Edexcel iGCSE Chemistry -1.44–1.45 Covalent Bonds and Electrostatic Attraction- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.44–1.45 Covalent Bonds and Electrostatic Attraction- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.44–1.45 Covalent Bonds and Electrostatic Attraction- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.44 know that a covalent bond is formed between atoms by the sharing of a pair of electrons
1.45 understand covalent bonds in terms of electrostatic attractions
1.44 Covalent Bonding – Sharing of a Pair of Electrons
Definition:
A covalent bond is a shared pair of electrons between two atoms.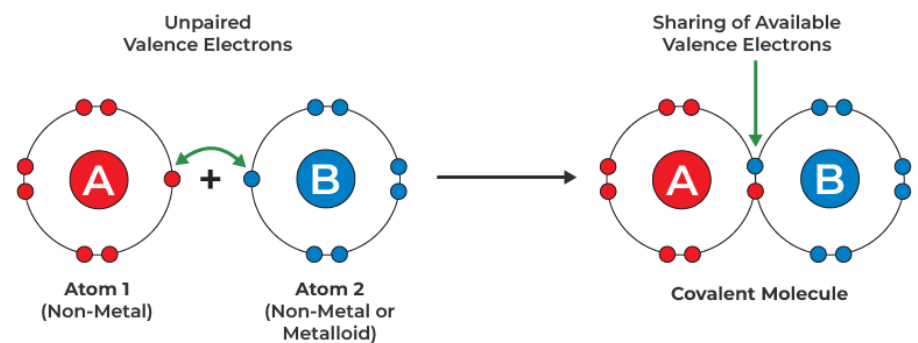
Covalent bonding usually occurs between non-metal atoms.
Why Do Atoms Form Covalent Bonds?
Atoms form covalent bonds to achieve a full outer electron shell.
Instead of transferring electrons (as in ionic bonding), atoms share electrons.
The shared electrons are attracted to both nuclei.
This electrostatic attraction between:
• The positive nuclei
• The shared pair of electrons
holds the atoms together.
Single Covalent Bond
A single covalent bond consists of one shared pair of electrons.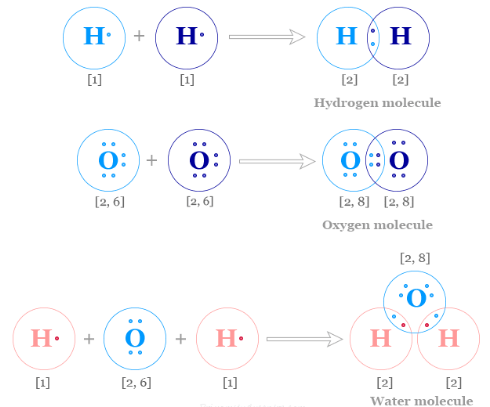
Example: Hydrogen molecule.
Each hydrogen has 1 electron. They share a pair to form:
\( \mathrm{H_2} \)
Each hydrogen now has 2 electrons in its outer shell (full for the first shell).
Example: Chlorine Molecule
Chlorine has electronic configuration 2,8,7.
Each chlorine atom shares one electron.
\( \mathrm{Cl_2} \)
Both chlorine atoms achieve a full outer shell (8 electrons).
Multiple Covalent Bonds
If more than one pair of electrons is shared:
| Bond Type | Shared Pairs | Example | Lewis Diagram |
|---|---|---|---|
| Single bond | 1 pair | \( \mathrm{H_2} \) | 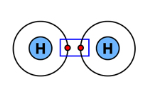 |
| Double bond | 2 pairs | \( \mathrm{O_2} \) | 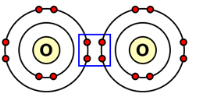 |
| Triple bond | 3 pairs | \( \mathrm{N_2} \) | 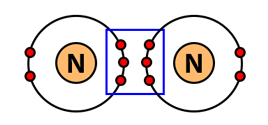 |
Key GCSE Definition to Memorise
A covalent bond is the strong electrostatic attraction between a shared pair of electrons and the nuclei of the bonded atoms.
Example 1 (Conceptual):
Why do two hydrogen atoms form a covalent bond?
▶️ Answer/Explanation
Each hydrogen atom has 1 electron.
They share their electrons to form one shared pair.
Both achieve a full outer shell of 2 electrons.
Example 2 (Deduction):
How many covalent bonds does oxygen form in \( \mathrm{O_2} \)?
▶️ Answer/Explanation
Oxygen has 6 outer electrons.
It needs 2 more to complete its shell.
Two pairs are shared.
Therefore, a double covalent bond is formed.
Example 3 (Hard):
Explain fully why covalent bonds are strong even though electrons are shared rather than transferred.
▶️ Answer/Explanation
The shared pair of electrons is attracted to both positively charged nuclei.
This creates strong electrostatic attraction.
The electrons hold the atoms together.
A large amount of energy is required to break this shared attraction.
1.45 Covalent Bonds in Terms of Electrostatic Attractions
Definition :
A covalent bond is the strong electrostatic attraction between a shared pair of electrons and the positively charged nuclei of the bonded atoms.
This definition is more detailed than simply saying “sharing of electrons”.
