Edexcel iGCSE Chemistry -1.36 Practical: Formula of a Metal Oxide- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.36 Practical: Formula of a Metal Oxide- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.36 Practical: Formula of a Metal Oxide- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide)
1.36 Practical: Determining the Formula of a Metal Oxide
The empirical formula of a metal oxide can be determined experimentally by:
- Combustion (e.g. magnesium oxide)
- Reduction (e.g. copper(II) oxide)
In both cases, mass measurements are used to calculate the mole ratio of metal to oxygen.
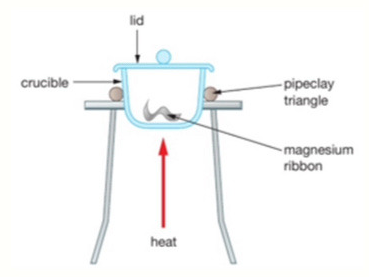
Method 1: Combustion of Magnesium
Magnesium is heated in a crucible and reacts with oxygen in air.
\( 2\mathrm{Mg} + \mathrm{O_2} \rightarrow 2\mathrm{MgO} \)
Measurements Taken:
1. Mass of empty crucible
2. Mass of crucible + magnesium
3. Mass of crucible + magnesium oxide
Key Calculation:
Mass of magnesium = (crucible + Mg) − crucible Mass of magnesium oxide
= (crucible + MgO) − crucible Mass of oxygen
= mass of MgO − mass of Mg
Convert masses of Mg and O to moles and find simplest ratio.
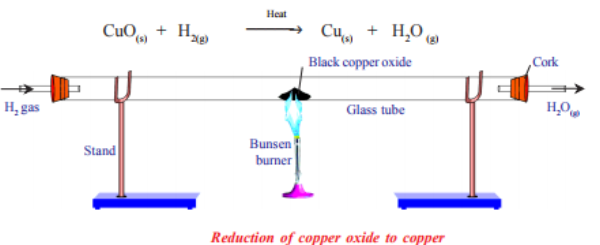
Method 2: Reduction of Copper(II) Oxide
Copper(II) oxide is heated with a reducing agent (e.g. hydrogen or carbon).
\( \mathrm{CuO} + \mathrm{H_2} \rightarrow \mathrm{Cu} + \mathrm{H_2O} \)
Oxygen is removed from CuO.
Measurements Taken:
1. Mass of copper(II) oxide before heating
2. Mass of copper after heating
Key Calculation:
Mass of oxygen removed = initial mass − final mass
Convert Cu and O masses to moles and determine ratio.
| Method | Mass Change | What Is Calculated |
|---|---|---|
| Combustion | Mass increases | Oxygen gained |
| Reduction | Mass decreases | Oxygen removed |
Sources of Error (Exam Evaluation)
• Product lost during heating
• Incomplete reaction
• Magnesium reacting with nitrogen
• Not heating to constant mass
Example 1 (Conceptual):
Why must the crucible be reheated until constant mass?
▶️ Answer/Explanation
To ensure the reaction has completed.
If mass is still changing, oxygen may still be reacting.
Constant mass confirms full reaction.
Example 2 (Numerical):
Mass of magnesium = 1.2 g Mass of magnesium oxide = 2.0 g Determine the empirical formula.
(Ar: Mg=24, O=16)
▶️ Answer/Explanation
Mass of oxygen
2.0 − 1.2 = 0.8 g
Moles
Mg: \( \dfrac{1.2}{24} = 0.05 \)
O: \( \dfrac{0.8}{16} = 0.05 \)
Ratio = 1:1 → MgO
Example 3 (Hard):
4.0 g of copper(II) oxide is reduced to 3.2 g of copper. Determine the empirical formula of the oxide.
(Ar: Cu=64, O=16)
▶️ Answer/Explanation
Mass of oxygen removed
4.0 − 3.2 = 0.8 g
Moles
Cu: \( \dfrac{3.2}{64} = 0.05 \)
O: \( \dfrac{0.8}{16} = 0.05 \)
Ratio = 1:1 → CuO
