Edexcel iGCSE Chemistry -1.48 Intermolecular Forces and Boiling Points- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.48 Intermolecular Forces and Boiling Points- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.48 Intermolecular Forces and Boiling Points- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.48 explain why melting and boiling points increase with increasing relative molecular mass
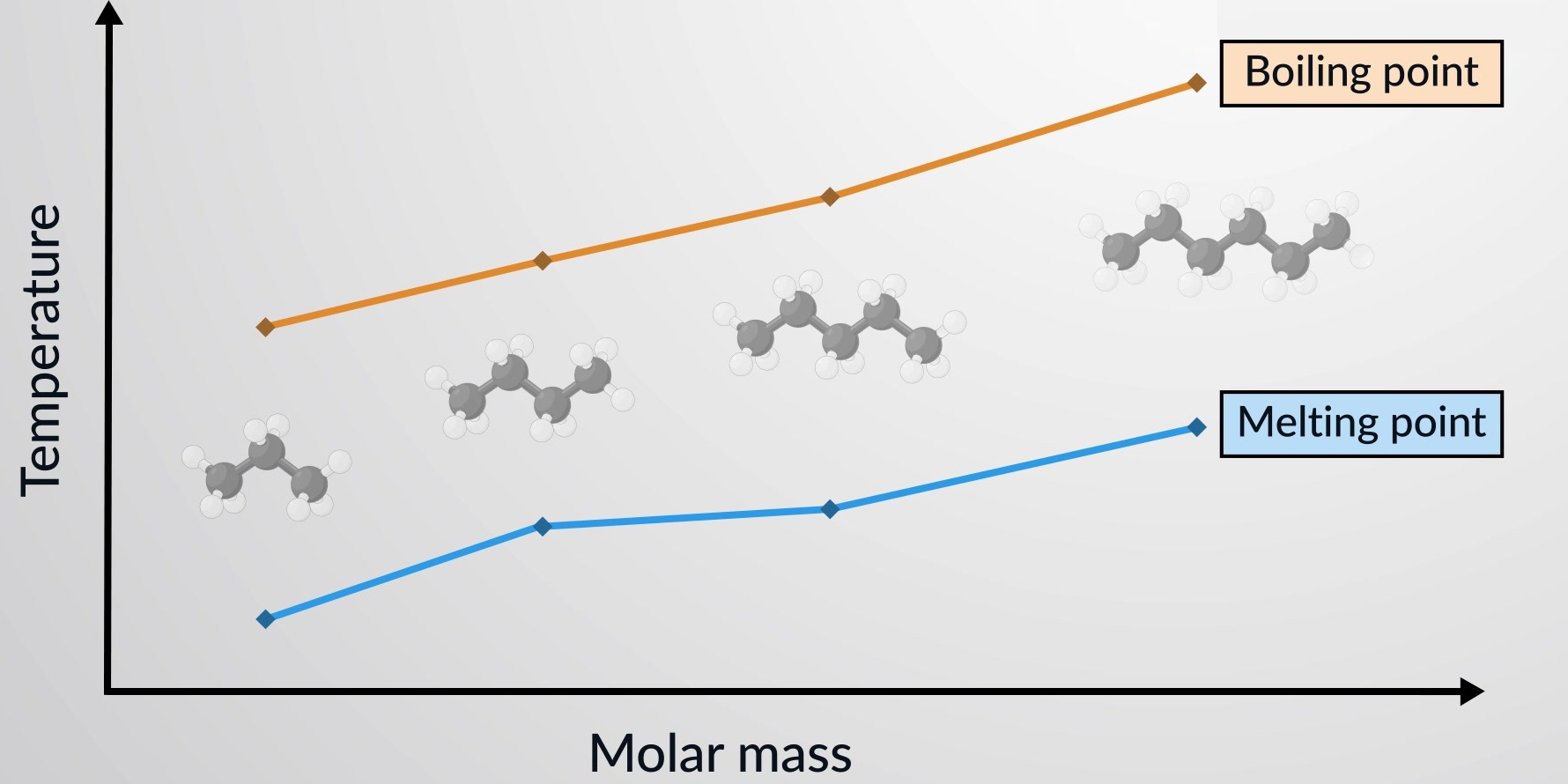
1.48 Effect of Relative Molecular Mass on Melting and Boiling Points
Key Idea:
For simple molecular substances, melting and boiling points generally increase as relative molecular mass (\( M_r \)) increases.
