Edexcel iGCSE Chemistry -1.31 Preparation of Simple Compounds- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.31 Preparation of Simple Compounds- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.31 Preparation of Simple Compounds- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.31 understand how the formulae of simple compounds can be obtained experimentally, including metal oxides, water and salts containing water of crystallisation
1.31 Determining the Formulae of Simple Compounds Experimentally
The formula of a compound can be determined by measuring masses before and after a reaction.
This allows us to calculate the ratio of moles of each element in the compound.
General Experimental Method
- Measure initial mass.
- React substance.
- Measure final mass.
- Calculate mass change.
- Convert masses to moles.
- Find simplest whole-number ratio.
1. Determining the Formula of a Metal Oxide
A metal is heated in oxygen.
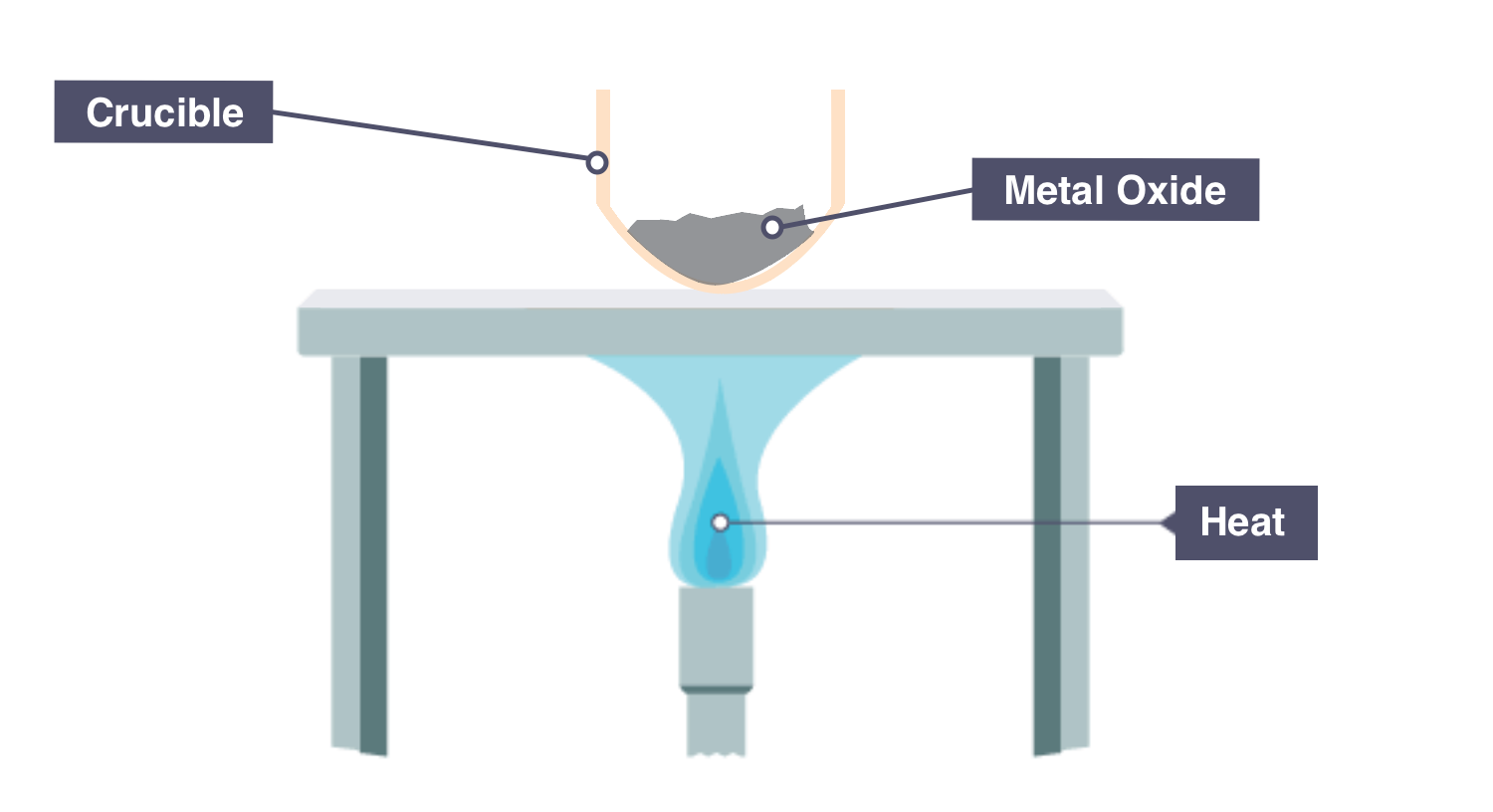
Mass increases because oxygen combines with the metal.
