Edexcel iGCSE Chemistry -1.19–1.22 Electronic Configuration and Periodic Position- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.19–1.22 Electronic Configuration and Periodic Position- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.19–1.22 Electronic Configuration and Periodic Position- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.19 understand how to deduce the electronic configurations of the first 20 elements from their positions in the Periodic Table
1.22 understand how the electronic configuration of a main group element is related to its position in the Periodic Table
1.19 Deducing Electronic Configurations of the First 20 Elements
The electronic configuration of an atom shows how its electrons are arranged in energy levels (shells).
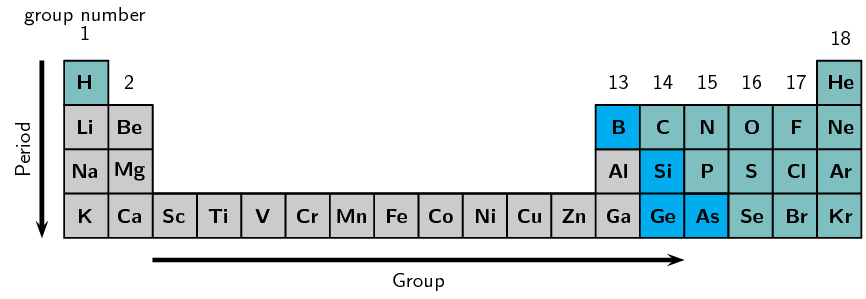
For the first 20 elements, electrons fill shells in a simple pattern.
Key Rules for the First 20 Elements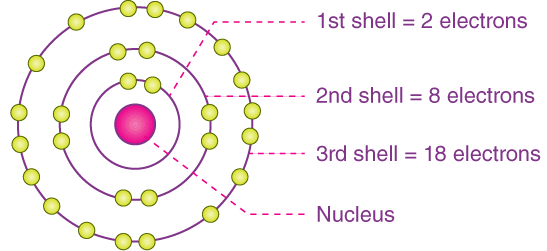
• Electrons fill the lowest energy shell first.
• Maximum electrons per shell (GCSE level):
First shell = 2
Second shell = 8
Third shell = 8 (for first 20 elements)
This gives the pattern: 2, 8, 8, 2 (up to calcium).
Using the Periodic Table to Deduce Configuration
- The atomic number gives the total number of electrons.
- The period number gives the number of occupied shells.
- The group number (for Groups 1–7) gives the number of outer shell electrons.
Examples of Configurations
| Element | Atomic Number | Electronic Configuration |
|---|---|---|
| Hydrogen | 1 | 1 |
| Carbon | 6 | 2,4 |
| Oxygen | 8 | 2,6 |
| Sodium | 11 | 2,8,1 |
| Chlorine | 17 | 2,8,7 |
| Calcium | 20 | 2,8,8,2 |
Why Group and Period Help
Group number (1–7) = outer electrons Period number = number of shells
Example: Chlorine is in Group 7, Period 3.
→ 3 shells → 7 outer electrons → Configuration: 2,8,7
Important GCSE Note
After calcium (atomic number 20), the filling order becomes more complex. At GCSE, you only need to know the first 20 elements.
Example 1 (Conceptual):
Why do elements in the same group have similar chemical properties?
▶️ Answer/Explanation
They have the same number of outer shell electrons.
Chemical reactions involve outer electrons.
Therefore, elements in the same group react in similar ways.
Example 2 (Numerical):
Deduce the electronic configuration of phosphorus (atomic number 15).
▶️ Answer/Explanation
Phosphorus has 15 electrons.
First shell: 2 Second shell: 8 Remaining: 5
Configuration = 2,8,5
Example 3 (Hard):
An element is in Group 2, Period 3 and has atomic number 12. Deduce its electronic configuration and explain your reasoning.
▶️ Answer/Explanation
Atomic number 12 means there are 12 electrons.
Period 3 means 3 occupied shells.