Why Does This Happen?
Simple molecular substances are held together by intermolecular forces of attraction.
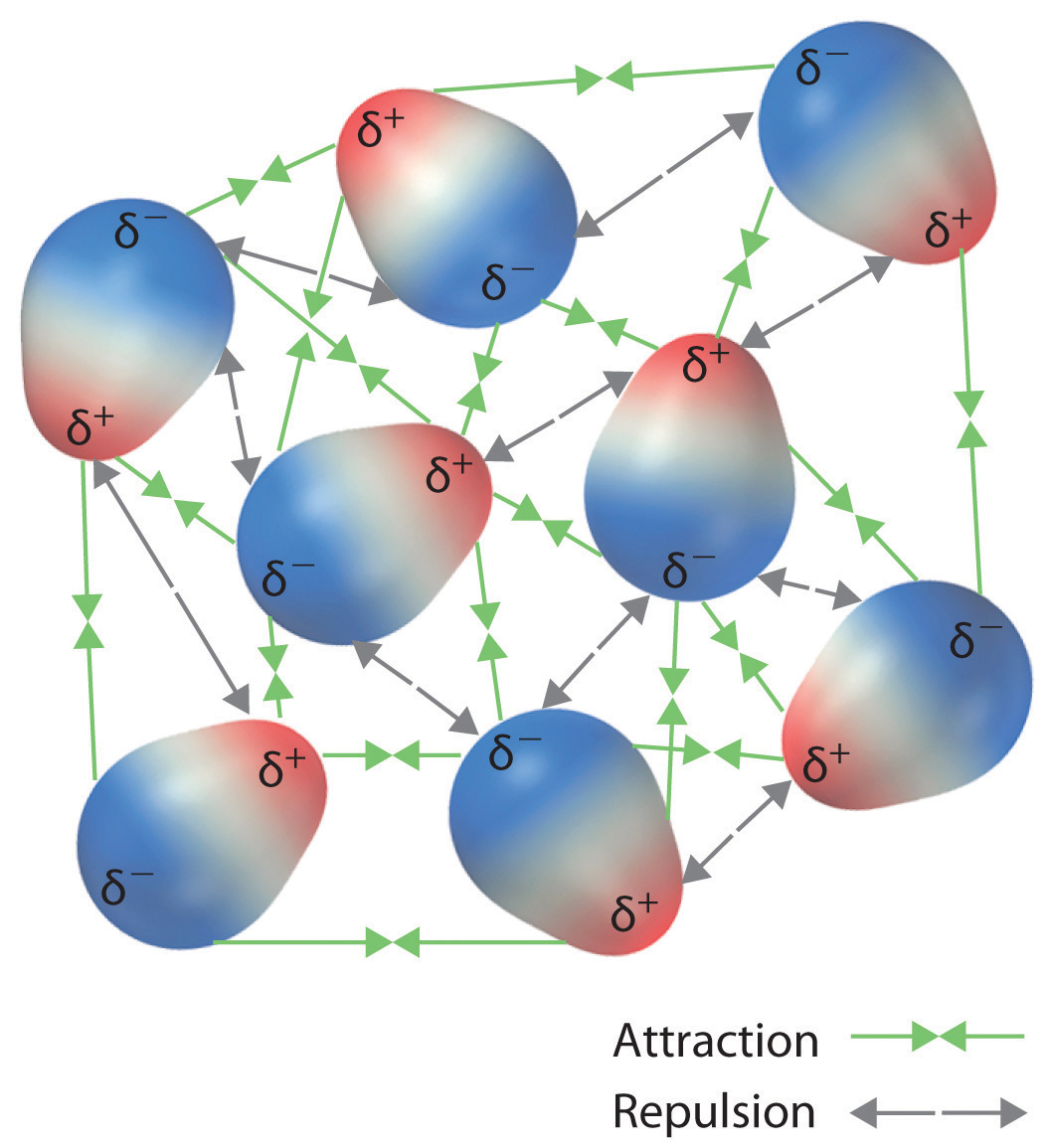
These forces become stronger as: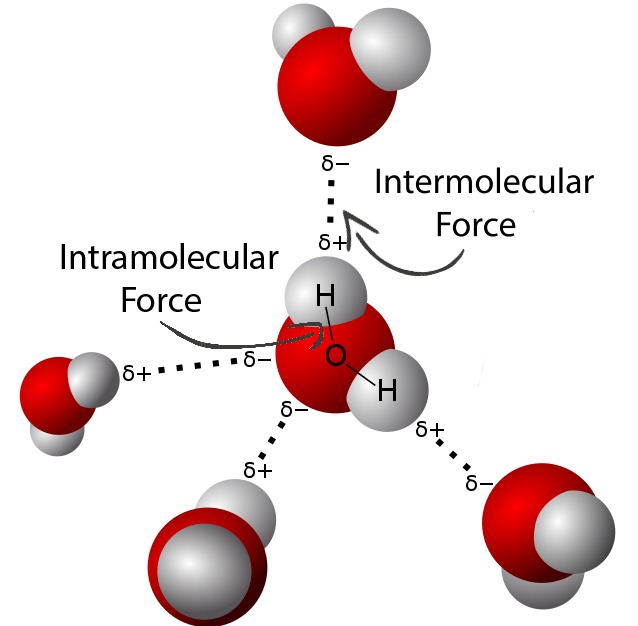
- Molecules become larger
- The number of electrons increases
- The electron cloud becomes more easily distorted
Stronger intermolecular forces mean:
More energy is required to separate molecules.
Therefore, melting and boiling points increase.
Important Clarification
The covalent bonds inside molecules do not get stronger.
It is the intermolecular forces between molecules that increase in strength.
Example: Group 7 (Halogens)
| Molecule | \( M_r \) | State at rtp |
|---|---|---|
| \( \mathrm{F_2} \) | 38 | Gas |
| \( \mathrm{Cl_2} \) | 71 | Gas |
| \( \mathrm{Br_2} \) | 160 | Liquid |
| \( \mathrm{I_2} \) | 254 | Solid |
As \( M_r \) increases down the group, melting and boiling points increase.
GCSE Explanation Structure
- Simple molecular structure
- Weak intermolecular forces
- Increasing molecular size → stronger intermolecular forces
- More energy required to overcome forces
- Therefore higher melting and boiling points
Example 1 (Conceptual):
Why does iodine have a higher boiling point than chlorine?
▶️ Answer/Explanation
Iodine has a larger relative molecular mass.
It has more electrons.
Intermolecular forces are stronger.
More energy is needed to overcome these forces.
Example 2 (Numerical):
Which has the higher boiling point: \( \mathrm{CH_4} \) (\( M_r = 16 \)) or \( \mathrm{C_2H_6} \) (\( M_r = 30 \))?
▶️ Answer/Explanation
\( \mathrm{C_2H_6} \) has a higher relative molecular mass.
It has more electrons.
Stronger intermolecular forces are present.
Therefore, ethane has the higher boiling point.
Example 3 (Hard ):
Explain fully why the melting point increases from \( \mathrm{F_2} \) to \( \mathrm{I_2} \).
▶️ Answer/Explanation
All halogens are simple molecular substances.
They are held together by intermolecular forces.
Going down the group, the molecules increase in size.
The number of electrons increases.
Intermolecular forces become stronger.
More energy is required to overcome these forces.
Therefore, melting point increases.
