Edexcel iGCSE Chemistry -1.52–1.53C Metallic Lattices and Metallic Bonding- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.52–1.53C Metallic Lattices and Metallic Bonding- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.52–1.53C Metallic Lattices and Metallic Bonding- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.52C know how to represent a metallic lattice by a 2-D diagram
1.53C understand metallic bonding in terms of electrostatic attractions
1.52C Representing a Metallic Lattice Using a 2-D Diagram
Definition:
A metallic lattice is a regular arrangement of positive metal ions surrounded by a sea of delocalised electrons.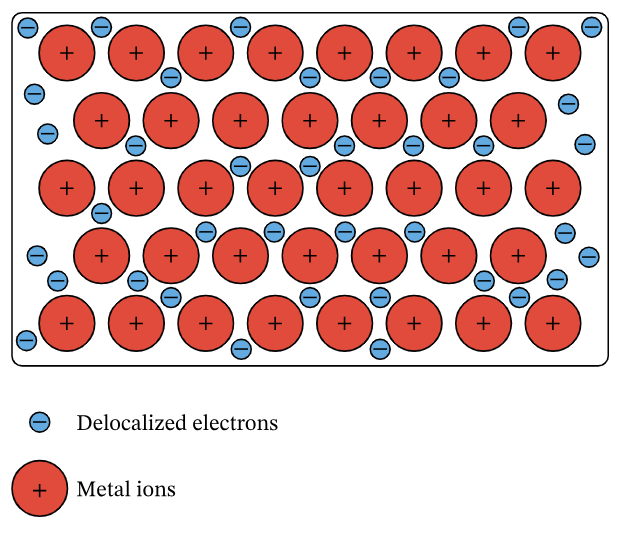
Metals have a giant structure held together by metallic bonding.
Metallic Bonding Explained
Metal atoms lose their outer electrons.
These electrons become delocalised (free to move).
The structure contains:
- Positive metal ions arranged in layers
- A ‘sea’ of delocalised electrons between them
The metallic bond is the strong electrostatic attraction between positive metal ions and delocalised electrons.
How to Draw a 2-D Metallic Lattice
In GCSE exams, a 2-D representation should show:
