values- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.11 understand how a chromatogram provides information about the composition of a mixture
1.12 understand how to use the calculation of Rf values to identify the components of a mixture
1.11 How a Chromatogram Shows the Composition of a Mixture
A chromatogram is the result obtained from chromatography. It provides information about the number of substances in a mixture and can help identify them.
In paper chromatography, different substances move different distances depending on:
• Their solubility in the solvent (mobile phase)
• Their attraction to the paper (stationary phase)
What a Chromatogram Shows
1. Number of substances present
Each spot represents a different substance.
2. Whether a sample is pure or a mixture
• One spot → pure substance
• More than one spot → mixture
3. Identification of substances
Substances can be identified by comparing:
• The position of spots
• The \( R_f \) value
\( R_f = \dfrac{\text{distance travelled by substance}}{\text{distance travelled by solvent}} \)
Rf values must be measured in the same solvent and under the same conditions for valid comparison.
Interpreting Spot Patterns
| Observation | Conclusion |
|---|---|
| One spot | Pure substance |
| Multiple spots | Mixture of substances |
| Spots at same height as reference | Same substance present |
| Different heights | Different substances |
Why Substances Travel Different Distances
A substance that is more soluble in the solvent travels further.
A substance that is more strongly attracted to the paper travels a shorter distance.
This difference allows separation and identification.
Example 1 (Conceptual):
A sample produces three spots on a chromatogram. What does this show?
▶️ Answer/Explanation
The sample is a mixture.
Each spot represents a different substance.
Example 2 (Numerical):
A dye spot travels \( 3.2 \, \mathrm{cm} \). The solvent front travels \( 8.0 \, \mathrm{cm} \). Calculate the \( R_f \) value.
▶️ Answer/Explanation
\( R_f = \dfrac{3.2}{8.0} \)
\( R_f = 0.40 \)
Example 3 (Hard ):
A student runs chromatography on an unknown ink and two known dyes (A and B). The unknown produces two spots: one at the same height as A and one at the same height as B. Explain what this shows and why height comparison is important.
▶️ Answer/Explanation
The unknown ink is a mixture.
It contains dye A and dye B because its spots are at the same height as the reference dyes.
Spots at the same height have travelled the same distance.
This means they have the same \( R_f \) value under the same conditions.
Therefore, they are likely the same substance.
1.12 Using Rf Values to Identify the Components of a Mixture
In paper chromatography, substances are separated based on differences in their movement through a solvent.
To identify substances, we calculate the Rf value.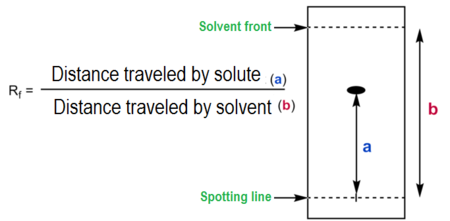
\( R_f = \dfrac{\text{distance travelled by substance}}{\text{distance travelled by solvent front}} \)
The Rf value has no units and is always between 0 and 1.
How to Measure Rf Accurately
1. Measure from the pencil baseline to the centre of the spot.
2. Measure from the baseline to the solvent front.
3. Substitute values into the formula.
Important: Rf values must be compared under the same experimental conditions (same solvent, same temperature, same type of paper).
How Rf Identifies Components
If an unknown substance has the same Rf value as a known substance (under identical conditions), they are likely the same compound.
If a chromatogram shows multiple spots with different Rf values, the substance is a mixture.
What Rf Values Tell Us
| Observation | Interpretation |
|---|---|
| One Rf value | Pure substance |
| Two or more Rf values | Mixture |
| Matching Rf with standard | Substance identified |
Why Substances Have Different Rf Values
A substance with greater solubility in the solvent travels further, giving a larger Rf.
A substance more strongly attracted to the stationary phase travels less, giving a smaller Rf.
Example 1 (Conceptual):
An unknown sample produces two spots with Rf values of 0.32 and 0.75. What does this show?
▶️ Answer/Explanation
The sample is a mixture.
Two different Rf values indicate two different substances are present.
Example 2 (Numerical):
A spot travels \( 5.4 \, \mathrm{cm} \). The solvent front travels \( 9.0 \, \mathrm{cm} \). Calculate the Rf value.
▶️ Answer/Explanation
\( R_f = \dfrac{5.4}{9.0} \)
\( R_f = 0.60 \)
Example 3 (Hard):
A student analyses an unknown food colouring using chromatography. The unknown has an Rf value of 0.42. Known dyes X, Y and Z have Rf values of 0.42, 0.68 and 0.25 respectively under the same conditions. Explain fully how the student can identify the dye.
▶️ Answer/Explanation
The unknown dye has an Rf value of 0.42.
Dye X also has an Rf value of 0.42 under the same experimental conditions.
Because the Rf values match and conditions are identical, the unknown dye is likely dye X.
Rf values must only be compared when the solvent, paper and temperature are the same.
