Edexcel iGCSE Chemistry -1.41 Ionic Bonding and Electrostatic Attraction- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.41 Ionic Bonding and Electrostatic Attraction- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.41 Ionic Bonding and Electrostatic Attraction- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.41 understand ionic bonding in terms of electrostatic attractions
1.41 Ionic Bonding in Terms of Electrostatic Attractions
Ionic bonding occurs between positive and negative ions.
It is defined as the electrostatic attraction between oppositely charged ions.
What Is Electrostatic Attraction?
Electrostatic attraction is the force between opposite charges.
Positive ions attract negative ions.
This attraction holds ions together in a giant ionic lattice.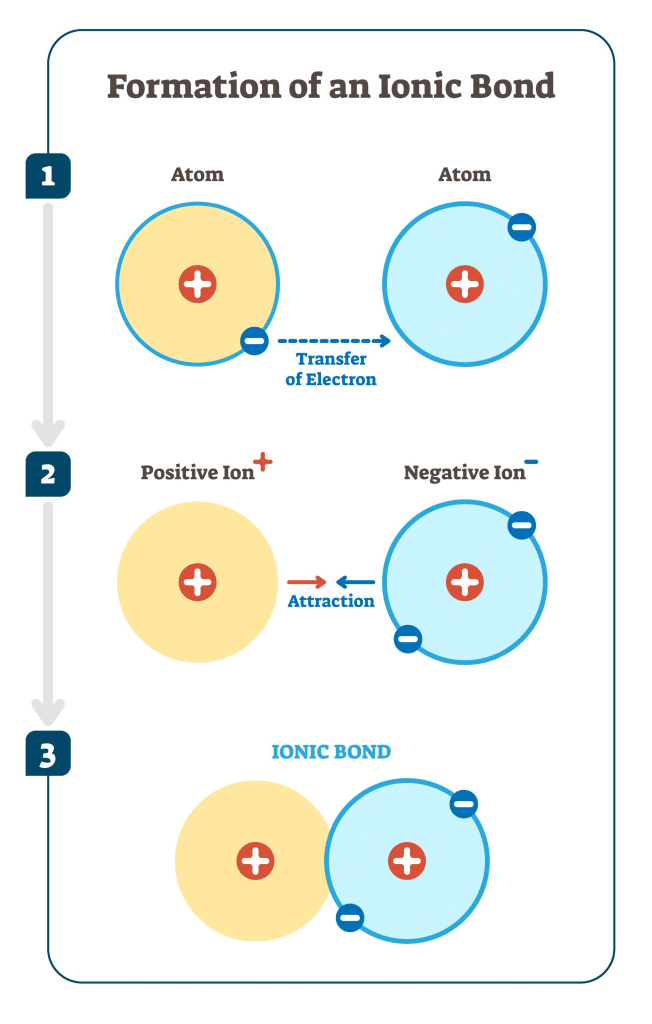
Formation of Ionic Bonds
Step 1: Electron transfer occurs (metal → non-metal).
Step 2: Positive and negative ions form.
Step 3: Strong electrostatic attractions act in all directions.
These attractions form a regular 3D lattice structure.
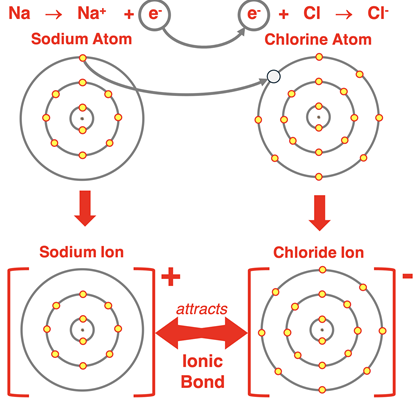
Example: Sodium Chloride
\( \mathrm{Na} \rightarrow \mathrm{Na^+} + e^- \)
\( \mathrm{Cl} + e^- \rightarrow \mathrm{Cl^-} \)
After transfer, Na+ and Cl− attract strongly.
The attraction acts between many ions, not just one pair.
Properties Explained by Ionic Bonding
| Property | Explanation |
|---|---|
| High melting point | Strong electrostatic forces require large energy to break |
| Brittle | If layers shift, like charges repel |
| Conducts when molten | Ions are free to move |
| Does not conduct when solid | Ions fixed in lattice |
Key GCSE Definition
An ionic bond is the strong electrostatic attraction between oppositely charged ions.
Example 1 (Conceptual):
Why does sodium chloride have a high melting point?
▶️ Answer/Explanation
There are strong electrostatic attractions between Na+ and Cl−.
A large amount of energy is required to overcome these forces.
Example 2 (Numerical ):
Magnesium oxide has a higher melting point than sodium chloride. Why?
▶️ Answer/Explanation
MgO contains Mg2+ and O2− ions.
The charges are higher than Na+ and Cl−.
Stronger electrostatic attraction occurs.
Therefore, more energy is needed to melt MgO.
Example 3 (Hard):
Explain fully why ionic compounds conduct electricity when molten but not when solid.
▶️ Answer/Explanation
In a solid ionic compound, ions are held in fixed positions in a lattice.
They cannot move, so charge cannot flow.
When molten, the lattice breaks down.
The ions are free to move.
Moving charged particles carry electrical current.
