Edexcel iGCSE Chemistry -1.20–1.21 Metals, Non-metals, and Classification of Elements- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.20–1.21 Metals, Non-metals, and Classification of Elements- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.20–1.21 Metals, Non-metals, and Classification of Elements- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.20 understand how to use electrical conductivity and the acid-base character of oxides to classify elements as metals or non-metals
1.21 identify an element as a metal or a non-metal according to its position in the Periodic Table
1.20 Using Electrical Conductivity and Oxides to Classify Elements as Metals or Non-Metals
Elements can be classified as metals or non-metals based on their physical and chemical properties.
Two important properties used at GCSE level are:
• Electrical conductivity
• The acid-base character of their oxides
1. Electrical Conductivity
Metals conduct electricity.
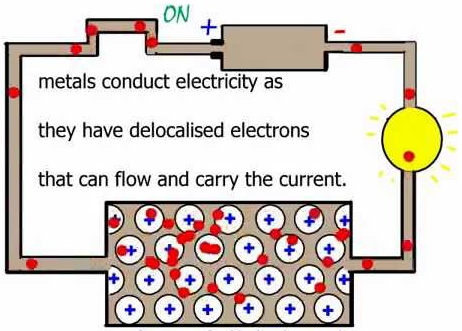
This is because metals have delocalised (free) electrons that can move and carry charge.
Non-metals do not conduct electricity (except graphite).
They do not have free moving charged particles.
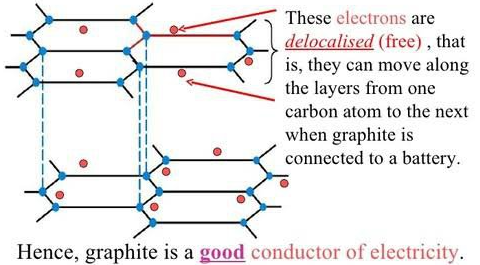
Important Exception: Graphite conducts electricity even though it is a non-metal, because it has delocalised electrons.
2. Acid-Base Character of Oxides
When elements react with oxygen, they form oxides.
The type of oxide formed helps classify the element.
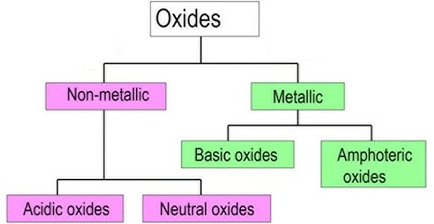
Metal oxides are usually basic.
They react with acids to form salt + water.
Example: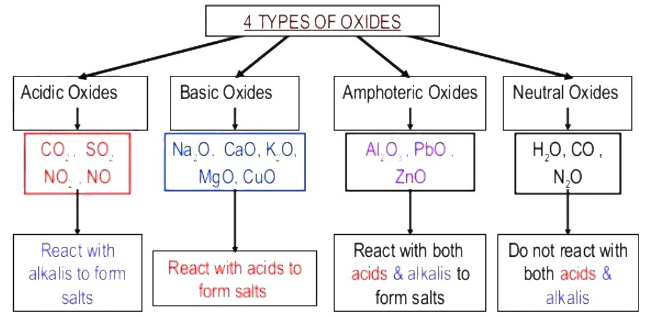
Magnesium oxide + hydrochloric acid → magnesium chloride + water
Non-metal oxides are usually acidic.
They dissolve in water to form acidic solutions.
Example:
Sulfur dioxide dissolves in water to form an acidic solution.
Comparison: Metals vs Non-Metals
| Property | Metals | Non-Metals |
|---|---|---|
| Electrical conductivity | Good conductor | Poor conductor (except graphite) |
| Type of oxide | Basic oxide | Acidic oxide |
| Reaction with acids | Forms salt + water | Usually no reaction |
Using These Properties to Classify an Unknown Element
If it conducts electricity and forms a basic oxide → Metal If it does not conduct electricity and forms an acidic oxide → Non-metal
Example 1 (Conceptual):
An element forms an oxide that neutralises acids. What does this suggest about the element?
▶️ Answer/Explanation
Neutralising acids shows the oxide is basic.
Basic oxides are formed by metals.
Therefore, the element is a metal.
Example 2 (Numerical):
An unknown element has electronic configuration 2,8,1 and conducts electricity. Classify it.
▶️ Answer/Explanation
Configuration 2,8,1 means it is in Group 1.
Group 1 elements are metals.
Since it conducts electricity, it is a metal.
Example 3 (Hard):
Element X does not conduct electricity in solid form. Its oxide dissolves in water to form a solution that turns blue litmus red. Explain how this information classifies element X.
