Edexcel iGCSE Chemistry -1.35C Gas Volume Calculations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.35C Gas Volume Calculations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.35C Gas Volume Calculations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.35C understand how to carry out calculations involving gas volumes and the molar volume of a gas (24 dm³ and 24 000 cm³ at room temperature and pressure (rtp))
1.35C Gas Volumes and Molar Volume at rtp
At room temperature and pressure (rtp), one mole of any gas occupies a fixed volume.
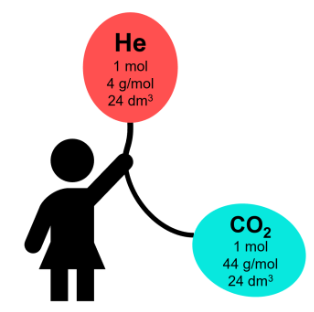
Molar Volume of a Gas at rtp
\( 1 \text{ mol of gas} = 24 \text{ dm}^3 \)
\( 1 \text{ mol of gas} = 24\,000 \text{ cm}^3 \)
This applies to all gases at rtp.
Key Formula
\( n = \dfrac{V}{24} \) (if volume in dm3)
\( n = \dfrac{V}{24\,000} \) (if volume in cm3)
Rearranged:
\( V = n \times 24 \)
Link With Chemical Equations
For gas reactions:
1. Use the balanced equation.
2. Use mole ratios.
3. Convert moles to gas volume using 24 dm3.
| Volume Unit | Formula to Use |
|---|---|
| dm3 | \( n = \dfrac{V}{24} \) |
| cm3 | \( n = \dfrac{V}{24\,000} \) |
Example 1 (Conceptual):
Why do all gases have the same molar volume at rtp?
▶️ Answer/Explanation
At rtp, gas particles are far apart.
The volume depends on number of particles, not their identity.
One mole always contains the same number of particles.
Therefore, all gases occupy 24 dm3 per mole.
Example 2 (Numerical ):
Calculate the volume of 0.50 mol of oxygen gas at rtp.
▶️ Answer/Explanation
\( V = 0.50 \times 24 \)
\( = 12 \text{ dm}^3 \)
Answer: 12 dm3
Example 3 (Hard):
Calcium reacts with hydrochloric acid:
\( \mathrm{Ca} + 2\mathrm{HCl} \rightarrow \mathrm{CaCl_2} + \mathrm{H_2} \)
If 2.4 g of calcium reacts, calculate the volume of hydrogen gas produced at rtp.
(Ar of Ca = 40)
▶️ Answer/Explanation
Step 1: Calculate moles of calcium
\( n = \dfrac{2.4}{40} = 0.06 \text{ mol} \)
Step 2: Use mole ratio
1 mol Ca → 1 mol H₂
0.06 mol Ca produces 0.06 mol H₂
Step 3: Convert to volume
\( V = 0.06 \times 24 \)
\( = 1.44 \text{ dm}^3 \)
Answer: 1.44 dm3
