Edexcel iGCSE Chemistry -1.54C Properties of Metals- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.54C Properties of Metals- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.54C Properties of Metals- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.54C explain typical physical properties of metals, including electrical conductivity and malleability
1.54C Physical Properties of Metals
Key Idea:
The typical physical properties of metals can be explained by metallic bonding.
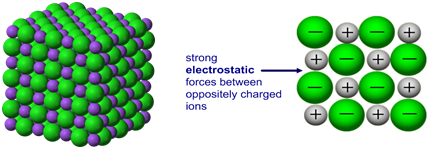
Metals consist of:
- Positive metal ions arranged in layers
- A sea of delocalised electrons
- Strong electrostatic attraction between them
1. Electrical Conductivity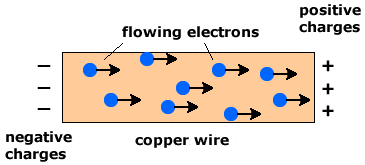
Metals are good electrical conductors.
This is because:
- Delocalised electrons are free to move
- These electrons carry electrical charge
- Moving charged particles create an electric current
Unlike ionic compounds, metals conduct electricity in both solid and molten states.
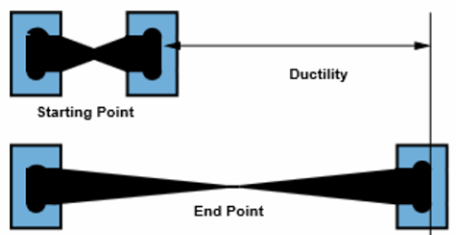
2. Malleability and Ductility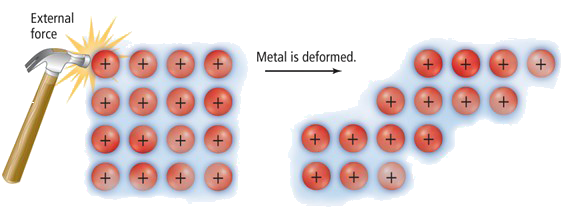
Malleable: Can be hammered into sheets.
Ductile: Can be drawn into wires.
This happens because:
- Layers of positive ions can slide over each other

- Delocalised electrons maintain attraction
- The metallic bond is not broken when layers shift
This prevents the metal from shattering.
3. High Melting and Boiling Points
Many metals have high melting points because:
- Metallic bonds are strong electrostatic attractions

- A large amount of energy is required to overcome them
4. Thermal Conductivity
Metals also conduct heat well because:
- Delocalised electrons transfer energy quickly
- Vibrations pass efficiently through closely packed ions
| Property | Explanation Using Metallic Bonding |
|---|---|
| Electrical conductivity | Free delocalised electrons carry charge |
| Malleability | Layers slide but attraction remains |
| High melting point | Strong electrostatic attraction |
| Thermal conductivity | Electrons transfer kinetic energy |
GCSE Explanation Structure
- Giant metallic lattice
- Positive ions + delocalised electrons
- Strong electrostatic attraction
- Property explained using movement of electrons or sliding of layers
Example 1 (Conceptual):
Why does aluminium conduct electricity when solid?
▶️ Answer/Explanation
Aluminium has delocalised electrons.
These electrons are free to move.
Moving charged electrons carry electrical current.
Example 2 (Application):
Why can copper be drawn into wires?
▶️ Answer/Explanation
Layers of positive ions can slide.
Delocalised electrons maintain attraction.
The metallic bond remains intact.
Example 3 (Hard):
Explain fully why metals are malleable but ionic compounds are brittle.
▶️ Answer/Explanation
In metals, positive ions are held by delocalised electrons.
When layers slide, attraction remains.
In ionic compounds, ions are fixed in a lattice.
If layers shift, like charges repel.
This causes the structure to shatter.
Therefore metals are malleable but ionic solids are brittle.