Edexcel iGCSE Chemistry -1.49–1.50 Giant Covalent Structures; Diamond, Graphite and Fullerenes- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.49–1.50 Giant Covalent Structures; Diamond, Graphite and Fullerenes- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.49–1.50 Giant Covalent Structures; Diamond, Graphite and Fullerenes- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.49 explain why substances with giant covalent structures are solids with high melting and boiling points
1.50 explain how the structures of diamond, graphite and C₆₀ fullerene influence their physical properties
1.49 Giant Covalent Structures – High Melting and Boiling Points
Definition:
A giant covalent structure (also called a giant molecular structure) is a continuous network of atoms bonded together by strong covalent bonds.
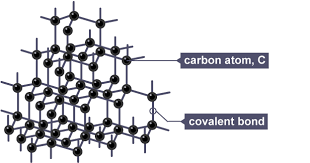
There are no separate molecules.
Examples
- \( \mathrm{Diamond} \)
- \( \mathrm{Graphite} \)
- \( \mathrm{Silicon\ dioxide\ (SiO_2)} \)
Structure Features
- Each atom forms several strong covalent bonds.
- The structure extends in three dimensions (except graphite layers).
- All atoms are linked in a giant network.
Why They Have High Melting and Boiling Points
The structure contains many strong covalent bonds.
To melt or boil the substance:
Strong covalent bonds must be broken.
Breaking covalent bonds requires a very large amount of energy.
Strong bonds → Large energy required → Very high melting/boiling points
| Structure Type | Forces Broken on Melting | Melting Point |
|---|---|---|
| Simple molecular | Intermolecular forces | Low |
| Giant covalent | Covalent bonds | Very high |
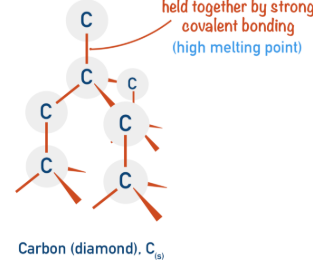
Example: Diamond
Each carbon atom forms four covalent bonds in a tetrahedral structure.
