Edexcel iGCSE Chemistry -1.28–1.29 Amount of Substance and Reacting Mass Calculations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.28–1.29 Amount of Substance and Reacting Mass Calculations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.28–1.29 Amount of Substance and Reacting Mass Calculations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.28 understand how to carry out calculations involving amount of substance, relative atomic mass (Ar) and relative formula mass (Mr)
1.29 calculate reacting masses using experimental data and chemical equations
1.28 Calculations Involving Amount of Substance, Ar and Mr
At GCSE level, you must be able to calculate the amount of substance (in moles) using relative atomic mass (Ar) or relative formula mass (Mr).
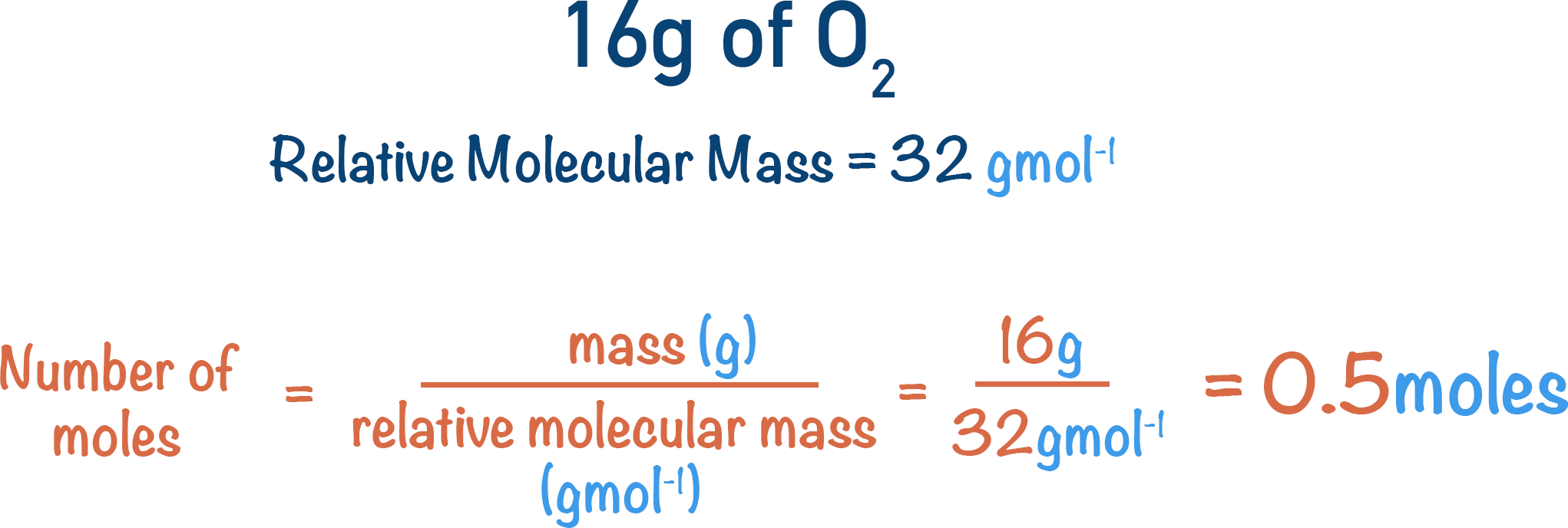
Core Formula
\( n = \dfrac{m}{M_r} \)
Where:
\( n \) = number of moles (mol) \( m \) = mass (g) \( M_r \) = relative formula mass
You may also rearrange the formula:
\( m = n \times M_r \)
Using Ar and Mr
For single elements:
Use Ar value (e.g. Mg has Ar = 24)
For compounds:
First calculate Mr Then substitute into the formula
Step-by-Step Problem Method
1. Write down the formula.
2. Calculate Mr (if needed).
3. Use \( n = \dfrac{m}{M_r} \).
4. Rearrange if needed.
Example 1 (Conceptual):
Why must we calculate Mr before finding moles of a compound?