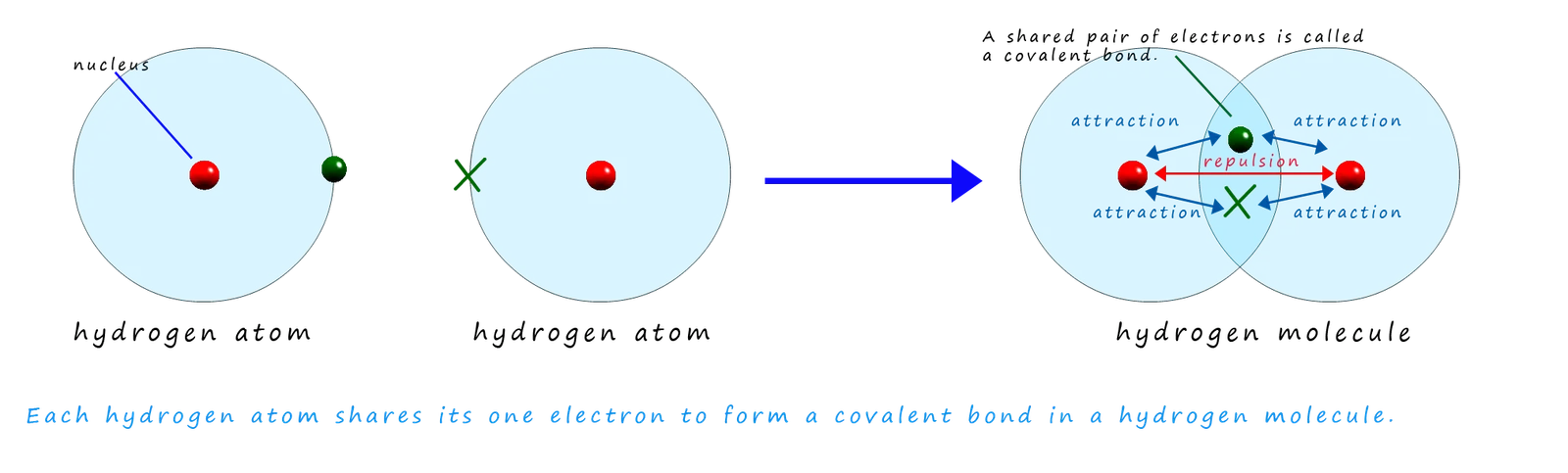
What Actually Causes the Attraction?
Atoms contain:
• A positively charged nucleus (due to protons)
• Negatively charged electrons
When a pair of electrons is shared between two atoms:
• Both nuclei attract the shared electrons
• The shared electrons hold the nuclei together
This electrostatic attraction forms the covalent bond.
Example: Hydrogen Molecule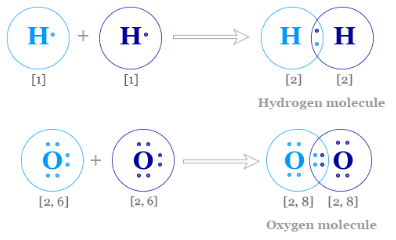
Each hydrogen atom has 1 electron.
They share one pair of electrons to form:
\( \mathrm{H_2} \)
The shared electrons are attracted to both hydrogen nuclei.
This attraction holds the atoms together.
Example: Oxygen Molecule
Oxygen has 6 outer electrons.
Two pairs are shared between the atoms:
\( \mathrm{O_2} \)
Each shared pair is attracted to both nuclei.
This creates strong bonding between the atoms.
Important Conceptual Point
The bond is not simply the electrons themselves.
The bond is the electrostatic force between:
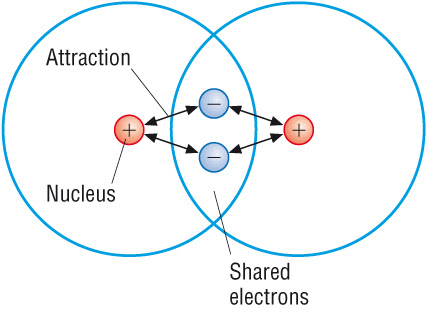
Shared electrons (negative) and Both positive nuclei
| Bond Type | What Is Attracting? | Type of Force |
|---|---|---|
| Ionic | Positive ion & negative ion | Electrostatic attraction |
| Covalent | Nuclei & shared electrons | Electrostatic attraction |
Key GCSE Comparison
Ionic bonding = attraction between fully charged ions Covalent bonding = attraction between nuclei and shared electrons
Example 1 (Conceptual):
What is actually attracting in a covalent bond?
▶️ Answer/Explanation
The positively charged nuclei attract the shared pair of electrons.
This electrostatic force forms the bond.
Example 2 (Deduction):
Why is a double bond stronger than a single bond?
▶️ Answer/Explanation
A double bond has two shared pairs of electrons.
There is greater electrostatic attraction between nuclei and electrons.
Therefore, more energy is required to break it.
Example 3 (Hard):
Explain fully why a covalent bond is described as electrostatic attraction even though no ions are formed.
▶️ Answer/Explanation
Electrostatic forces occur between positive and negative charges.
In covalent bonding, nuclei are positively charged.
Shared electrons are negatively charged.
The attraction between these opposite charges forms the bond.
No full ions are needed for electrostatic attraction to occur.
