Edexcel iGCSE Chemistry -1.37–1.38 Formation and Charges of Ions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.37–1.38 Formation and Charges of Ions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.37–1.38 Formation and Charges of Ions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.37 understand how ions are formed by electron loss or gain
1.38 know the charges of these ions:
• metals in Groups 1, 2 and 3
• non-metals in Groups 5, 6 and 7
• Ag⁺, Cu²⁺, Fe²⁺, Fe³⁺, Pb²⁺, Zn²⁺
• hydrogen (H⁺), hydroxide (OH⁻), ammonium (NH₄⁺), carbonate (CO₃²⁻), nitrate (NO₃⁻), sulfate (SO₄²⁻)
1.37 Formation of Ions by Electron Loss or Gain
An ion is a charged particle formed when an atom gains or loses electrons.
Only electrons are transferred — the nucleus does not change.
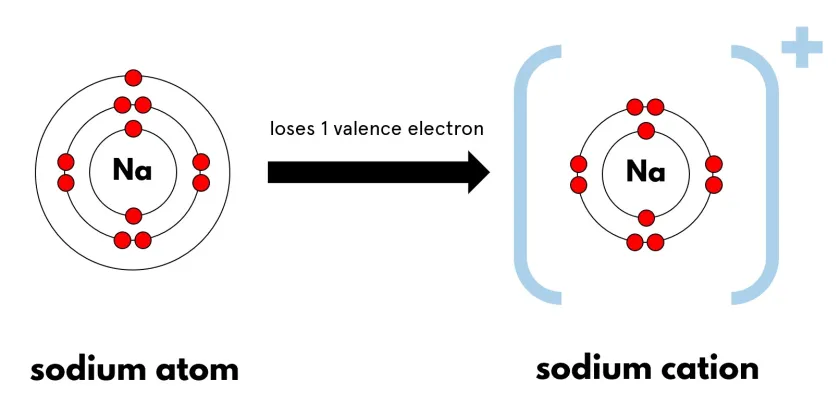
Formation of Positive Ions (Cations)
When an atom loses electrons, it forms a positive ion.
This happens because:
Protons > Electrons → Overall positive charge
Example: Sodium
Electronic configuration: 2,8,1 Loses 1 electron → 2,8
\( \mathrm{Na} \rightarrow \mathrm{Na^+} + e^- \)
Sodium forms a \( \mathrm{Na^+} \) ion.
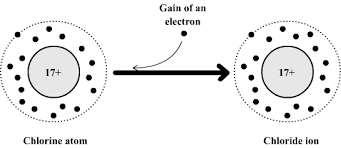
Formation of Negative Ions (Anions)
When an atom gains electrons, it forms a negative ion.
This happens because:
Electrons > Protons → Overall negative charge
Example: Chlorine
Electronic configuration: 2,8,7 Gains 1 electron → 2,8,8
\( \mathrm{Cl} + e^- \rightarrow \mathrm{Cl^-} \)
Chlorine forms a \( \mathrm{Cl^-} \) ion.
Why Ions Form
Atoms form ions to achieve a full outer electron shell.
This gives a stable electronic configuration (like noble gases).
| Type of Element | What Happens to Electrons | Type of Ion Formed |
|---|---|---|
| Metals | Lose electrons | Positive ions (cations) |
| Non-metals | Gain electrons | Negative ions (anions) |
Key GCSE Point
The charge on an ion equals the number of electrons lost or gained.
Example 1 (Conceptual):
Why does magnesium form a 2+ ion?
▶️ Answer/Explanation
Magnesium has electronic configuration 2,8,2.
It loses 2 electrons to achieve 2,8.
Therefore, it forms \( \mathrm{Mg^{2+}} \).
Example 2 (Numerical):
An atom has electronic configuration 2,8,6. What ion will it form?
▶️ Answer/Explanation
It needs 2 more electrons to achieve a full shell (2,8,8).
It will gain 2 electrons.
It forms a 2− ion.
Example 3 (Hard):
Explain fully why metals tend to form positive ions while non-metals form negative ions.
▶️ Answer/Explanation
Metals have few electrons in their outer shell.
It is easier for them to lose electrons than gain many.
Losing electrons gives them a full outer shell.
Non-metals have nearly full outer shells.
It is easier for them to gain electrons.
Therefore, metals form positive ions and non-metals form negative ions.
1.38 Charges of Common Ions
You must know the charges of common ions for GCSE exams.
Ion charges are determined by how many electrons are lost or gained.
1. Metals in Groups 1, 2 and 3
Metals lose electrons to form positive ions.
| Group | Electrons Lost | Ion Charge |
|---|---|---|
| 1 | Lose 1 | +1 |
| 2 | Lose 2 | +2 |
| 3 | Lose 3 | +3 |
2. Non-metals in Groups 5, 6 and 7
Non-metals gain electrons to form negative ions.
| Group | Electrons Gained | Ion Charge |
|---|---|---|
| 5 | Gain 3 | −3 |
| 6 | Gain 2 | −2 |
| 7 | Gain 1 | −1 |
3. Transition Metal and Other Common Positive Ions
| Ion | Charge |
|---|---|
| Ag+ | +1 |
| Cu2+ | +2 |
| Fe2+ | +2 |
| Fe3+ | +3 |
| Pb2+ | +2 |
| Zn2+ | +2 |
4. Common Polyatomic Ions
| Ion | Formula | Charge |
|---|---|---|
| Hydrogen | H+ | +1 |
| Hydroxide | OH− | −1 |
| Ammonium | NH4+ | +1 |
| Carbonate | CO32− | −2 |
| Nitrate | NO3− | −1 |
| Sulfate | SO42− | −2 |
Key GCSE Memory Tip
Group number of metals = ion charge Group number from 8 (for non-metals) = ion charge
Example 1 (Conceptual):
Why does oxygen form O2−?
▶️ Answer/Explanation
Oxygen is in Group 6.
It gains 2 electrons to complete its outer shell.
Therefore, it forms O2−.
Example 2 (Numerical):
What ion will aluminium form?
▶️ Answer/Explanation
Aluminium is in Group 3.
It loses 3 electrons.
It forms Al3+.
Example 3 (Hard ):
Write the formula of iron(III) sulfate.
▶️ Answer/Explanation
Fe3+ and SO42−
Balance charges:
2 × (+3) = +6 3 × (−2) = −6
Formula = Fe2(SO4)3
