Edexcel iGCSE Chemistry -1.25 Word and Symbol Equations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.25 Word and Symbol Equations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.25 Word and Symbol Equations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.25 write word equations and balanced chemical equations (including state symbols):
• for reactions studied in this specification
• for unfamiliar reactions where suitable information is provided
1.25 Writing Word Equations and Balanced Chemical Equations (Including State Symbols)
Chemical equations show what happens in a chemical reaction.

At GCSE level, you must be able to write:
- Word equations
- Balanced symbol equations
- Equations with state symbols
1. Word Equations
A word equation uses the names of substances.
Reactants → Products
Example:
Magnesium + oxygen → magnesium oxide
2. Balanced Chemical Equations
A balanced chemical equation uses symbols and formulae.
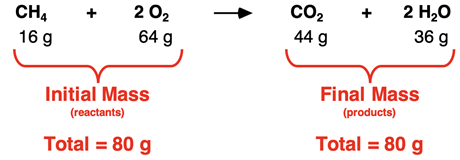
It must follow the Law of Conservation of Mass:
Number of atoms of each element must be the same on both sides.
Example:
\( 2\mathrm{Mg} + \mathrm{O_2} \rightarrow 2\mathrm{MgO} \)
3. State Symbols
State symbols show the physical state of each substance:
- \( (\mathrm{s}) \) solid
- \( (\mathrm{l}) \) liquid
- \( (\mathrm{g}) \) gas
- \( (\mathrm{aq}) \) aqueous (dissolved in water)
Example with state symbols:
\( 2\mathrm{Mg}(\mathrm{s}) + \mathrm{O_2}(\mathrm{g}) \rightarrow 2\mathrm{MgO}(\mathrm{s}) \)
Steps for Balancing Equations
1. Write correct chemical formulae.
2. Count atoms on each side.
3. Add coefficients (numbers in front).
4. Do NOT change chemical formulae.
Common Reaction Types from This Specification
| Reaction Type | General Word Equation |
|---|---|
| Metal + oxygen | Metal + oxygen → metal oxide |
| Metal + acid | Metal + acid → salt + hydrogen |
| Acid + base | Acid + base → salt + water |
| Combustion | Substance + oxygen → oxide(s) |
Example 1 (Conceptual):
Why must chemical equations be balanced?
▶️ Answer/Explanation
Atoms cannot be created or destroyed in a chemical reaction.
The number of each type of atom must be the same on both sides.
This follows the law of conservation of mass.
Example 2 (Numerical):
Balance the equation:
\( \mathrm{H_2} + \mathrm{O_2} \rightarrow \mathrm{H_2O} \)
▶️ Answer/Explanation
\( 2\mathrm{H_2} + \mathrm{O_2} \rightarrow 2\mathrm{H_2O} \)
Now there are 4 hydrogen atoms and 2 oxygen atoms on both sides.
Example 3 (Hard):
Calcium reacts with hydrochloric acid to form calcium chloride and hydrogen gas. Write the balanced chemical equation including state symbols.
▶️ Answer/Explanation
Word equation:
Calcium + hydrochloric acid → calcium chloride + hydrogen
Balanced symbol equation:
\( \mathrm{Ca}(\mathrm{s}) + 2\mathrm{HCl}(\mathrm{aq}) \rightarrow \mathrm{CaCl_2}(\mathrm{aq}) + \mathrm{H_2}(\mathrm{g}) \)
There is 1 Ca, 2 H and 2 Cl on both sides.
