Edexcel iGCSE Chemistry -1.39 Writing Ionic Formulae- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.39 Writing Ionic Formulae- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.39 Writing Ionic Formulae- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.39 write formulae for compounds formed between the ions listed above
1.39 Writing Formulae for Ionic Compounds
Ionic compounds are formed between positive ions (cations) and negative ions (anions).
The total positive charge must equal the total negative charge.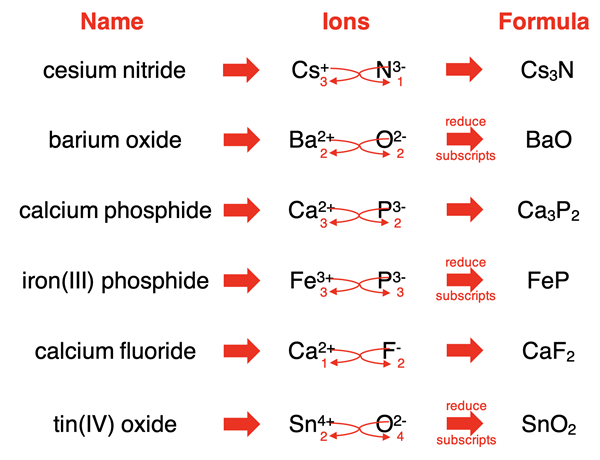
Step-by-Step Method
1. Write the ions with their charges.
2. Balance the total charges.
3. Use the smallest whole number ratio.
4. Use brackets for polyatomic ions if more than one is needed.
Balancing Charges (Criss-Cross Method)
The number of each ion is chosen so that:
Total positive charge = Total negative charge
| Positive Ion | Negative Ion | Formula |
|---|---|---|
| Na+ | Cl− | NaCl |
| Mg2+ | Cl− | MgCl2 |
| Al3+ | O2− | Al2O3 |
Using Polyatomic Ions
Brackets are used if more than one polyatomic ion is needed.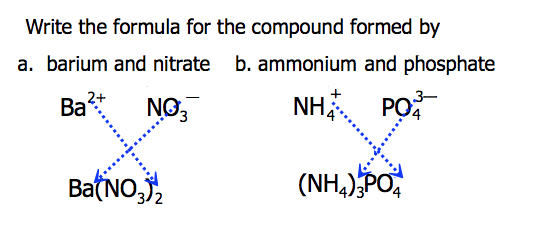
Examples:
Ca2+ + NO3− → Ca(NO3)2
Fe3+ + SO42− → Fe2(SO4)3
NH4+ + CO32− → (NH4)2CO3
Common Mistake to Avoid
Do not change the small numbers inside a polyatomic ion.
For example: NO3− stays NO3, not NO6.
Example 1 (Conceptual):
Why is magnesium oxide written as MgO and not Mg2O?
▶️ Answer/Explanation
Mg forms Mg2+ and oxygen forms O2−.
The charges already balance 1:1.
So the simplest ratio is MgO.
Example 2 (Numerical):
Write the formula of aluminium nitrate.
▶️ Answer/Explanation
Al3+ and NO3−
Need three nitrate ions to balance +3.
Formula = Al(NO3)3
Example 3 (Hard ):
Write the formula of iron(II) hydroxide and iron(III) hydroxide.
▶️ Answer/Explanation
Iron(II) hydroxide:
Fe2+ and OH−
Need 2 hydroxide ions.
Formula = Fe(OH)2
Iron(III) hydroxide:
Fe3+ and OH−
Need 3 hydroxide ions.
Formula = Fe(OH)3