All atoms are bonded strongly in a 3D network.
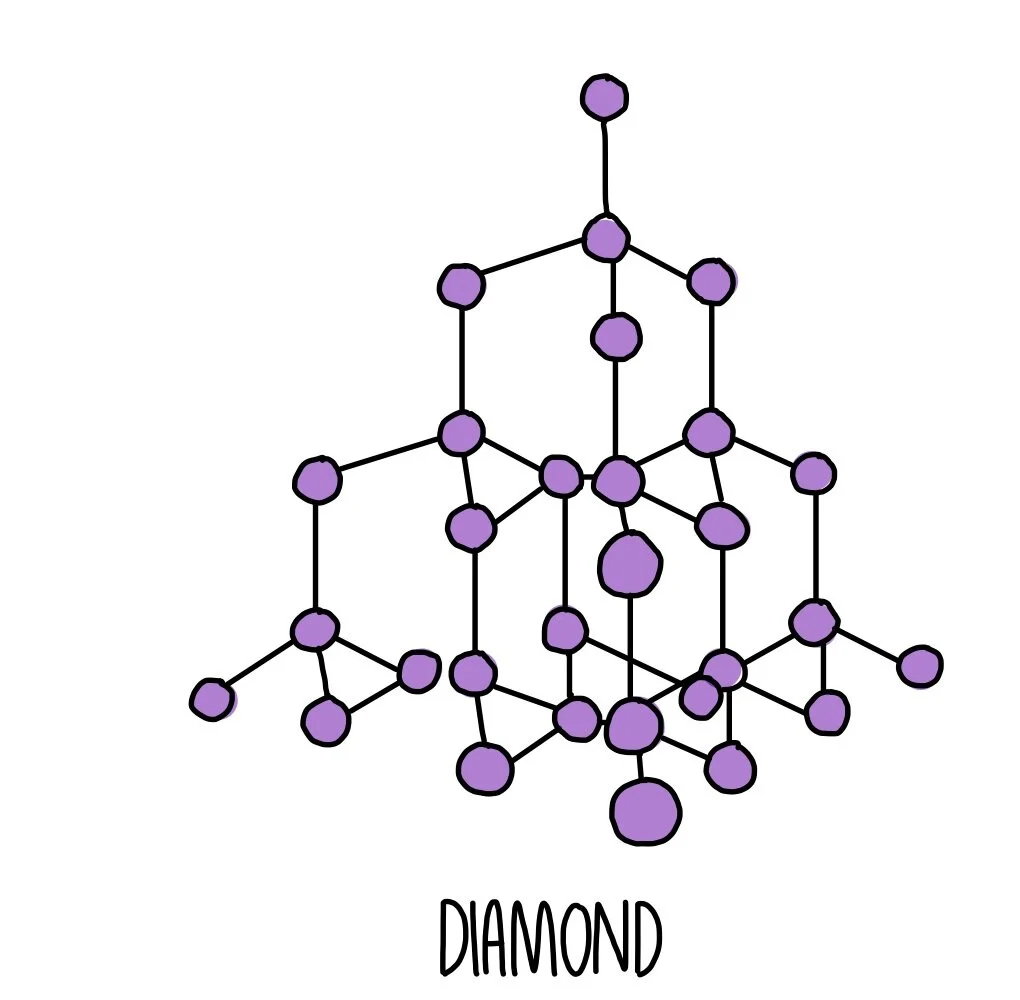
Therefore, diamond has an extremely high melting point.
Example: Silicon Dioxide
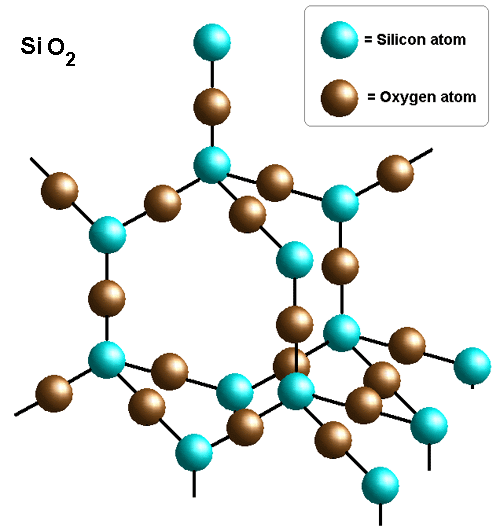
\( \mathrm{SiO_2} \)
Each silicon atom bonds to four oxygen atoms.
A giant 3D network is formed.
Strong covalent bonds give a high melting point.
GCSE Explanation Structure
- Giant covalent structure
- Many strong covalent bonds throughout
- Bonds must be broken to melt
- Large amount of energy required
- Therefore high melting and boiling points
Example 1 (Conceptual):
Why does diamond have a very high melting point?
▶️ Answer/Explanation
Diamond has a giant covalent structure.
There are many strong covalent bonds.
A large amount of energy is required to break these bonds.
Example 2 (Comparison):
Explain why carbon dioxide has a low melting point but silicon dioxide has a high melting point.
▶️ Answer/Explanation
\( \mathrm{CO_2} \) is simple molecular.
Only intermolecular forces are broken when melting.
\( \mathrm{SiO_2} \) has a giant covalent structure.
Strong covalent bonds must be broken.
Much more energy is required.
Example 3 (Hard):
Explain fully why giant covalent substances are solids at room temperature.
▶️ Answer/Explanation
Giant covalent substances consist of a network of atoms.
Atoms are joined by strong covalent bonds.
These bonds extend throughout the structure.
Large amounts of energy are required to break them.
At room temperature, there is not enough energy to break the bonds.
Therefore, they remain solid.
1.50 Structure and Properties of Diamond, Graphite and C60 Fullerene
All three substances are forms (allotropes) of carbon.
They contain only carbon atoms but have different structures.
Different structures lead to different physical properties.
