Edexcel iGCSE Chemistry -1.14–1.15 Atoms, Molecules, and Structure of the Atom- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.14–1.15 Atoms, Molecules, and Structure of the Atom- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.14–1.15 Atoms, Molecules, and Structure of the Atom- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.14 know what is meant by the terms atom and molecule
1.15 know the structure of an atom in terms of the positions, relative masses and relative charges of sub-atomic particles
1.14 The Terms Atom and Molecule
Understanding the difference between atoms and molecules is essential in GCSE Chemistry.
Atom
An atom is the smallest particle of an element that can exist.
Atoms cannot be broken down into simpler substances by chemical reactions.
Each element is made of only one type of atom.
Examples:
• Helium exists as single atoms: \( \mathrm{He} \)
• Sodium atoms are represented as: \( \mathrm{Na} \)
Molecule
A molecule is two or more atoms chemically bonded together.
Molecules can be:
• Molecules of elements (same type of atom)
• Molecules of compounds (different types of atoms)
Examples of Molecules of Elements:
• Oxygen: \( \mathrm{O_2} \)
• Nitrogen: \( \mathrm{N_2} \)
Examples of Molecules of Compounds:
• Water: \( \mathrm{H_2O} \)
• Carbon dioxide: \( \mathrm{CO_2} \)
Key Differences
| Feature | Atom | Molecule |
|---|---|---|
| Definition | Smallest particle of an element | Two or more bonded atoms |
| Bonding | Single particle | Atoms chemically bonded |
| Example | \( \mathrm{He} \) | \( \mathrm{O_2} \), \( \mathrm{H_2O} \) |
Common Exam Confusion
• \( \mathrm{O} \) is an atom, but \( \mathrm{O_2} \) is a molecule.
• A molecule is not the same as a compound — it can be made of the same element.
Example 1 (Conceptual):
Is \( \mathrm{N_2} \) an atom or a molecule? Explain.
▶️ Answer/Explanation
It is a molecule.
It contains two nitrogen atoms chemically bonded together.
Example 2 (Numerical):
How many atoms are present in one molecule of \( \mathrm{H_2O} \)?
▶️ Answer/Explanation
\( \mathrm{H_2O} \) contains 2 hydrogen atoms and 1 oxygen atom.
Total atoms = 3.
Example 3 (Hard):
Explain fully why helium exists as single atoms but oxygen exists as molecules.
▶️ Answer/Explanation
