Edexcel iGCSE Chemistry -1.13 Core Practical: Paper Chromatography- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.13 Core Practical: Paper Chromatography- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.13 Core Practical: Paper Chromatography- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.13 practical: investigate paper chromatography using inks/food colourings
1.13 Practical: Investigating Paper Chromatography Using Inks or Food Colourings
Paper chromatography is used to separate and identify the components of mixtures such as inks and food colourings.
The separation occurs because different substances have different:
• Solubilities in the solvent (mobile phase)
• Attractions to the paper (stationary phase)
Aim
To separate and identify the dyes present in inks or food colourings using paper chromatography.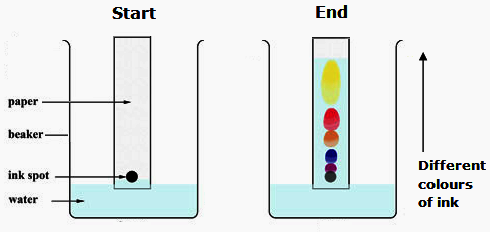
Apparatus
- Chromatography paper
- Pencil (not pen)
- Ruler
- Beaker with solvent (e.g. water)
- Capillary tube
- Lid or watch glass
Method
1. Draw a pencil baseline about 1 cm from the bottom of the paper.
2. Place small spots of ink/food colouring on the baseline.
3. Place the paper in a beaker containing a shallow layer of solvent.
4. Ensure the solvent level is below the baseline.
5. Allow the solvent to travel up the paper.
6. Remove the paper before the solvent reaches the top.
7. Mark the solvent front immediately and allow the paper to dry.
Why Pencil Is Used
Pen ink would dissolve in the solvent and interfere with results. Pencil contains graphite, which does not dissolve.
Calculating Rf
\( R_f = \dfrac{\text{distance travelled by dye}}{\text{distance travelled by solvent}} \)
Rf values are used to identify dyes by comparison with known substances.
Control Variables
| Variable | How Controlled |
|---|---|
| Type of solvent | Use same solvent for all samples |
| Temperature | Keep lab conditions constant |
| Paper type | Use same chromatography paper |
What Results Show
• One spot → pure substance
• Multiple spots → mixture
• Matching Rf values → same substance (under same conditions)
Example 1 (Conceptual):
Why must the solvent level be below the baseline at the start of the experiment?
▶️ Answer/Explanation
If the solvent touches the spots directly, the dyes will dissolve into the solvent instead of travelling up the paper.
This would prevent proper separation.
Example 2 (Numerical):
A blue dye travels \( 4.8 \, \mathrm{cm} \). The solvent front travels \( 8.0 \, \mathrm{cm} \). Calculate the Rf value.
▶️ Answer/Explanation
\( R_f = \dfrac{4.8}{8.0} \)
\( R_f = 0.60 \)
Example 3 (Hard):
Suggest two improvements to increase the accuracy and reliability of a chromatography experiment and explain why they help.
▶️ Answer/Explanation
Use a lid to cover the beaker.
This prevents solvent evaporation and keeps conditions consistent.
Repeat the experiment and calculate average Rf values.
This reduces random errors and improves reliability.