1. Diamond
Structure:
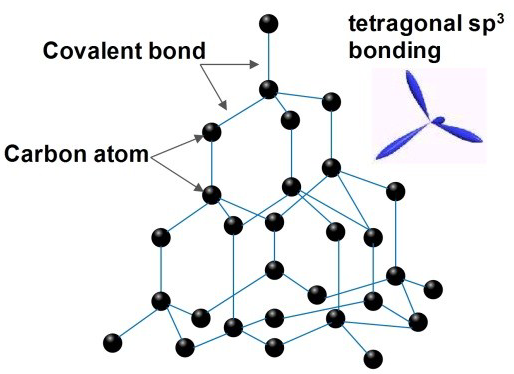
- Each carbon atom forms four covalent bonds.
- Tetrahedral arrangement.
- Giant 3D covalent network.
- No delocalised electrons.
Properties Explained:
- Very hard → Strong covalent bonds in all directions.
- Very high melting point → Many strong covalent bonds must be broken.
- Does not conduct electricity → No free or delocalised electrons.
2. Graphite
Structure:
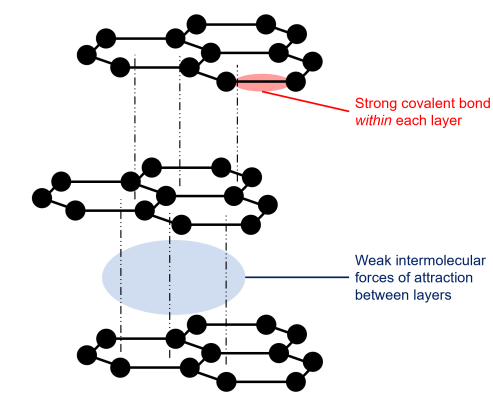
- Each carbon forms three covalent bonds.
- Layers of hexagonal rings.
- One electron per carbon is delocalised.
- Weak intermolecular forces between layers.
Properties Explained:
- Conducts electricity → Delocalised electrons can move along layers.
- Soft and slippery → Layers can slide over each other.
- High melting point → Strong covalent bonds within layers.
3. C60 Fullerene
Structure:
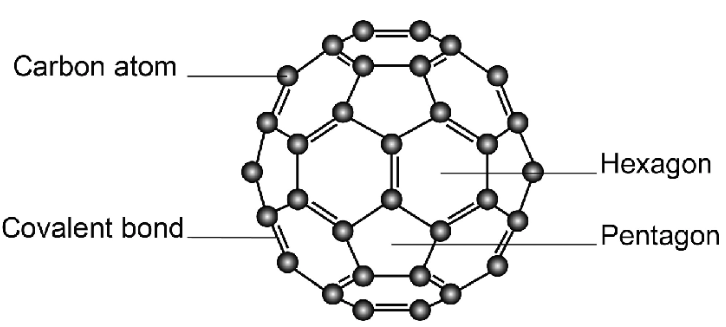
- Simple molecular substance.
- Molecules contain 60 carbon atoms.
- Spherical “football” shape.
- Weak intermolecular forces between molecules.
Properties Explained:
- Low melting point (compared to diamond/graphite) → Only intermolecular forces broken.
- Soft → Weak forces between molecules.
- Poor electrical conductor → No continuous network of delocalised electrons.
| Property | Diamond | Graphite | C60 |
|---|---|---|---|
| Structure Type | Giant covalent | Layered giant covalent | Simple molecular |
| Hardness | Very hard | Soft | Soft |
| Melting Point | Very high | Very high | Lower |
| Electrical Conductivity | No | Yes | Poor |
Key GCSE Linking Phrase
Different structures → Different bonding arrangements → Different physical properties.
Example 1 (Conceptual):
Why is diamond very hard but graphite is soft?
▶️ Answer/Explanation
Diamond has strong covalent bonds in all directions.
Graphite has layers with weak forces between them.
The layers can slide over each other.
Example 2 (deduction):
Why does graphite conduct electricity but diamond does not?
▶️ Answer/Explanation
Graphite has delocalised electrons that can move.
Diamond has no free electrons.
Moving electrons are needed for conduction.
Example 3 (Hard):
Explain fully why C60 has a much lower melting point than diamond.
▶️ Answer/Explanation
Diamond has a giant covalent structure.
Strong covalent bonds must be broken to melt it.
C60 consists of separate molecules.
Only weak intermolecular forces are broken when it melts.
Therefore, much less energy is required.