▶️ Answer/Explanation
The element does not conduct electricity, so it is likely a non-metal.
Its oxide dissolves in water and turns blue litmus red.
Turning blue litmus red shows the solution is acidic.
Acidic oxides are formed by non-metals.
Therefore, element X is a non-metal.
1.21 Identifying Metals and Non-Metals from Their Position in the Periodic Table
The position of an element in the Periodic Table allows us to classify it as a metal or a non-metal.
General Layout of the Periodic Table
Metals are found on the left-hand side and in the centre of the Periodic Table.
Non-metals are found on the right-hand side of the Periodic Table.
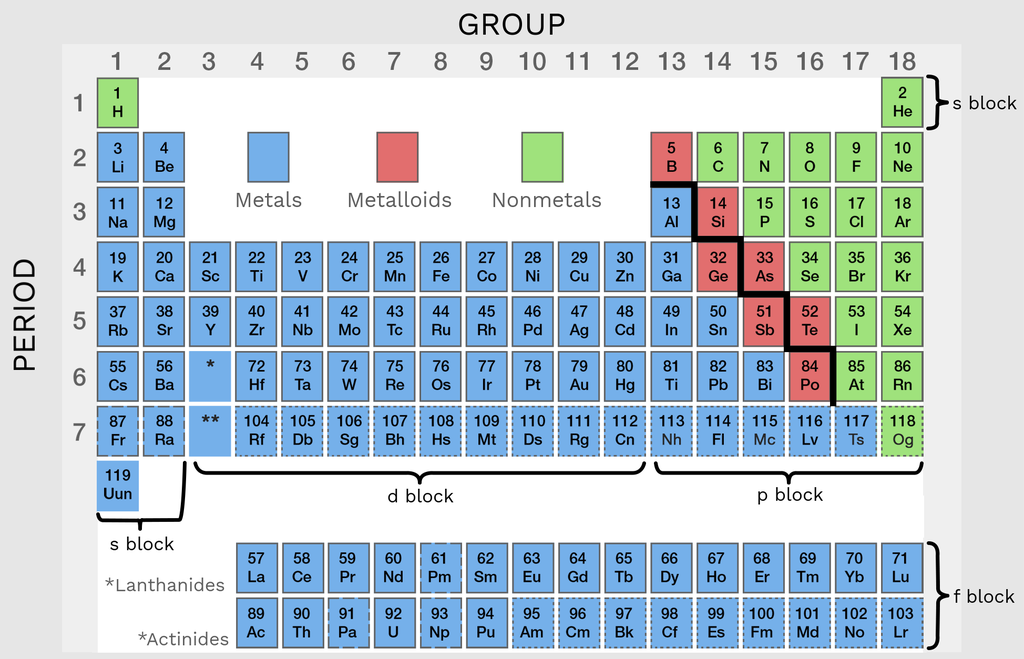
The elements are separated by a zig-zag line (sometimes called the staircase line).
Typical Positions
• Group 1 → Metals
• Group 2 → Metals
• Transition block (centre) → Metals
• Group 7 → Non-metals
• Group 0 → Non-metals
Hydrogen is a special case — it is in Group 1 but is a non-metal.
Elements close to the zig-zag line may show mixed properties, but at GCSE level they are usually classified by their general position.
Comparison Table
| Feature | Metals | Non-Metals |
|---|---|---|
| Location | Left and centre | Right side |
| Example Groups | 1, 2, Transition metals | 7 and 0 |
| General behaviour | Lose electrons | Gain or share electrons |
How to Answer in Exams
If on left → metal
If on right → non-metal
Always refer to position in the Periodic Table in your explanation.
Example 1 (Conceptual):
Element X is in Group 2. Is it a metal or a non-metal?
▶️ Answer/Explanation
Group 2 elements are on the left-hand side of the Periodic Table.
Therefore, it is a metal.
Example 2 (Numerical):
An element has atomic number 15. Identify whether it is a metal or non-metal using its position in the Periodic Table.
▶️ Answer/Explanation
Atomic number 15 is phosphorus.
Phosphorus is in Group 5 on the right-hand side.
Therefore, it is a non-metal.
Example 3 (Hard):
Explain fully how the position of calcium in the Periodic Table shows that it is a metal.
▶️ Answer/Explanation
Calcium is in Group 2 and Period 4.
Group 2 elements are found on the left-hand side of the Periodic Table.
Elements on the left-hand side are metals.
Calcium also has two outer electrons and forms positive ions by losing them.
This behaviour is typical of metals.