Mass of oxygen = final mass − initial mass
Convert both metal and oxygen to moles and find ratio.
2. Determining the Formula of Water
Hydrogen reacts with oxygen to form water.
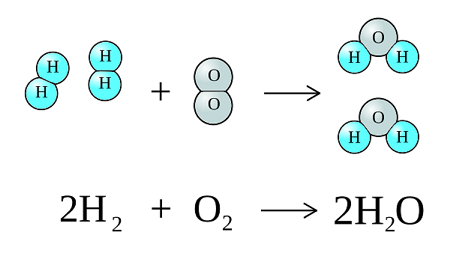
Experimental results show the ratio of hydrogen to oxygen is 2:1 in moles.
Formula = \( \mathrm{H_2O} \)
3. Determining Water of Crystallisation (Hydrated Salts)
Some salts contain water molecules trapped in their crystal structure.
Example: hydrated copper sulfate.
When heated, water is removed and mass decreases.
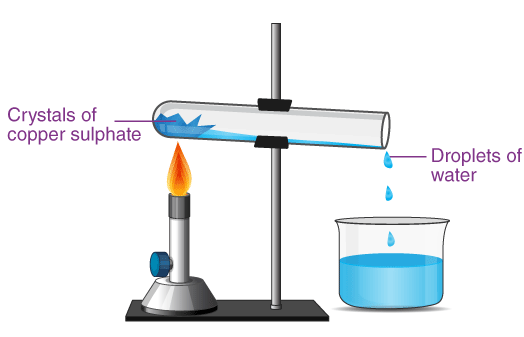
Mass of water lost = initial mass − final mass
You calculate moles of salt and moles of water to find the value of \( x \) in:
\( \mathrm{Salt \cdot xH_2O} \)
Summary Comparison
| Compound Type | Mass Change | What Is Calculated |
|---|---|---|
| Metal oxide | Mass increases | Oxygen gained |
| Water | Ratio measured | H:O mole ratio |
| Hydrated salt | Mass decreases | Water lost |
Example 1 (Conceptual):
Why does the mass increase when magnesium is heated in air?
▶️ Answer/Explanation
Magnesium reacts with oxygen.
Oxygen atoms combine with magnesium atoms.
The added oxygen increases the mass.
Example 2 (Numerical):
A student heats 2.4 g of magnesium to form 4.0 g of magnesium oxide. Determine the empirical formula.
(Ar: Mg = 24, O = 16)
▶️ Answer/Explanation
Step 1: Mass of oxygen
4.0 − 2.4 = 1.6 g
Step 2: Convert to moles
Mg: \( \dfrac{2.4}{24} = 0.10 \)
O: \( \dfrac{1.6}{16} = 0.10 \)
Step 3: Ratio
1 : 1
Formula = MgO
Example 3 (Hard):
5.0 g of hydrated copper sulfate is heated until constant mass. The final mass is 3.2 g. Determine the value of \( x \) in \( \mathrm{CuSO_4 \cdot xH_2O} \).
(Ar: Cu=64, S=32, O=16, H=1)
▶️ Answer/Explanation
Step 1: Mass of water lost
5.0 − 3.2 = 1.8 g
Step 2: Moles of CuSO₄
\( M_r(\mathrm{CuSO_4}) = 64 + 32 + 64 = 160 \)
\( \dfrac{3.2}{160} = 0.02 \text{ mol} \)
Step 3: Moles of water
\( M_r(\mathrm{H_2O}) = 18 \)
\( \dfrac{1.8}{18} = 0.10 \text{ mol} \)
Step 4: Ratio
0.02 : 0.10
Divide by 0.02 → 1 : 5
x = 5