1. A regular arrangement of positive ions (e.g. \( \mathrm{Na^+} \)).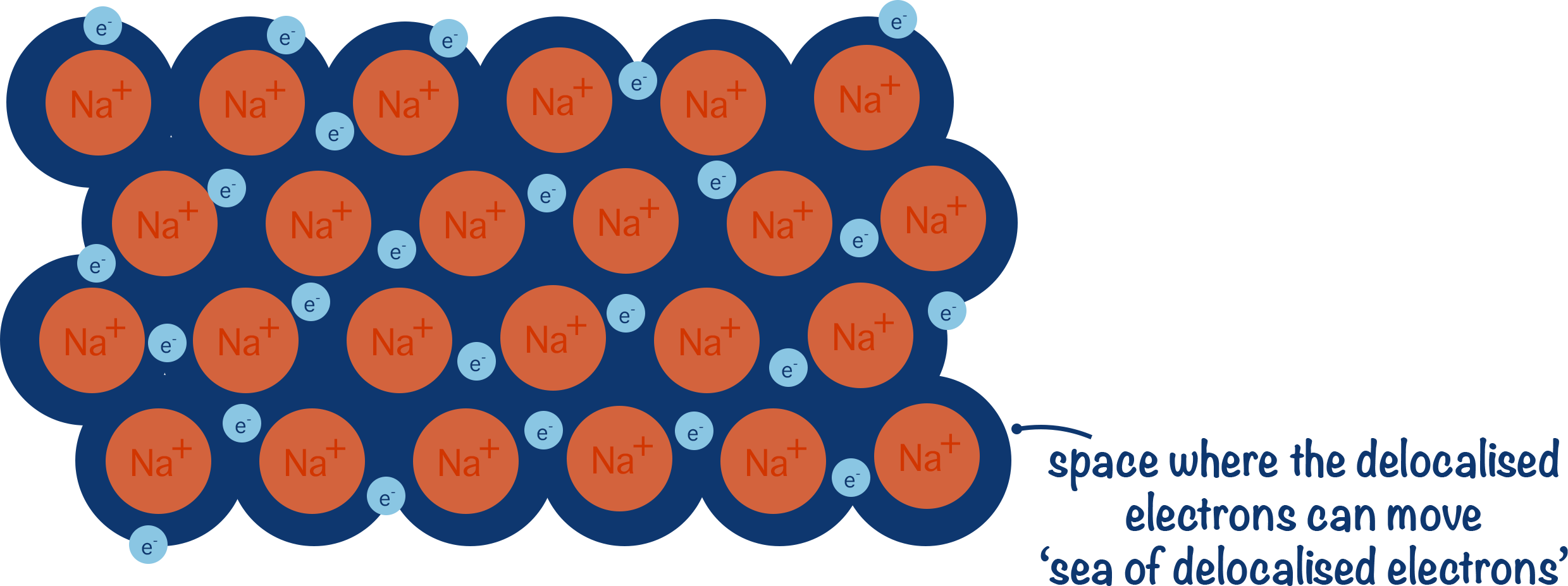
2. Charges clearly shown.
3. Small dots or crosses between ions to represent delocalised electrons.
4. Electrons not attached to any single ion.
Example (Sodium Metal)
\( \mathrm{Na} \rightarrow \mathrm{Na^+} + e^- \)
In the lattice:
• Many \( \mathrm{Na^+} \) ions in rows
• Delocalised electrons moving between them
Important: The diagram is 2-D, but the real structure is 3-D.
| Feature | What Must Be Shown |
|---|---|
| Ions | Positive charges labelled |
| Arrangement | Regular, repeating pattern |
| Electrons | Delocalised between ions |
How Structure Explains Properties
• Conduct electricity → Delocalised electrons can move
• High melting point → Strong attraction between ions and electrons
• Malleable → Layers of ions can slide but remain bonded
Example 1 (Conceptual):
Why must the ions be shown as positive in a metallic lattice diagram?
▶️ Answer/Explanation
Metal atoms lose outer electrons.
They form positive ions.
The positive charge must be shown to represent metallic bonding correctly.
Example 2 (Application):
Why must electrons be shown between ions rather than attached to one ion?
▶️ Answer/Explanation
Electrons are delocalised.
They are free to move throughout the lattice.
If attached to one ion, the diagram would incorrectly show covalent bonding.
Example 3 (Hard):
Explain fully how a 2-D metallic lattice diagram represents electrical conductivity in metals.
▶️ Answer/Explanation
The diagram shows positive metal ions in a regular pattern.
It also shows delocalised electrons between the ions.
These electrons are free to move throughout the lattice.
Moving charged particles carry electrical current.
Therefore, metals conduct electricity.
1.53C Metallic Bonding in Terms of Electrostatic Attractions
Definition:
Metallic bonding is the strong electrostatic attraction between positive metal ions and delocalised electrons.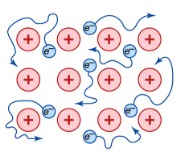
How Metallic Bonding Forms
Metal atoms have few outer shell electrons (usually 1–3).
These outer electrons become delocalised and are no longer attached to one specific atom.
As a result:
- Positive metal ions are formed
- A ‘sea’ of delocalised electrons surrounds them
The electrostatic attraction between the positively charged ions and negatively charged delocalised electrons holds the structure together.
Why the Bond is Strong
The metallic bond is strong because:
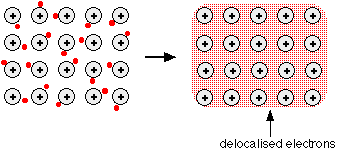
- There are many positive ions
- There are many delocalised electrons
- Electrostatic attraction acts in all directions throughout the lattice
A large amount of energy is required to overcome these attractions.
How Metallic Bonding Explains Properties
| Property | Explanation |
|---|---|
| High melting point | Strong electrostatic attraction requires lots of energy to overcome |
| Electrical conductivity | Delocalised electrons can move and carry charge |
| Malleability | Layers of ions can slide while attraction is maintained |
Comparison with Other Bonding
| Bond Type | Electrostatic Attraction Between |
|---|---|
| Ionic | Positive and negative ions |
| Covalent | Nuclei and shared electron pair |
| Metallic | Positive metal ions and delocalised electrons |
Key GCSE Explanation Structure
- Metal atoms lose outer electrons
- Positive ions formed
- Electrons become delocalised
- Strong electrostatic attraction between ions and electrons
- This attraction forms the metallic bond
Example 1 (Conceptual):
What particles are attracting in metallic bonding?
▶️ Answer/Explanation
Positive metal ions attract delocalised electrons.
This electrostatic attraction forms the metallic bond.
Example 2 (Application):
Why does copper conduct electricity?
▶️ Answer/Explanation
Copper has delocalised electrons.
These electrons are free to move.
Moving charged electrons carry electrical current.
Example 3 (Hard):
Explain fully why magnesium has a higher melting point than sodium in terms of metallic bonding.
▶️ Answer/Explanation
Magnesium forms \( \mathrm{Mg^{2+}} \) ions.
Sodium forms \( \mathrm{Na^+} \) ions.
Magnesium releases two delocalised electrons per atom.
There is stronger electrostatic attraction between ions and electrons in magnesium.
More energy is required to break these attractions.
Therefore magnesium has a higher melting point.
