Edexcel iGCSE Chemistry -1.34C Concentration Calculations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.34C Concentration Calculations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.34C Concentration Calculations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.34C understand how to carry out calculations involving amount of substance, volume and concentration (in mol/dm³) of solution
1.34C Calculations Involving Amount of Substance, Volume and Concentration (mol/dm3)
In solution chemistry, you must be able to calculate relationships between:
- Amount of substance (moles, \( n \))
- Volume (dm3)
- Concentration (mol/dm3)
Key Formula
\( c = \dfrac{n}{V} \)
Where:
\( c \) = concentration (mol/dm3)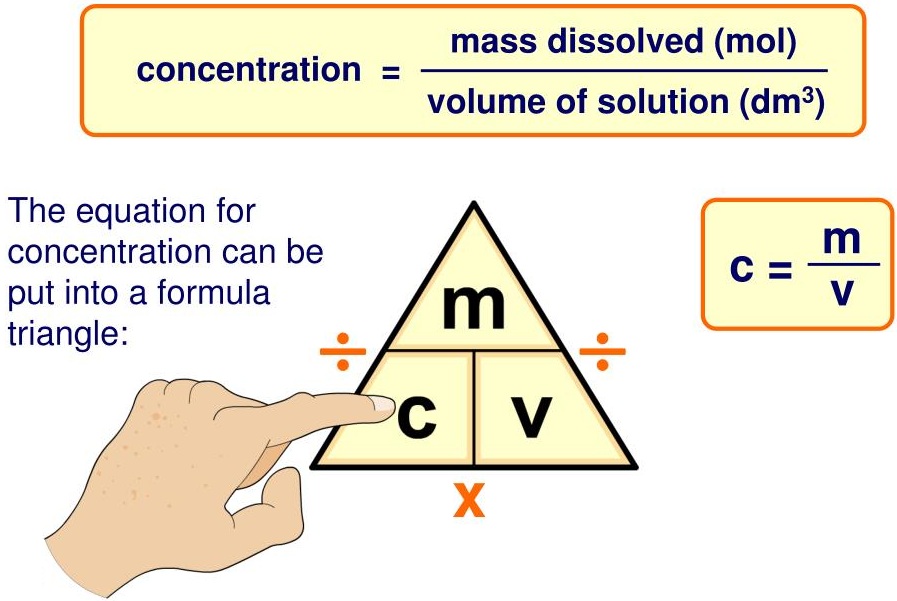
\( n \) = number of moles (mol)
\( V \) = volume (dm3)
Rearrangements:
\( n = c \times V \)
\( V = \dfrac{n}{c} \)
Important Unit Conversion
Volume must be in dm3.
\( 1000 \text{ cm}^3 = 1 \text{ dm}^3 \)
To convert: divide cm3 by 1000
Link with Mass Calculations
If mass is given:
Step 1: \( n = \dfrac{m}{M_r} \)
Step 2: Use \( c = \dfrac{n}{V} \)
| Quantity | Symbol | Units |
|---|---|---|
| Moles | \( n \) | mol |
| Concentration | \( c \) | mol/dm3 |
| Volume | \( V \) | dm3 |
Example 1 (Conceptual):
Why must volume be converted to dm3 before using the concentration formula?
▶️ Answer/Explanation
Concentration is measured in mol per dm3.
If volume is in cm3, the units will be incorrect.
Therefore, volume must be converted to dm3.
Example 2 (Numerical):
Calculate the concentration of a solution containing 0.50 mol of sodium chloride in 250 cm3 of solution.
▶️ Answer/Explanation
Step 1: Convert volume
\( 250 \div 1000 = 0.250 \text{ dm}^3 \)
Step 2: Use formula
\( c = \dfrac{0.50}{0.250} \)
\( = 2.0 \text{ mol/dm}^3 \)
Answer: 2.0 mol/dm3
Example 3 (Hard):
4.0 g of sodium hydroxide (Mr = 40) is dissolved to make 500 cm3 of solution. Calculate the concentration in mol/dm3.
▶️ Answer/Explanation
Step 1: Calculate moles
\( n = \dfrac{4.0}{40} = 0.10 \text{ mol} \)
Step 2: Convert volume
\( 500 \div 1000 = 0.50 \text{ dm}^3 \)
Step 3: Calculate concentration
\( c = \dfrac{0.10}{0.50} \)
\( = 0.20 \text{ mol/dm}^3 \)
Answer: 0.20 mol/dm3
