Edexcel iGCSE Chemistry -1.40 Dot-and-Cross Diagrams for Ionic Bonding- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.40 Dot-and-Cross Diagrams for Ionic Bonding- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.40 Dot-and-Cross Diagrams for Ionic Bonding- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.40 draw dot-and-cross diagrams to show the formation of ionic compounds by electron transfer (only outer electrons need be shown)
1.40 Dot-and-Cross Diagrams for Ionic Compounds
Dot-and-cross diagrams are used to show ionic bonding through electron transfer.
They show how a metal atom transfers electrons to a non-metal atom to form oppositely charged ions.
What Must Be Shown in the Diagram
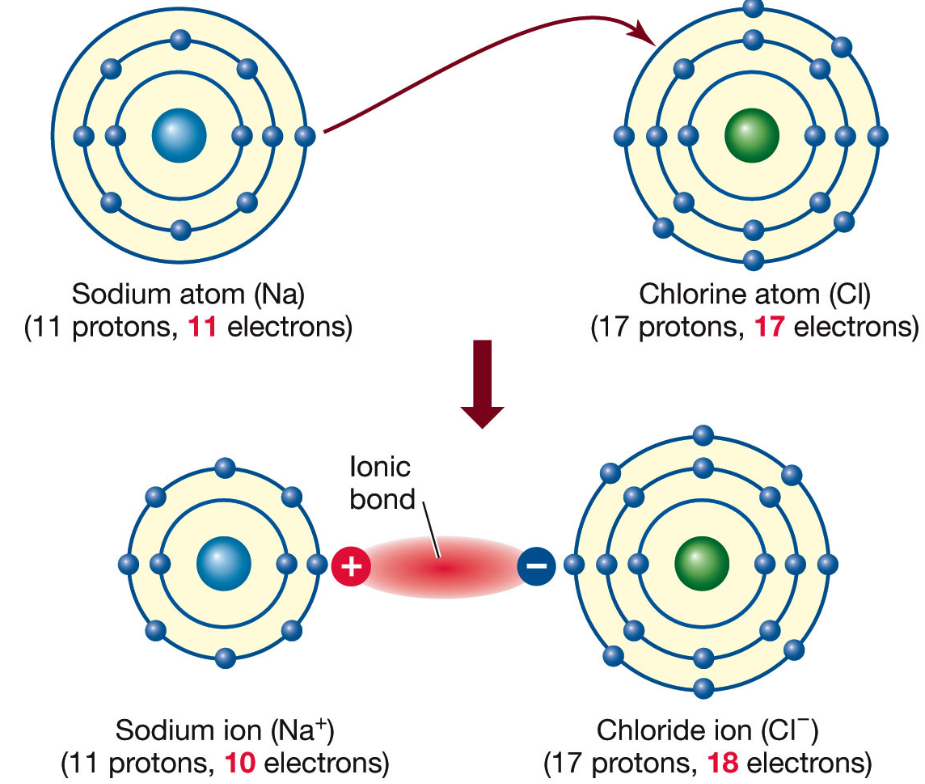
- Only outer (valence) electrons
- Dots for one atom and crosses for the other
- Electron transfer from metal to non-metal
- Square brackets around each ion
- The correct charge on each ion
- Full outer shells after transfer
Inner shells are not drawn at GCSE level.
Why Electron Transfer Occurs
Atoms transfer electrons to achieve a full outer shell.
Metals lose electrons because they have few outer electrons.
Non-metals gain electrons because their outer shell is nearly full.
Result: Stable electronic configuration (like noble gases)
Group Trends (Exam Essential)
The number of electrons transferred depends on the group number.
- Group 1 → Lose 1 electron → +1
- Group 2 → Lose 2 electrons → +2
- Group 3 → Lose 3 electrons → +3
- Group 5 → Gain 3 electrons → −3
- Group 6 → Gain 2 electrons → −2
- Group 7 → Gain 1 electron → −1
Balancing Charges in Ionic Compounds
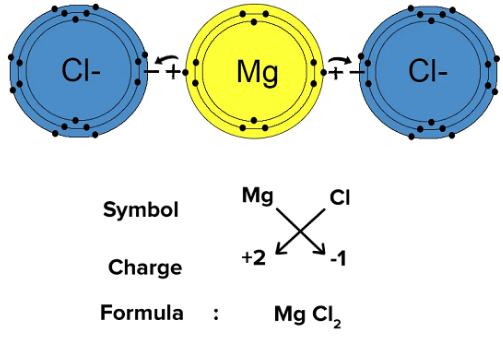
The total positive charge must equal the total negative charge.
