Edexcel iGCSE Chemistry -1.42–1.43 Giant Ionic Lattices, Melting Points, and Electrical Conductivity- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.42–1.43 Giant Ionic Lattices, Melting Points, and Electrical Conductivity- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.42–1.43 Giant Ionic Lattices, Melting Points, and Electrical Conductivity- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.42 understand why compounds with giant ionic lattices have high melting and boiling points
1.43 know that ionic compounds do not conduct electricity when solid, but do conduct electricity when molten and in aqueous solution
1.42 Why Giant Ionic Lattices Have High Melting and Boiling Points
Ionic compounds form giant ionic lattices.
A giant ionic lattice is a regular three-dimensional arrangement of alternating positive and negative ions.
Structure of a Giant Ionic Lattice
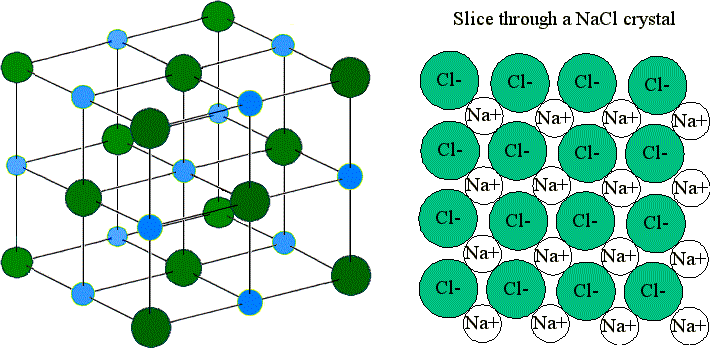
- Each positive ion is surrounded by negative ions.
- Each negative ion is surrounded by positive ions.
- Strong electrostatic attractions act in all directions throughout the structure.
Reason for High Melting and Boiling Points
There are strong electrostatic forces of attraction between oppositely charged ions.
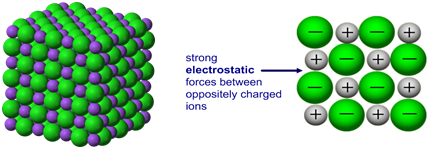
A large amount of energy is required to overcome these forces.
More energy → Higher melting point More energy → Higher boiling point
Factors Affecting Strength of Ionic Bonding
| Factor | Effect on Attraction | Effect on Melting Point |
|---|---|---|
| Higher ionic charge | Stronger attraction | Higher melting point |
| Smaller ion size | Stronger attraction (closer together) | Higher melting point |
Comparison Example
Magnesium oxide (MgO) vs sodium chloride (NaCl):
MgO contains Mg2+ and O2− NaCl contains Na+ and Cl−
Because MgO has ions with higher charges, the electrostatic attraction is stronger.
Therefore, MgO has a much higher melting point.
Key GCSE Explanation
Giant ionic lattices have high melting and boiling points because strong electrostatic forces between oppositely charged ions require large amounts of energy to overcome.
Example 1 (Conceptual):
Why does sodium chloride have a high melting point?
▶️ Answer/Explanation
There are strong electrostatic attractions between Na+ and Cl−.
A large amount of energy is needed to break these forces.
Example 2 (Comparison):
Explain why magnesium oxide has a higher melting point than sodium chloride.
▶️ Answer/Explanation
MgO has Mg2+ and O2− ions.
The charges are greater than in NaCl.
Stronger electrostatic attraction exists.
More energy is required to overcome the lattice.
Example 3 (Hard):
Explain fully why ionic compounds have high boiling points as well as high melting points.
▶️ Answer/Explanation
Ionic compounds consist of a giant lattice of oppositely charged ions.
Strong electrostatic forces act in all directions.
Melting requires overcoming many strong attractions.
Boiling requires complete separation of ions.
Both processes require large amounts of energy.
1.43 Electrical Conductivity of Ionic Compounds
Ionic compounds contain positive and negative ions.
Whether they conduct electricity depends on whether the ions are free to move.
Ionic Compounds as Solids
In the solid state, ions are held in fixed positions in a giant lattice.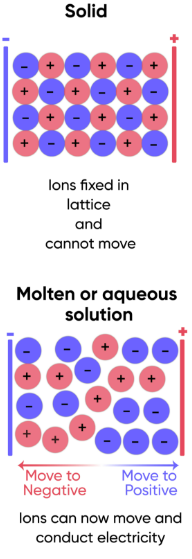
They cannot move.
No movement of charged particles → No electrical conduction
Therefore: Solid ionic compounds do not conduct electricity.
Ionic Compounds When Molten
When melted, the lattice breaks down.
The ions become free to move.
Moving charged ions → Electrical current flows
Therefore: Molten ionic compounds conduct electricity.
Ionic Compounds in Aqueous Solution
When dissolved in water, ionic compounds separate into ions.
Example: \( \mathrm{NaCl(s)} \rightarrow \mathrm{Na^+(aq)} + \mathrm{Cl^-(aq)} \)
The ions are free to move in solution.
Therefore: Ionic compounds in aqueous solution conduct electricity.
| State | Are Ions Free to Move? | Conducts Electricity? |
|---|---|---|
| Solid | No | No |
| Molten | Yes | Yes |
| Aqueous | Yes | Yes |
Key GCSE Explanation
Ionic compounds conduct electricity only when ions are free to move.
Example 1 (Conceptual):
Why does solid sodium chloride not conduct electricity?
▶️ Answer/Explanation
The ions are fixed in a lattice.
They cannot move.
Therefore, no current can flow.
Example 2 (Comparison):
Explain why molten magnesium oxide conducts electricity but solid magnesium oxide does not.
▶️ Answer/Explanation
In solid MgO, ions are fixed in a lattice.
When molten, the lattice breaks down.
The ions become free to move and carry charge.
Example 3 (Hard):
Explain fully why an aqueous solution of copper(II) sulfate conducts electricity.
▶️ Answer/Explanation
Copper(II) sulfate dissolves in water.
It separates into Cu2+ and SO42− ions.
These ions are free to move in the solution.
Moving charged particles allow current to flow.
