Edexcel iGCSE Chemistry -1.32–1.33 Empirical and Molecular Formulae and Calculations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.32–1.33 Empirical and Molecular Formulae and Calculations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.32–1.33 Empirical and Molecular Formulae and Calculations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.32 know what is meant by the terms empirical formula and molecular formula
1.33 calculate empirical and molecular formulae from experimental data
1.32 Empirical Formula and Molecular Formula
Chemical formulae show the number and ratio of atoms in a substance.
There are two important types of formula you must know at GCSE level:

- Empirical formula
- Molecular formula
Empirical Formula
The empirical formula shows the simplest whole-number ratio of atoms in a compound.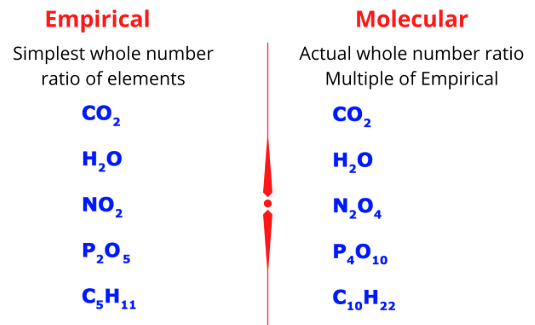
Example:
Hydrogen peroxide has molecular formula \( \mathrm{H_2O_2} \).
The simplest ratio 2:2 simplifies to 1:1.
Empirical formula = \( \mathrm{HO} \)
Molecular Formula
The molecular formula shows the actual number of atoms of each element in one molecule.
Example:
Glucose = \( \mathrm{C_6H_{12}O_6} \)
Its empirical formula is:
Divide by 6 → \( \mathrm{CH_2O} \)
Key Difference
| Feature | Empirical Formula | Molecular Formula |
|---|---|---|
| Shows | Simplest ratio | Actual number of atoms |
| May be simplified? | Yes | No |
| Example | CH₂O | C₆H₁₂O₆ |
Important Relationship
The molecular formula is always a multiple of the empirical formula.
\( \text{Molecular formula} = (\text{Empirical formula})_n \)
Example 1 (Conceptual):
Why can different compounds have the same empirical formula?
▶️ Answer/Explanation
The empirical formula only shows the simplest ratio.
Different compounds can have actual numbers that reduce to the same ratio.
For example, \( \mathrm{CH_2O} \) could represent formaldehyde or glucose in simplest form.
Example 2 (Numerical):
Find the empirical formula of \( \mathrm{C_2H_4} \).
▶️ Answer/Explanation
Simplify the ratio 2:4.
Divide by 2 → 1:2
Empirical formula = \( \mathrm{CH_2} \)
Example 3 (Hard):
A compound has empirical formula \( \mathrm{CH_2} \) and Mr = 56. Determine its molecular formula.
▶️ Answer/Explanation
Step 1: Calculate empirical formula mass
\( M_r(\mathrm{CH_2}) = 12 + (2 \times 1) = 14 \)
Step 2: Divide molecular mass by empirical mass
\( \dfrac{56}{14} = 4 \)
Step 3: Multiply formula by 4
\( (\mathrm{CH_2})_4 = \mathrm{C_4H_8} \)
Molecular formula = \( \mathrm{C_4H_8} \)
1.33 Calculating Empirical and Molecular Formulae from Experimental Data
Experimental data (usually masses or percentage composition) can be used to determine:
- The empirical formula (simplest ratio)
- The molecular formula (actual number of atoms)
