Edexcel iGCSE Chemistry -1.4–1.5C Solutions and Solubility- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.4–1.5C Solutions and Solubility- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.4–1.5C Solutions and Solubility- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.4 know what is meant by the terms:
• solvent
• solute
• solution
• saturated solution
1.5C know what is meant by the term solubility in the units g per 100 g of solvent
1.4 Key Terms: Solvent, Solute, Solution and Saturated Solution
When substances mix to form a solution, specific terms are used to describe the roles of the substances involved.
Solute
A solute is the substance that is dissolved in a liquid. 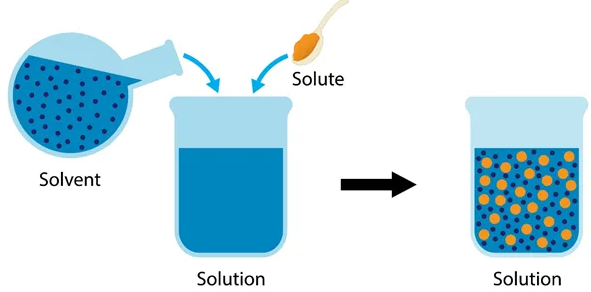
It is usually present in a smaller amount.
Example: In salt water, the salt is the solute.
Solvent
A solvent is the substance that dissolves the solute.
It is usually present in a larger amount.
Example: In salt water, the water is the solvent.
Solution
A solution is the clear mixture formed when a solute dissolves in a solvent.
The particles of solute are evenly spread throughout the solvent.
Solution = Solute + Solvent
Saturated Solution
A saturated solution is a solution that contains the maximum amount of solute that can dissolve at a given temperature.
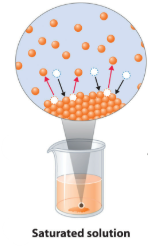
If more solute is added, it will not dissolve and will remain as a solid.
The amount of solute that can dissolve usually increases as temperature increases.
Comparison of the Terms
| Term | Definition | Example (Salt Water) |
|---|---|---|
| Solute | Substance that is dissolved | Salt |
| Solvent | Substance that does the dissolving | Water |
| Solution | Uniform mixture of solute and solvent | Salt water |
| Saturated Solution | Maximum solute dissolved at a given temperature | Salt water where extra salt will not dissolve |
Example 1 (Conceptual):
A student adds sugar to tea and stirs. Identify the solute, solvent and solution.
▶️ Answer/Explanation
Solute: Sugar (it dissolves).
Solvent: Tea (main liquid that dissolves the sugar).
Solution: Sweetened tea.
Example 2 (Numerical):
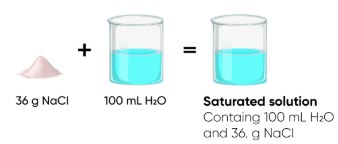
At \( 20^\circ \mathrm{C} \), the maximum amount of salt that dissolves in \( 100 \, \mathrm{cm^3} \) of water is \( 36 \, \mathrm{g} \).
If \( 40 \, \mathrm{g} \) of salt is added, how much remains undissolved?
▶️ Answer/Explanation
Maximum dissolved = \( 36 \, \mathrm{g} \)
Added = \( 40 \, \mathrm{g} \)
Undissolved = \( 40 – 36 = 4 \, \mathrm{g} \)
The solution is saturated, and \( 4 \, \mathrm{g} \) remains as solid.
Example 3 (Hard):
A student heats a saturated solution and observes that more solute dissolves. Explain why using particle theory.
▶️ Answer/Explanation
Heating increases the kinetic energy of solvent particles.
The solvent particles move faster and are better able to overcome the attractive forces holding the solute particles together.
As a result, more solute particles can separate and dissolve, increasing solubility.
1.5C Solubility (in g per 100 g of Solvent)
Solubility is a measure of how much solute can dissolve in a given amount of solvent at a particular temperature.
Definition of Solubility
Solubility is the maximum mass of solute that can dissolve in 100 g of solvent at a stated temperature.
Solubility unit = \( \mathrm{g\ per\ 100\ g\ of\ solvent} \)
This value describes a saturated solution.
Important Points
• Solubility depends on temperature.
• The solvent mass is fixed at \( 100 \, \mathrm{g} \) when using this unit.
• If more solute is added than the solubility value, it will not dissolve.
Example Meaning
If a substance has a solubility of \( 36 \, \mathrm{g\ per\ 100\ g\ water} \) at \( 20^\circ \mathrm{C} \), it means:
A maximum of \( 36 \, \mathrm{g} \) of the solute can dissolve in \( 100 \, \mathrm{g} \) of water at \( 20^\circ \mathrm{C} \).
Solubility vs Concentration
| Feature | Solubility | Concentration |
|---|---|---|
| Definition | Maximum mass that can dissolve | Amount dissolved in a given volume |
| Unit | \( \mathrm{g/100g\ solvent} \) | \( \mathrm{g/dm^3} \) or \( \mathrm{g/cm^3} \) |
| Temperature Dependent? | Yes | Not necessarily maximum |
Example 1 (Conceptual):
A salt has a solubility of \( 50 \, \mathrm{g/100g\ water} \) at \( 30^\circ \mathrm{C} \). What does this mean?
▶️ Answer/Explanation
It means a maximum of \( 50 \, \mathrm{g} \) of salt can dissolve in \( 100 \, \mathrm{g} \) of water at \( 30^\circ \mathrm{C} \).
Any extra salt added will remain undissolved.
Example 2 (Numerical):
The solubility of a substance is \( 25 \, \mathrm{g/100g\ water} \) at \( 40^\circ \mathrm{C} \).
Calculate the maximum mass that can dissolve in \( 250 \, \mathrm{g} \) of water.
▶️ Answer/Explanation
If \( 100 \, \mathrm{g} \) water dissolves \( 25 \, \mathrm{g} \)
Then \( 250 \, \mathrm{g} \) water dissolves:
\( \dfrac{25}{100} \times 250 = 62.5 \, \mathrm{g} \)
So the maximum mass that dissolves is \( 62.5 \, \mathrm{g} \).
Example 3 (Hard):
At \( 20^\circ \mathrm{C} \), the solubility of a salt is \( 30 \, \mathrm{g/100g\ water} \). A student adds \( 45 \, \mathrm{g} \) of salt to \( 100 \, \mathrm{g} \) of water.
Describe what happens and calculate how much remains undissolved.
▶️ Answer/Explanation
Only \( 30 \, \mathrm{g} \) can dissolve because this is the solubility at \( 20^\circ \mathrm{C} \).
Undissolved mass = \( 45 – 30 = 15 \, \mathrm{g} \)
The solution becomes saturated and \( 15 \, \mathrm{g} \) remains as solid at the bottom.
