Edexcel iGCSE Chemistry -1.18 Arrangement of the Periodic Table- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.18 Arrangement of the Periodic Table- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.18 Arrangement of the Periodic Table- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.18 understand how elements are arranged in the Periodic Table:
• in order of atomic number
• in groups and periods
1.18 Arrangement of Elements in the Periodic Table
The Periodic Table arranges all known elements in a systematic way based on their atomic structure and chemical properties.
Arrangement in Order of Atomic Number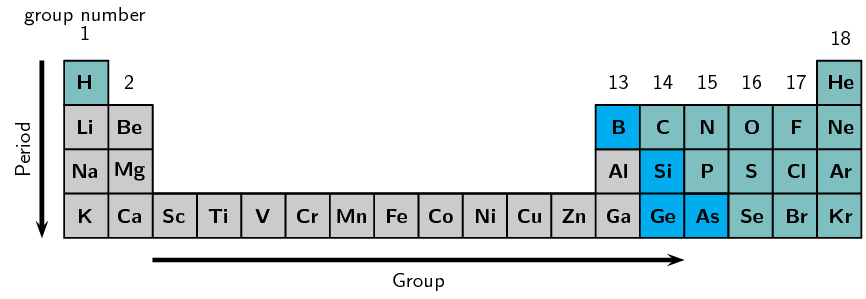
Elements are arranged in increasing atomic number.
Atomic number = number of protons
Each element has one more proton than the element before it.
The atomic number determines the number of electrons in a neutral atom, which controls chemical behaviour.
Groups
Groups are the vertical columns in the Periodic Table.
Elements in the same group have:
• The same number of outer shell (valence) electrons
• Similar chemical properties
Example:
Group 1 → 1 outer electron Group 7 → 7 outer electrons Group 0 → Full outer shell
This explains why elements in the same group react in similar ways.
Periods
Periods are the horizontal rows in the Periodic Table.
The period number tells us:
• The number of occupied electron shells
As you move across a period:
• Atomic number increases • Number of protons increases • Number of outer electrons increases
Groups vs Periods Comparison
| Feature | Group (Column) | Period (Row) |
|---|---|---|
| Direction | Vertical | Horizontal |
| Electrons in outer shell | Same | Increase across |
| Number of shells | Increase down group | Same within period |
| Chemical properties | Similar | Gradually change |
Key GCSE Understanding
• Reactivity trends are explained by electron structure.
• Group number (1–7) tells you outer electrons (for main group elements).
• Period number tells you number of shells.
Example 1 (Conceptual):
Why do elements in Group 1 have similar chemical properties?
▶️ Answer/Explanation
They all have one electron in their outer shell.
Chemical reactions involve outer electrons.
Since they lose one electron easily, they react in similar ways.
Example 2 (Numerical):
An element has atomic number 16 and is in Period 3. How many shells and outer electrons does it have?
▶️ Answer/Explanation
Period 3 means 3 occupied electron shells.
Atomic number 16 means 16 electrons.
Electron arrangement: 2,8,6.
So it has 6 outer electrons.
Example 3 (Hard):
Explain fully how the position of an element in the Periodic Table relates to its atomic structure.
▶️ Answer/Explanation
Elements are arranged in order of increasing atomic number.
The atomic number equals the number of protons in the nucleus.
The group number shows the number of electrons in the outer shell (for main group elements).
The period number shows the number of occupied electron shells.
Elements in the same group have similar chemical properties because they have the same outer electron structure.
