Edexcel iGCSE Chemistry -1.24 Noble Gases- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.24 Noble Gases- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.24 Noble Gases- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.24 understand why the noble gases (Group 0) do not readily react
1.24 Why Noble Gases (Group 0) Do Not Readily React
The noble gases are found in Group 0 of the Periodic Table.
They are very unreactive (inert) compared with other elements.
Electronic Structure of Noble Gases
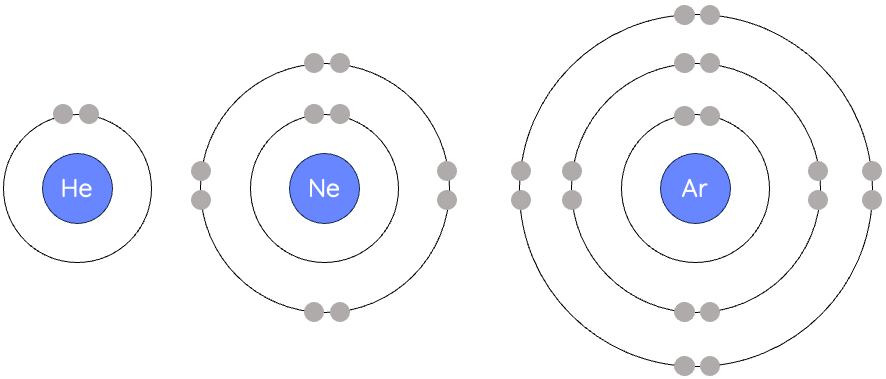
Noble gases have a full outer electron shell.
Examples of electronic configurations:
Helium: 2
Neon: 2,8
Argon: 2,8,8
A full outer shell is a very stable arrangement.
Why Full Shells Are Stable
Atoms react in order to achieve a full outer shell.
Noble gases already have:
• Maximum number of outer electrons
• No tendency to gain electrons
• No tendency to lose electrons
• No need to share electrons
Therefore, they do not easily form ions or compounds.
Key GCSE Explanation
Noble gases are unreactive because they have a full outer electron shell, which is a stable electronic configuration.
Comparison with Other Groups
| Group | Outer Electrons | Tendency | Reactivity |
|---|---|---|---|
| 1 | 1 | Lose 1 electron | Very reactive |
| 7 | 7 | Gain 1 electron | Very reactive |
| 0 | Full shell | No gain or loss | Very unreactive |
Important Note
Although noble gases are very unreactive, heavier noble gases (like xenon) can form compounds under extreme conditions. However, at GCSE level they are considered unreactive.
Example 1 (Conceptual):
Why does neon not react with other elements?
▶️ Answer/Explanation
Neon has electronic configuration 2,8.
Its outer shell is full.
It does not need to gain or lose electrons.
Therefore, it is unreactive.
Example 2 (Numerical):
An element has electronic configuration 2,8,8. Predict its reactivity.
▶️ Answer/Explanation
It has a full outer shell.
It is a Group 0 element.
It will be very unreactive.
Example 3 (Hard):
Explain fully why Group 0 elements do not readily form ions or compounds.
▶️ Answer/Explanation
Group 0 elements have full outer electron shells.
A full outer shell is a stable electronic configuration.
They do not need to gain electrons to become stable.
They do not need to lose electrons either.
Therefore, they have no strong tendency to form ions.
As a result, they do not readily form compounds.