Group 2 means 2 outer shell electrons.
First shell: 2 Second shell: 8 Third shell: 2
Configuration = 2,8,2
1.22 Relationship Between Electronic Configuration and Position in the Periodic Table
For main group elements (Groups 1–7 and 0), the position of an element in the Periodic Table is directly related to its electronic configuration.
Group Number and Outer Electrons

For main group elements:
Group number = number of electrons in the outer shell
Examples:
Sodium (Group 1): 2,8,1 → 1 outer electron
Magnesium (Group 2): 2,8,2 → 2 outer electrons
Chlorine (Group 7): 2,8,7 → 7 outer electrons
Neon (Group 0): 2,8 → Full outer shell
Elements in the same group have similar chemical properties because they have the same number of outer shell electrons.
Period Number and Electron Shells
Period number = number of occupied electron shells
Examples:
Period 1 → 1 shell (e.g. hydrogen: 1)
Period 2 → 2 shells (e.g. oxygen: 2,6)
Period 3 → 3 shells (e.g. sodium: 2,8,1)
As you move across a period:
• Atomic number increases
• One electron is added each time
• Outer electrons increase across the row
Summary Table
| Feature | What It Tells Us | Linked To |
|---|---|---|
| Group number | Number of outer electrons | Reactivity & bonding |
| Period number | Number of shells | Energy levels |
| Atomic number | Total electrons | Electronic configuration |
Why This Matters for Chemical Reactivity
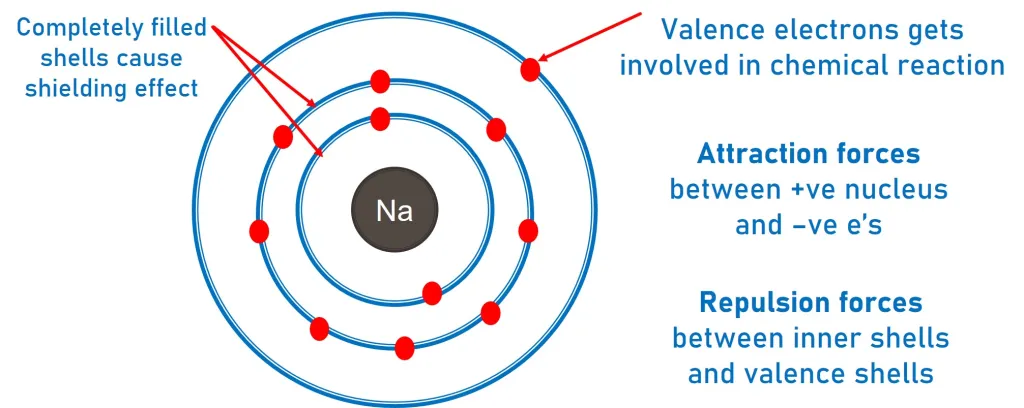
Chemical reactions involve the outer shell electrons.
Group 1 elements lose 1 electron easily.
Group 7 elements gain 1 electron easily.
Group 0 elements are unreactive because they have full outer shells.
Example 1 (Conceptual):
Why are Group 0 elements very unreactive?
▶️ Answer/Explanation
They have a full outer electron shell.
They do not need to gain or lose electrons, so they are chemically stable.
Example 2 (Numerical):
An element has electronic configuration 2,8,6. Deduce its group and period.
▶️ Answer/Explanation
There are 3 shells → Period 3.
There are 6 outer electrons → Group 6.
Example 3 (Hard):
Explain fully how the electronic configuration of chlorine (2,8,7) determines its position and reactivity in the Periodic Table.
▶️ Answer/Explanation
Chlorine has 3 occupied electron shells, so it is in Period 3.
It has 7 electrons in its outer shell, so it is in Group 7.
It needs one more electron to achieve a full outer shell.
Therefore, it tends to gain one electron in reactions.
This explains why chlorine is reactive and forms -1 ions.
