Edexcel iGCSE Chemistry -1.46 Dot-and-Cross Diagrams for Covalent Molecules- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.46 Dot-and-Cross Diagrams for Covalent Molecules- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.46 Dot-and-Cross Diagrams for Covalent Molecules- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.46 understand how to use dot-and-cross diagrams to represent covalent bonds in:
• diatomic molecules
• inorganic molecules
• organic molecules containing up to two carbon atoms
1.46 Dot-and-Cross Diagrams for Covalent Molecules
Dot-and-cross diagrams show how outer electrons are shared in covalent bonds.![]()
Only outer shell (valence) electrons are shown.
One atom’s electrons are shown as dots (•), the other’s as crosses (×).
General Rules
1. Show only outer electrons.
2. Show each shared pair clearly.
3. Each shared pair = one covalent bond.
4. Each atom must have a full outer shell (H needs 2, others usually 8).
5. Lone pairs must be shown.
A. Diatomic Molecules
1. Hydrogen
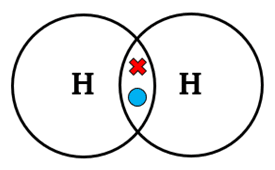
\( \mathrm{H_2} \)
Each H contributes 1 electron → 1 shared pair (single bond).
2. Oxygen
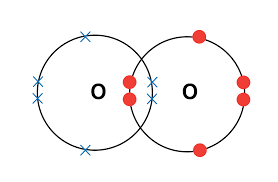
\( \mathrm{O_2} \)
Each O has 6 outer electrons → shares 2 pairs → double bond.
3. Nitrogen
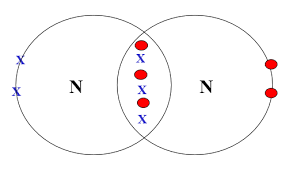
\( \mathrm{N_2} \)
Each N has 5 outer electrons → shares 3 pairs → triple bond.
4. Halogens (e.g. \( \mathrm{Cl_2} \))
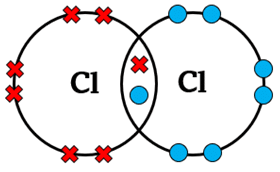
Halogens have 7 outer electrons → share 1 pair.
5. Hydrogen Halides (e.g. \( \mathrm{HCl} \))
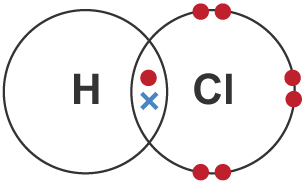
H shares 1 electron with halogen → single bond.
B. Inorganic Molecules
1. Water
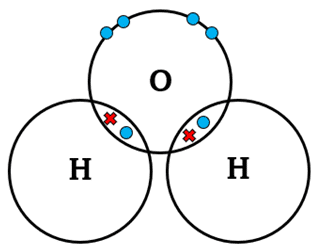
\( \mathrm{H_2O} \)
Oxygen (6 outer electrons) forms two single bonds with two hydrogen atoms.
Oxygen has two lone pairs remaining.
2. Ammonia
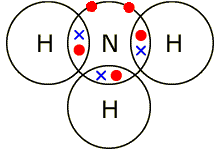
\( \mathrm{NH_3} \)
Nitrogen (5 outer electrons) forms three single bonds with hydrogen.
One lone pair remains on nitrogen.
3. Carbon Dioxide
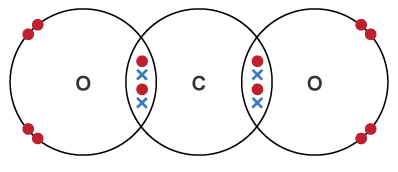
\( \mathrm{CO_2} \)
Carbon forms two double bonds (one to each oxygen).
Each oxygen keeps two lone pairs.
C. Organic Molecules (Up to Two Carbons)
1. Methane
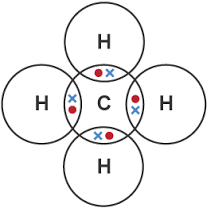
\( \mathrm{CH_4} \)
Carbon forms four single bonds with hydrogen.
2. Ethane
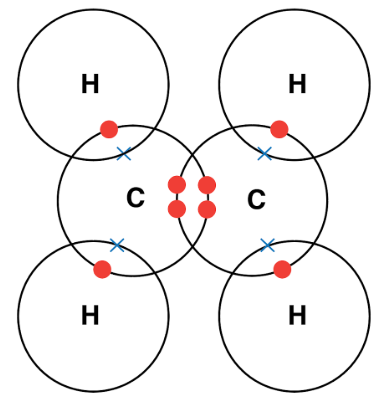
\( \mathrm{C_2H_6} \)
Single bond between carbons + each carbon bonded to three hydrogens.
3. Ethene
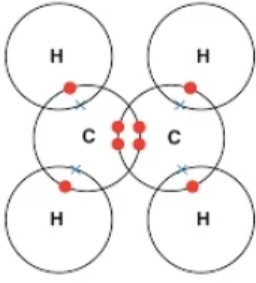
\( \mathrm{C_2H_4} \)
Double bond between carbons + each carbon bonded to two hydrogens.
4. Haloalkanes (e.g. \( \mathrm{CH_3Cl} \))
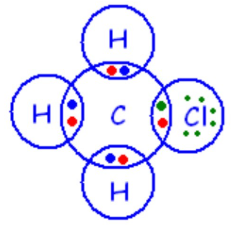
One hydrogen replaced by halogen.
| Molecule | Bond Type | Lone Pairs Present? |
|---|---|---|
| \( \mathrm{H_2} \) | Single | No |
| \( \mathrm{O_2} \) | Double | Yes |
| \( \mathrm{N_2} \) | Triple | Yes |
| \( \mathrm{CO_2} \) | Double bonds | Yes |
Example 1 (Conceptual):
Why does nitrogen form a triple bond in \( \mathrm{N_2} \)?
▶️ Answer/Explanation
Nitrogen has 5 outer electrons.
It needs 3 more to complete its shell.
Three shared pairs form a triple bond.
Example 2 (Deduction):
How many lone pairs are present on oxygen in water?
▶️ Answer/Explanation
Oxygen has 6 outer electrons.
2 pairs are used for bonding.
2 lone pairs remain.
Example 3 (Hard):
Explain how the bonding differs between ethane and ethene.
▶️ Answer/Explanation
Ethane contains a single bond between carbon atoms.
Ethene contains a double bond between carbon atoms.
A double bond means two shared pairs instead of one.
This makes ethene more reactive.
