Edexcel iGCSE Chemistry -1.55C Electrical Conductivity- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.55C Electrical Conductivity- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.55C Electrical Conductivity- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.55C understand why covalent compounds do not conduct electricity
1.55C Why Covalent Compounds Do Not Conduct Electricity
Key Principle:
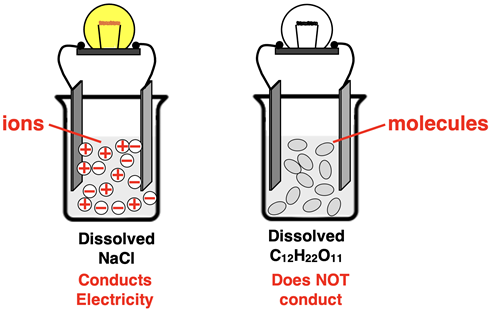
Electrical conductivity requires mobile charged particles.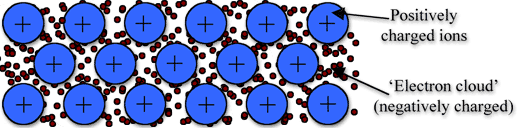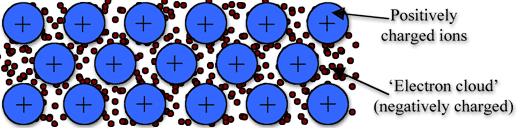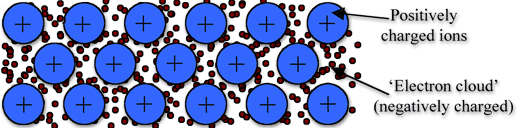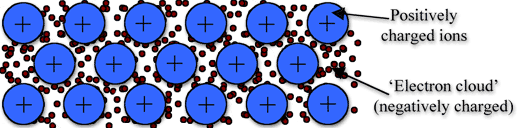
These mobile particles can be:
- Ions (as in molten ionic compounds)
- Delocalised electrons (as in metals)
Structure of Covalent Compounds
Covalent compounds consist of neutral molecules.
Within each molecule:
- Electrons are shared in covalent bonds
- Electrons are localised between specific atoms
- No ions are present
Because there are no free-moving charged particles, current cannot flow.
Behaviour in Different States
| State | Charged Particles Present? | Conducts Electricity? |
|---|---|---|
| Solid | No | No |
| Molten | No | No |
| Aqueous (if molecular) | No | No |
Important Clarification
Melting a covalent compound does not produce ions.
The molecules remain neutral.
Therefore, even when molten, most covalent compounds do not conduct electricity.
Example: Methane
\( \mathrm{CH_4} \)
- Methane consists of neutral molecules with shared electron pairs.
- There are no mobile ions or electrons.
- Therefore, methane does not conduct electricity.
Exception: Graphite
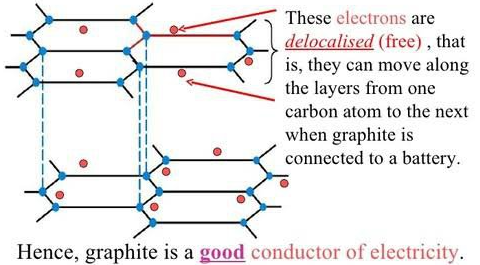
- Graphite conducts electricity because it has delocalised electrons.
- However, this is not typical of covalent compounds.
Example 1 (Conceptual):
Why does carbon dioxide not conduct electricity when molten?
▶️ Answer/Explanation
\( \mathrm{CO_2} \) consists of neutral molecules.
Melting does not form ions.
No mobile charged particles are present.
Therefore, no current flows.
Example 2 (Comparison):
Explain why molten sodium chloride conducts electricity but molten iodine does not.
▶️ Answer/Explanation
Molten sodium chloride contains free ions.
Molten iodine consists of neutral molecules.
Iodine has no mobile charged particles.
Therefore, iodine does not conduct.
Example 3 (Hard):
Explain fully why most covalent compounds remain poor electrical conductors in all physical states.
▶️ Answer/Explanation
Covalent compounds consist of molecules with shared electron pairs.
Electrons are localised in bonds.
There are no free ions.
Melting does not produce mobile charged particles.
Without mobile charges, electrical current cannot flow.
