Edexcel iGCSE Chemistry -1.3 Diffusion- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.3 Diffusion- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.3 Diffusion- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.3 understand how the results of experiments involving the dilution of coloured solutions and diffusion of gases can be explained
1.3 Explaining Dilution of Coloured Solutions and Diffusion of Gases
Experiments involving coloured solutions and gases provide evidence for the particle model of matter. They show that particles are in constant random motion and that substances mix because particles move and spread out.
Dilution of a Coloured Solution
In a typical experiment, a coloured solution (such as copper sulfate solution or food colouring in water) becomes lighter when more water is added.
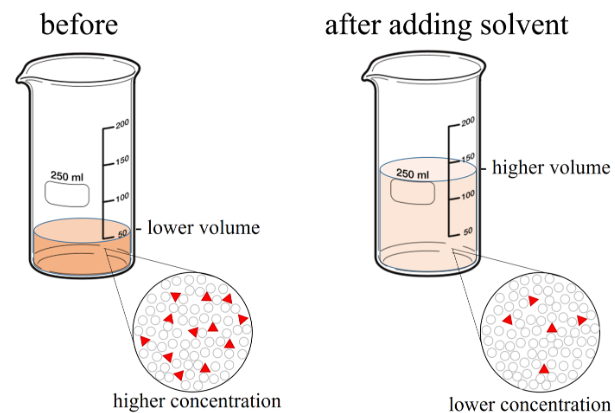
This can be explained using particle theory:
- The coloured particles (solute particles) do not disappear.
- When more solvent is added, the same number of coloured particles are spread through a larger volume.
- The concentration decreases (fewer coloured particles per unit volume).
The lighter colour is evidence that the particles are more widely spaced.
Diffusion of Gases
Diffusion is the net movement of particles from a region of high concentration to a region of low concentration.

A common experiment involves ammonia gas and hydrogen chloride gas diffusing from opposite ends of a tube and forming a white ring of ammonium chloride.
This happens because:
• Gas particles move rapidly and randomly.
• They spread out from areas of high concentration.
• Lighter particles move faster than heavier particles at the same temperature.
Why Diffusion is Faster in Gases than in Liquids
| Reason | Gases | Liquids |
|---|---|---|
| Particle spacing | Far apart | Close together |
| Particle speed | Very fast | Slower |
| Attractive forces | Very weak | Stronger |
Exam Tip
When explaining dilution or diffusion, always refer to:
• Particle movement (random motion)
• Concentration difference
• Particle spacing
Example 1 (Conceptual):
A crystal of potassium manganate(VII) is placed in water and the purple colour slowly spreads. Explain why stirring is not required.
▶️ Answer/Explanation
Water and manganate particles are in constant random motion.
The manganate particles move from an area of high concentration near the crystal to areas of lower concentration in the water.
This process is diffusion, so stirring is not needed.
Example 2 (Numerical):
A solution contains \( 8 \, \mathrm{g} \) of dye in \( 200 \, \mathrm{cm^3} \) of water. It is diluted to \( 400 \, \mathrm{cm^3} \).
Calculate the new concentration in \( \mathrm{g/cm^3} \).
▶️ Answer/Explanation
Initial concentration:
\( \dfrac{8}{200} = 0.04 \, \mathrm{g/cm^3} \)
After dilution:
\( \dfrac{8}{400} = 0.02 \, \mathrm{g/cm^3} \)
The concentration halves because the same mass is spread through double the volume.
Example 3 (Hard ):
In a long glass tube, ammonia gas is placed at one end and hydrogen chloride gas at the other. A white ring forms closer to the hydrogen chloride end. Explain fully using particle theory.
▶️ Answer/Explanation
Both gases diffuse because their particles move randomly and rapidly.
Diffusion occurs from areas of high concentration to low concentration.
Ammonia has a lower relative molecular mass than hydrogen chloride, so its particles move faster at the same temperature.
Because ammonia diffuses faster, it travels a greater distance in the same time.
Therefore, the white ammonium chloride ring forms closer to the hydrogen chloride end.