Method to Find Empirical Formula from Mass Data
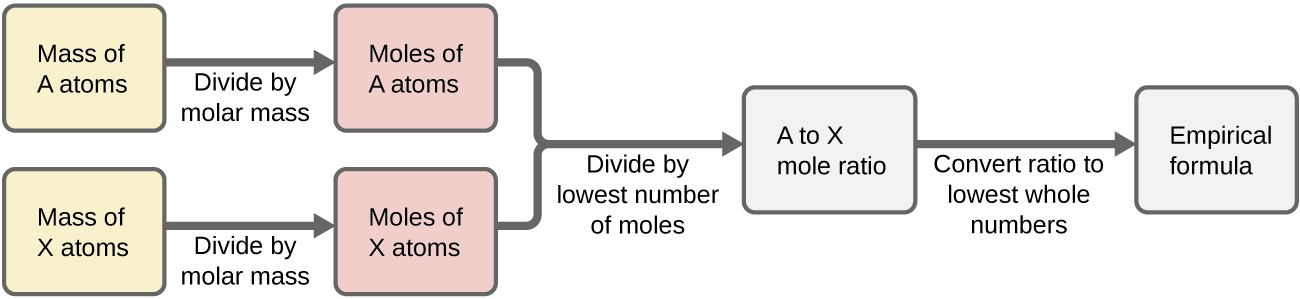
1. Write down the masses of each element.
2. Convert mass to moles (divide by Ar).
3. Divide all mole values by the smallest mole value.
4. Round to the nearest whole number.
If Percentages Are Given
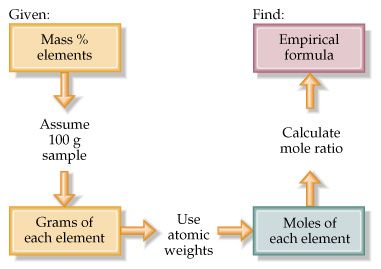
Assume 100 g of the compound.
This means:
Percentage value = mass in grams
Then follow the same mole steps.
Finding Molecular Formula
Once the empirical formula is known:
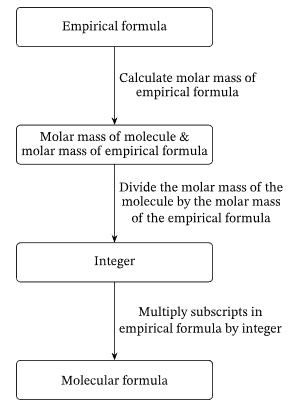
- Calculate empirical formula mass.
- Divide given Mr by empirical formula mass.
- Multiply empirical formula by that number.
\( \text{Molecular formula} = (\text{Empirical formula})_n \)
Example 1 (Conceptual):
Why must mole ratios be used instead of mass ratios when determining empirical formula?
▶️ Answer/Explanation
Chemical formulae are based on numbers of atoms.
Masses depend on atomic mass.
Moles convert mass into number of particles.
Therefore, mole ratios must be used.
Example 2 (Numerical):
A compound contains 40% carbon, 6.7% hydrogen and 53.3% oxygen. Determine the empirical formula.
(Ar: C=12, H=1, O=16)
▶️ Answer/Explanation
Step 1: Assume 100 g
C = 40 g H = 6.7 g O = 53.3 g
Step 2: Convert to moles
C: \( \dfrac{40}{12} = 3.33 \) H: \( \dfrac{6.7}{1} = 6.7 \) O: \( \dfrac{53.3}{16} = 3.33 \)
Step 3: Divide by smallest (3.33)
C: 1 H: 2 O: 1
Empirical formula = \( \mathrm{CH_2O} \)
Example 3 (Hard):
A compound contains 52.2% carbon, 13.0% hydrogen and 34.8% oxygen. Its Mr is 46. Determine its molecular formula.
(Ar: C=12, H=1, O=16)
▶️ Answer/Explanation
Step 1: Assume 100 g
C: 52.2 g H: 13.0 g O: 34.8 g
Step 2: Convert to moles
C: \( \dfrac{52.2}{12} = 4.35 \) H: \( \dfrac{13.0}{1} = 13.0 \) O: \( \dfrac{34.8}{16} = 2.18 \)
Step 3: Divide by smallest (2.18)
C: 2 H: 6 O: 1
Empirical formula = \( \mathrm{C_2H_6O} \)
Step 4: Empirical mass
\( 2(12) + 6(1) + 16 = 24 + 6 + 16 = 46 \)
Step 5: Compare with given Mr
\( \dfrac{46}{46} = 1 \)
Molecular formula = \( \mathrm{C_2H_6O} \)