For example:
Mg2+ and Cl− Need two Cl− to balance +2
Formula = MgCl2
Step-by-Step Method for Any Question
- Write electronic configurations.
- Identify how many electrons are lost/gained.
- Draw outer electrons only.
- Show electron transfer.
- Draw ions in brackets with charges.
- Check both ions have full outer shells.
Common Exam Mistakes
- Forgetting brackets around ions
- Not showing charges
- Incorrect number of transferred electrons
- Forgetting to balance ion ratio
- Drawing inner shells
Example 1 (Conceptual)
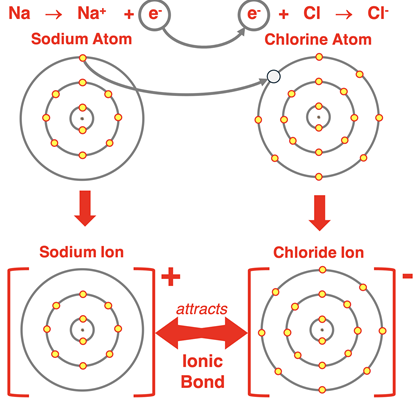
Explain how a dot-and-cross diagram shows the formation of sodium chloride.
▶️ Answer / Explanation
Sodium has 1 outer electron.
Chlorine has 7 outer electrons.
Sodium transfers 1 electron to chlorine.
[Na]+ [Cl]−
Chlorine shows 8 electrons (7 dots + 1 cross).
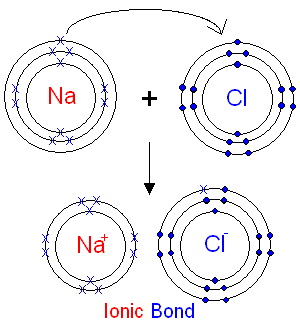
Both ions now have full outer shells.
Example 2 (Numerical)
Draw a dot-and-cross diagram for magnesium chloride.
▶️ Answer / Explanation
Magnesium loses 2 electrons.
Each chlorine gains 1 electron.
[Mg]2+ 2[Cl]−
Each chloride ion shows 7 original dots + 1 cross.
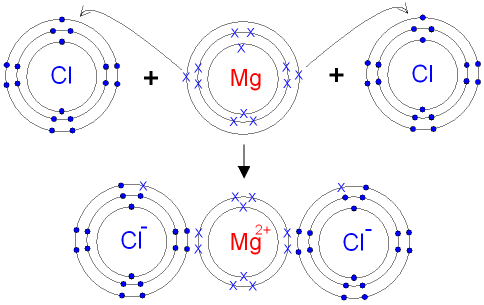
Magnesium shows no outer electrons.
Important: There must be TWO chloride ions.
Example 3 (Hard )
Draw and explain the dot-and-cross diagram for aluminium oxide.
▶️ Answer / Explanation
Aluminium loses 3 electrons.
Oxygen gains 2 electrons.
To balance charges:
2Al → 2Al3+ 3O → 3O2−
Formula = Al2O3
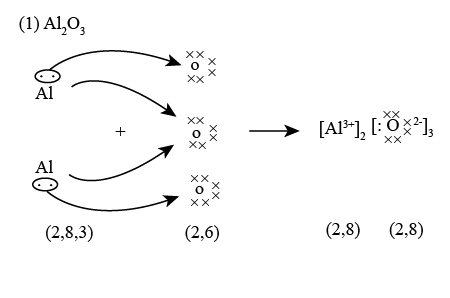
Each oxygen shows 6 original dots + 2 crosses.
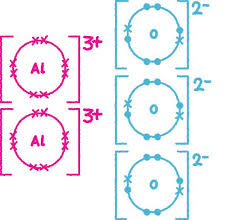
Each aluminium loses 3 electrons and is shown as Al3+.
Key Point: Charges must balance (+6 and −6).
