Edexcel iGCSE Chemistry -1.26Relative Formula Mass- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.26Relative Formula Mass- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.26Relative Formula Mass- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.26 calculate relative formula masses (including relative molecular masses) (Mr) from relative atomic masses (Ar)
1.26 Calculating Relative Formula Mass (Mr) and Relative Molecular Mass (Mr)
The relative formula mass (Mr) is the sum of the relative atomic masses (Ar) of all the atoms in a formula.
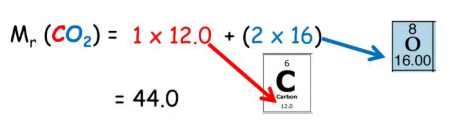
For simple covalent molecules, it is also called relative molecular mass.
Formula
\( M_r = \text{Sum of all } A_r \text{ values in the formula} \)
You must multiply each Ar by the number of that atom present.
Step-by-Step Method
1. Write down the formula.
2. Identify how many of each atom there are.
3. Multiply each Ar by the number of atoms.
4. Add all values together.
Example Ar Values (from Periodic Table)
| Element | Ar |
|---|---|
| H | 1 |
| C | 12 |
| O | 16 |
| Na | 23 |
| Cl | 35.5 |
Difference Between Formula Mass and Molecular Mass
| Type | Used For | Example |
|---|---|---|
| Relative molecular mass | Covalent substances | \( \mathrm{H_2O} \), \( \mathrm{CO_2} \) |
| Relative formula mass | Ionic compounds | \( \mathrm{NaCl} \), \( \mathrm{MgO} \) |
Example 1 (Conceptual):
Why does chlorine contribute 35.5 and not 35 to calculations?
▶️ Answer/Explanation
The value 35.5 is the relative atomic mass.
It is a weighted average of chlorine isotopes.
We must use Ar values from the Periodic Table.
Example 2 (Numerical):
Calculate the Mr of water, \( \mathrm{H_2O} \).
▶️ Answer/Explanation
Hydrogen: \( 2 \times 1 = 2 \) Oxygen: \( 1 \times 16 = 16 \)
\( M_r = 2 + 16 = 18 \)
Example 3 (Hard):
Calculate the Mr of calcium hydroxide, \( \mathrm{Ca(OH)_2} \).
▶️ Answer/Explanation
Ar values:
Ca = 40 O = 16 H = 1
There are two OH groups.
\( M_r = 40 + 2(16 + 1) \)
\( = 40 + 2(17) \)
\( = 40 + 34 = 74 \)
