Edexcel iGCSE Chemistry -1.16 Atomic Number, Mass Number, and Isotopes- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.16 Atomic Number, Mass Number, and Isotopes- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.16 Atomic Number, Mass Number, and Isotopes- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.16 know what is meant by the terms atomic number, mass number, isotopes and relative atomic mass (Ar)
1.16 Atomic Number, Mass Number, Isotopes and Relative Atomic Mass (Ar)
These terms describe important properties of atoms and help us understand how elements are represented in the periodic table.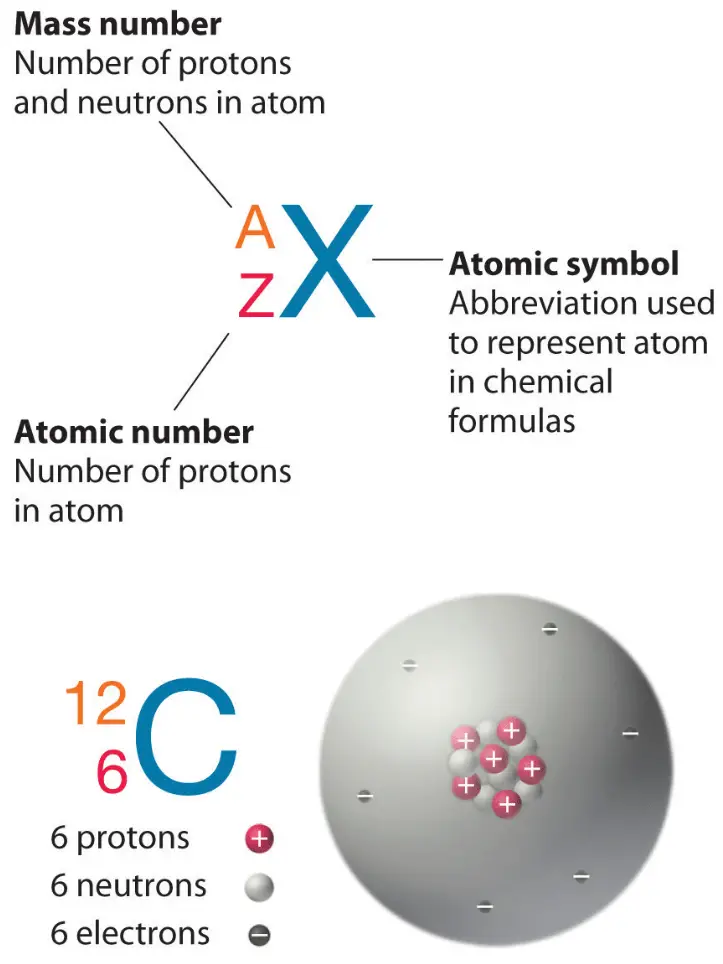
Atomic Number
The atomic number is the number of protons in the nucleus of an atom.
Atomic number = number of protons
In a neutral atom:
Number of electrons = atomic number
The atomic number determines the identity of the element.
Mass Number
The mass number is the total number of protons + neutrons in the nucleus.
Mass number = protons + neutrons
Number of neutrons can be calculated:
Neutrons = mass number − atomic number
Isotopes
Isotopes are atoms of the same element that have:
• The same number of protons
• Different numbers of neutrons
This means isotopes have:
• The same atomic number
• Different mass numbers
Isotopes have the same chemical properties because they have the same number of electrons.
Relative Atomic Mass (Ar)
The relative atomic mass (Ar) is the weighted mean mass of an element compared to \( \dfrac{1}{12} \) of the mass of a carbon-12 atom.

Because most elements exist as mixtures of isotopes, Ar is usually not a whole number.
Weighted mean calculation:
\( A_r = \dfrac{(m_1 \times a_1) + (m_2 \times a_2) + \dots}{100} \)
where \( m \) = isotopic mass and \( a \) = percentage abundance.
![]()
Comparison of Terms
| Term | Definition | Depends On |
|---|---|---|
| Atomic number | Number of protons | Protons only |
| Mass number | Protons + neutrons | Nucleus particles |
| Isotopes | Same protons, different neutrons | Neutron number |
| Relative atomic mass | Weighted mean of isotopes | Isotopic abundance |
Example 1 (Conceptual):
Why do isotopes of the same element have identical chemical properties?
▶️ Answer/Explanation
Chemical reactions involve electrons.
Isotopes have the same number of protons and therefore the same number of electrons.
So they react in the same way chemically.
Example 2 (Numerical):
An element has two isotopes:
Isotope A: mass 20, abundance 90% Isotope B: mass 22, abundance 10%
Calculate the relative atomic mass.
▶️ Answer/Explanation
\( A_r = \dfrac{(20 \times 90) + (22 \times 10)}{100} \)
\( = \dfrac{1800 + 220}{100} \)
\( = 20.2 \)
Example 3 (Hard):
An atom is written as \( \,^{37}_{17}\mathrm{Cl} \). Explain fully what this notation shows.
▶️ Answer/Explanation
The atomic number is 17, meaning there are 17 protons.
In a neutral atom, there are also 17 electrons.
The mass number is 37, meaning total protons and neutrons = 37.
Number of neutrons = \( 37 – 17 = 20 \).
This is an isotope of chlorine.