Helium is a noble gas with a full outer electron shell.
It is chemically stable and does not need to bond with other atoms.
Oxygen has six electrons in its outer shell.
It needs two more electrons to achieve a full outer shell.
Two oxygen atoms share electrons and form a covalent bond, creating \( \mathrm{O_2} \).
1.15 The Structure of the Atom
Atoms are made up of smaller particles called sub-atomic particles.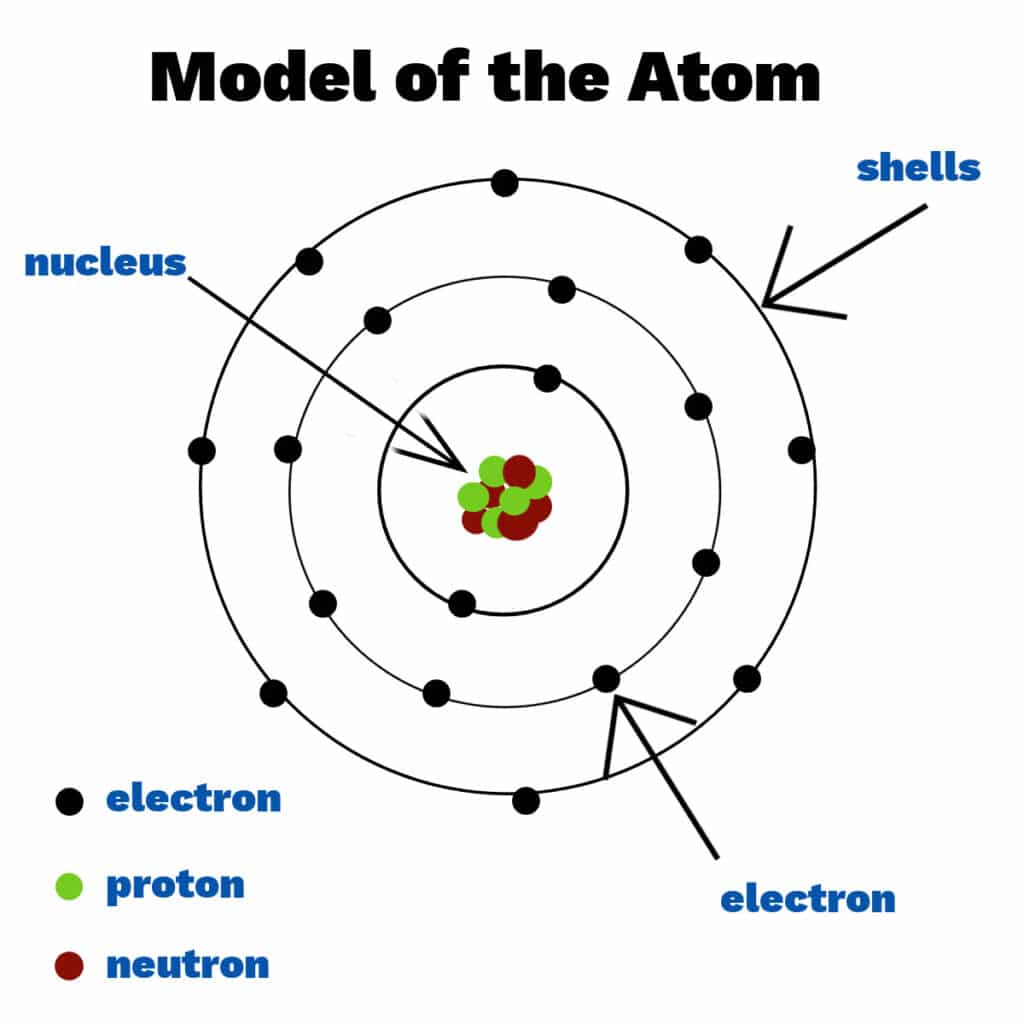
There are three main sub-atomic particles:
- Proton
- Neutron
- Electron
Positions of Sub-Atomic Particles
At the centre of the atom is the nucleus.
• The nucleus contains protons and neutrons.
Electrons move around the nucleus in energy levels (shells).
Relative Charges
Proton: \( +1 \)
Neutron: \( 0 \)
Electron: \( -1 \)
Atoms are electrically neutral because the number of protons equals the number of electrons.
Relative Masses
Proton: \( 1 \)
Neutron: \( 1 \)
Electron: \( \dfrac{1}{1836} \) (very small)
Almost all the mass of an atom is concentrated in the nucleus.
Summary Table
| Particle | Relative Charge | Relative Mass | Location |
|---|---|---|---|
| Proton | +1 | 1 | Nucleus |
| Neutron | 0 | 1 | Nucleus |
| Electron | −1 | \( \dfrac{1}{1836} \) | Shells around nucleus |
Key GCSE Understanding
• Atomic number = number of protons.
• Mass number = protons + neutrons.
• In a neutral atom: protons = electrons.
Example 1 (Conceptual):
Why is most of the mass of an atom found in the nucleus?
▶️ Answer/Explanation
Protons and neutrons have relative mass 1 and are located in the nucleus.
Electrons have a very small mass (\( \dfrac{1}{1836} \)), so they contribute very little to the total mass.
Example 2 (Numerical):
An atom has 12 protons, 12 neutrons and 12 electrons.
Calculate its mass number.
▶️ Answer/Explanation
Mass number = protons + neutrons
= \( 12 + 12 = 24 \)
Example 3 (Hard):
An atom has atomic number 17 and mass number 35. Describe fully the number and positions of its sub-atomic particles.
▶️ Answer/Explanation
Atomic number 17 means there are 17 protons.
In a neutral atom, there are also 17 electrons.
Mass number 35 means total protons and neutrons = 35.
Number of neutrons = \( 35 – 17 = 18 \).
Protons and neutrons are located in the nucleus.
Electrons are arranged in shells around the nucleus.
