Edexcel iGCSE Chemistry -1.47 Simple Molecular Substances- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.47 Simple Molecular Substances- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.47 Simple Molecular Substances- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.47 explain why substances with simple molecular structures are gases or liquids, or solids with low melting and boiling points
1.47 Simple Molecular Substances and Intermolecular Forces
Definition:
A simple molecular substance consists of small molecules held together by weak intermolecular forces.
![]()
Examples include:
\( \mathrm{O_2} \), \( \mathrm{N_2} \), \( \mathrm{CO_2} \), \( \mathrm{H_2O} \), \( \mathrm{CH_4} \)
Bonding Within vs Between Molecules
There are two types of forces present:
• Strong covalent bonds within each molecule
• Weak intermolecular forces between molecules
Important: Melting or boiling does NOT break covalent bonds.
It only overcomes intermolecular forces of attraction.
Why They Have Low Melting and Boiling Points
Intermolecular forces are weak compared with covalent bonds.
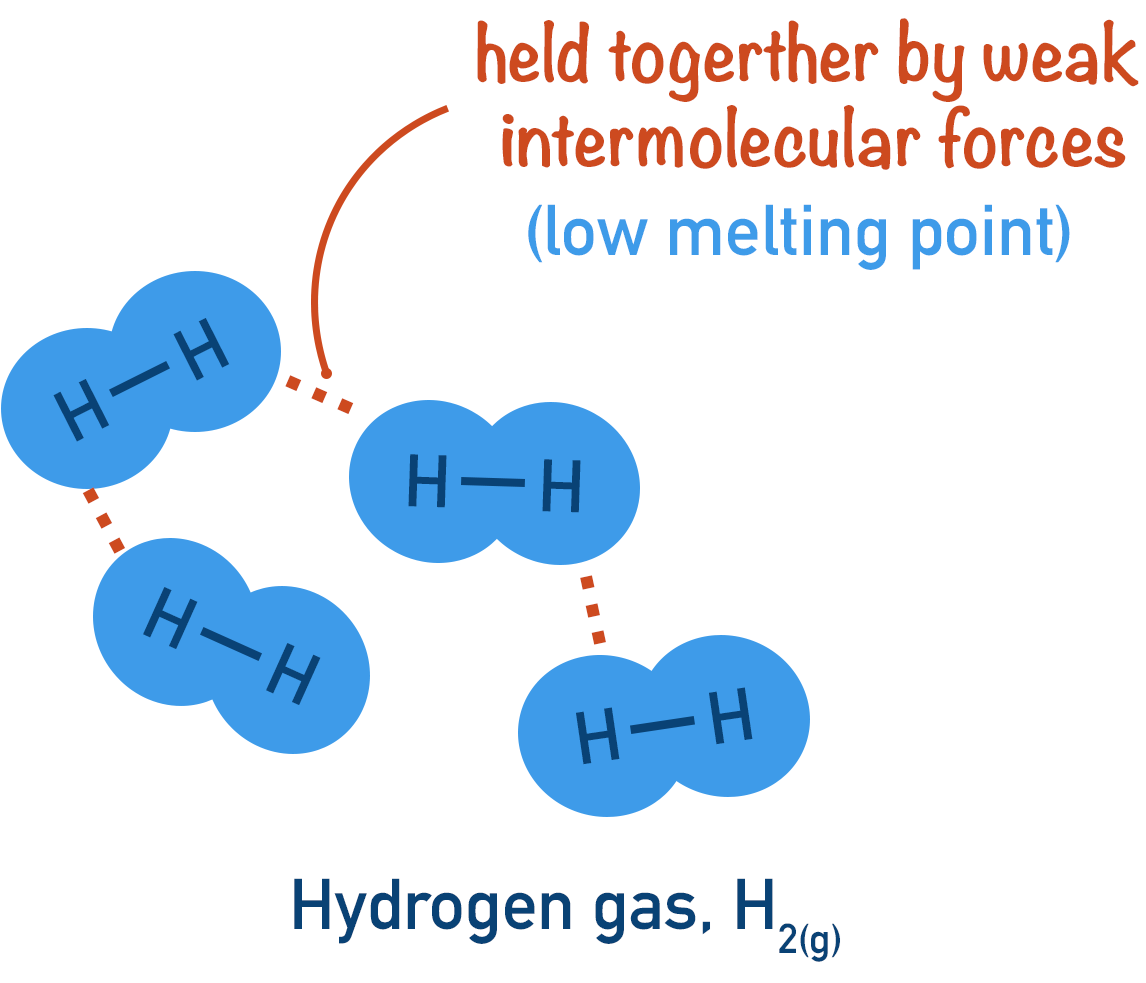
Only a small amount of energy is needed to overcome them.
Weak forces → Low energy needed → Low melting/boiling point
Therefore, simple molecular substances are often gases or liquids at room temperature.
State at Room Temperature
| Substance | State at rtp | Reason |
|---|---|---|
| \( \mathrm{O_2} \) | Gas | Very weak intermolecular forces |
| \( \mathrm{H_2O} \) | Liquid | Stronger intermolecular forces than O₂ |
| \( \mathrm{I_2} \) | Solid | Stronger intermolecular forces due to larger molecule |
Key GCSE Explanation Structure
• Simple molecular structure
• Strong covalent bonds within molecules
• Weak intermolecular forces between molecules
• Little energy required to overcome these forces
• Therefore low melting and boiling points
Example 1 (Conceptual):
Why does oxygen have a low boiling point?
▶️ Answer/Explanation
Oxygen has a simple molecular structure.
There are weak intermolecular forces between molecules.
Only a small amount of energy is needed to overcome them.
Example 2 (Comparison):
Explain why water has a higher boiling point than methane.
▶️ Answer/Explanation
Both are simple molecular substances.
Water has stronger intermolecular forces.
More energy is required to overcome these forces.
Therefore, water has a higher boiling point.
Example 3 (Hard):
Explain fully why carbon dioxide is a gas at room temperature despite containing strong covalent bonds.
▶️ Answer/Explanation
Carbon dioxide has strong covalent bonds within each molecule.
However, molecules are held together by weak intermolecular forces.
Boiling only requires overcoming intermolecular forces.
These forces are weak and require little energy.
Therefore, CO₂ has a low boiling point and is a gas at room temperature.