▶️ Answer/Explanation
The formula \( n = \dfrac{m}{M_r} \) requires the relative formula mass.
Compounds contain more than one type of atom.
Therefore, we must first add the Ar values to find Mr.
Example 2 (Numerical):
Calculate the number of moles in 12 g of carbon. (Ar of C = 12)
▶️ Answer/Explanation
\( n = \dfrac{12}{12} \)
\( = 1 \text{ mol} \)
Example 3 (Hard):
Calculate the number of moles in 49 g of sulfuric acid, \( \mathrm{H_2SO_4} \).
(Ar values: H = 1, S = 32, O = 16)
▶️ Answer/Explanation
Step 1: Calculate Mr
\( M_r = (2 \times 1) + 32 + (4 \times 16) \)
\( = 2 + 32 + 64 = 98 \)
Step 2: Use the mole formula
\( n = \dfrac{49}{98} \)
\( = 0.5 \text{ mol} \)
Answer: 0.5 mol
1.29 Calculating Reacting Masses Using Experimental Data and Chemical Equations
Reacting mass calculations use:
- A balanced chemical equation
- The mole concept
- Experimental mass data
You use the balanced equation to find the mole ratio between substances.
Core Method for Reacting Mass Calculations
1. Write the balanced equation.
2. Calculate moles of the known substance.
3. Use the mole ratio from the equation.
4. Convert moles back to mass (if needed).
Important Formulae
\( n = \dfrac{m}{M_r} \)
\( m = n \times M_r \)
Using Mole Ratios
In a balanced equation, coefficients give mole ratios.
Example: \( 2\mathrm{Mg} + \mathrm{O_2} \rightarrow 2\mathrm{MgO} \)
This means:
2 moles Mg react with 1 mole O₂ 2 moles Mg produce 2 moles MgO
Example 1 (Conceptual):
Why must a chemical equation be balanced before calculating reacting masses?
▶️ Answer/Explanation
The mole ratio comes from the balanced equation.
If the equation is not balanced, the ratio of reacting particles would be incorrect.
This would give the wrong mass calculation.
Example 2 (Numerical):
Calculate the mass of magnesium oxide formed when 12 g of magnesium reacts with excess oxygen.
Equation:
\( 2\mathrm{Mg} + \mathrm{O_2} \rightarrow 2\mathrm{MgO} \)
(Ar: Mg = 24, O = 16)
▶️ Answer/Explanation
Step 1: Calculate moles of Mg
\( n = \dfrac{12}{24} = 0.5 \text{ mol} \)
Step 2: Use mole ratio
2 mol Mg → 2 mol MgO Ratio is 1:1
0.5 mol Mg produces 0.5 mol MgO
Step 3: Convert to mass
Mr of MgO = 24 + 16 = 40
Mass = \( 0.5 \times 40 = 20 \text{ g} \)
Answer: 20 g
Example 3 (Hard):
25 g of calcium carbonate decomposes on heating.
\( \mathrm{CaCO_3}(\mathrm{s}) \rightarrow \mathrm{CaO}(\mathrm{s}) + \mathrm{CO_2}(\mathrm{g}) \)
Calculate the mass of carbon dioxide produced.
(Ar: Ca=40, C=12, O=16)
▶️ Answer/Explanation
Step 1: Calculate Mr of CaCO₃
\( M_r = 40 + 12 + (3 \times 16) \)
\( = 40 + 12 + 48 = 100 \)
Step 2: Calculate moles of CaCO₃
\( n = \dfrac{25}{100} = 0.25 \text{ mol} \)
Step 3: Mole ratio
1 mol CaCO₃ → 1 mol CO₂
0.25 mol CaCO₃ produces 0.25 mol CO₂
Step 4: Convert to mass
Mr of CO₂ = 12 + (2 × 16) = 44
Mass = \( 0.25 \times 44 = 11 \text{ g} \)
Answer: 11 g of CO₂
