Edexcel iGCSE Chemistry -1.23 Group Properties- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.23 Group Properties- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.23 Group Properties- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.23 understand why elements in the same group of the Periodic Table have similar chemical properties
1.23 Why Elements in the Same Group Have Similar Chemical Properties
Elements in the same group (vertical column) of the Periodic Table have similar chemical properties because they have the same number of electrons in their outer shell.
Key Principle
Chemical reactions involve only the outer shell (valence) electrons.
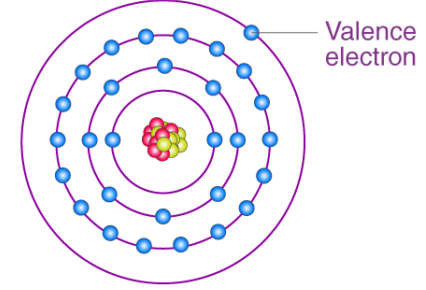
Group number (1–7) = number of outer shell electrons
Since elements in the same group have the same number of outer electrons, they tend to:
- Gain the same number of electrons
- Lose the same number of electrons
- Form similar types of ions
Example: Group 1 (Alkali Metals)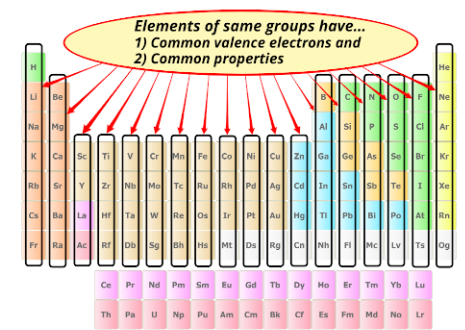
Electronic configurations:
Lithium: 2,1
Sodium: 2,8,1
Potassium: 2,8,8,1
All have 1 outer electron.
They all lose 1 electron to form \( +1 \) ions.
Example: Group 7 (Halogens)
Fluorine: 2,7
Chlorine: 2,8,7
Bromine: 2,8,18,7 (beyond first 20, but same pattern)
All have 7 outer electrons.
They all gain 1 electron to form \( -1 \) ions.
Summary Table
| Group | Outer Electrons | Typical Ion Formed | Chemical Behaviour |
|---|---|---|---|
| 1 | 1 | \( +1 \) | Lose 1 electron |
| 2 | 2 | \( +2 \) | Lose 2 electrons |
| 7 | 7 | \( -1 \) | Gain 1 electron |
| 0 | Full shell | None | Unreactive |
Why Properties Change Down a Group
Although chemical behaviour is similar, reactivity may change down a group because:
• Atoms get larger
• Outer electrons are further from the nucleus
• Attraction between nucleus and outer electrons changes
Example 1 (Conceptual):
Why do sodium and potassium react in similar ways with water?
▶️ Answer/Explanation
Both are in Group 1.
They both have 1 outer electron.
They both lose 1 electron to form +1 ions.
Therefore, they show similar chemical reactions.
Example 2 (Numerical):
An element has electronic configuration 2,8,2. Predict its chemical behaviour.
▶️ Answer/Explanation
It has 2 outer electrons.
It is in Group 2.
It will lose 2 electrons to form a \( +2 \) ion.
It will behave like other Group 2 metals.
Example 3 (Hard):
Explain fully why chlorine and bromine have similar chemical properties but different reactivities.
▶️ Answer/Explanation
Both chlorine and bromine are in Group 7.
They both have 7 outer shell electrons.
They both gain 1 electron to form -1 ions.
This gives them similar chemical properties.
However, bromine has more electron shells.
Its outer electrons are further from the nucleus.
The attraction between nucleus and outer electrons is weaker.
Therefore, bromine is less reactive than chlorine.
