Edexcel iGCSE Chemistry -1.56–1.57C Electrolysis of Ionic Compounds and Ions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.56–1.57C Electrolysis of Ionic Compounds and Ions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.56–1.57C Electrolysis of Ionic Compounds and Ions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.56C understand why ionic compounds conduct electricity only when molten or in aqueous solution
1.57C know that anion and cation are terms used to refer to negative and positive ions respectively
1.56C Electrical Conductivity of Ionic Compounds
Key Principle:
Electrical conductivity requires mobile charged particles.
In ionic compounds, the charged particles are ions.
Structure of Ionic Compounds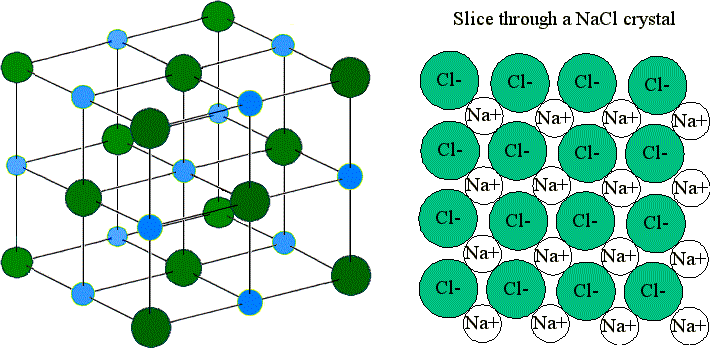
Ionic compounds consist of a giant ionic lattice.
They contain:
- Positive ions
- Negative ions
- Strong electrostatic attractions between oppositely charged ions
1. Solid Ionic Compounds
In the solid state:
- Ions are fixed in a regular lattice
- They cannot move
- There are no mobile charge carriers
Therefore, solid ionic compounds do not conduct electricity.
2. Molten Ionic Compounds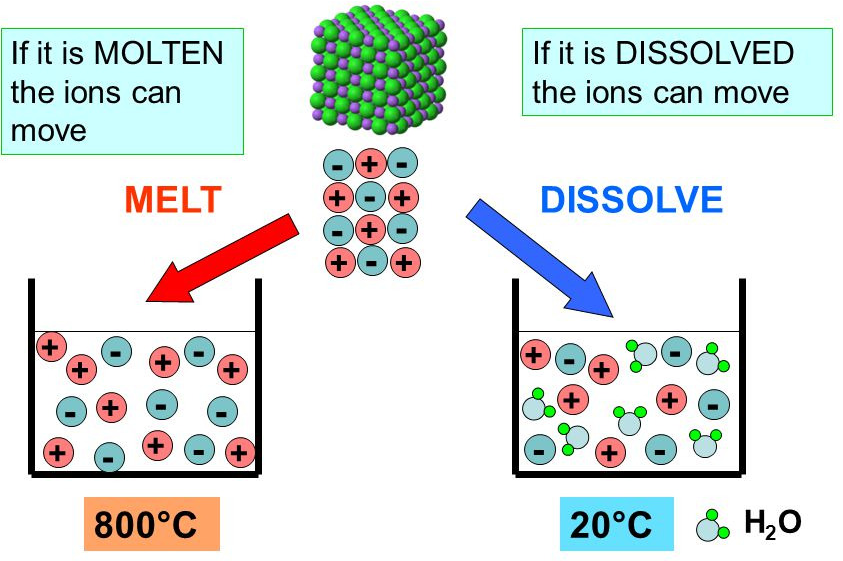
When heated until molten:
- The lattice breaks down
- Ions become free to move
- Mobile ions carry electrical charge
Therefore, molten ionic compounds conduct electricity.
3. Ionic Compounds in Aqueous Solution
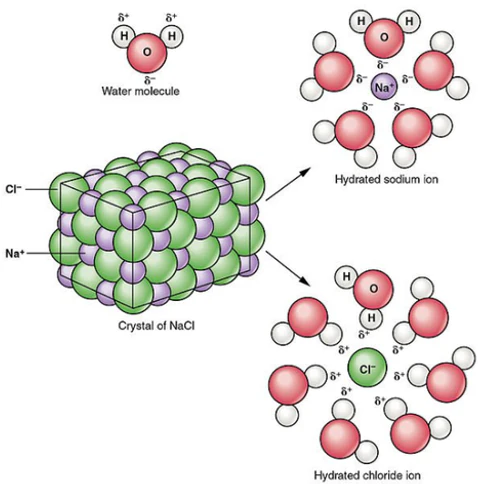
When dissolved in water:
\( \mathrm{NaCl(s)} \rightarrow \mathrm{Na^+(aq)} + \mathrm{Cl^-(aq)} \)
The ions separate and become free to move in solution.
Therefore, aqueous ionic solutions conduct electricity.
| State | Are Ions Free to Move? | Conducts Electricity? |
|---|---|---|
| Solid | No | No |
| Molten | Yes | Yes |
| Aqueous | Yes | Yes |
Example 1 (Conceptual):
Why does solid magnesium oxide not conduct electricity?
▶️ Answer/Explanation
\( \mathrm{MgO} \) consists of a giant ionic lattice.
The ions are fixed in position.
They cannot move.
Therefore, no electrical current flows.
Example 2 (Application):
Explain why molten potassium bromide conducts electricity.
▶️ Answer/Explanation
When molten, the ionic lattice breaks down.
\( \mathrm{K^+} \) and \( \mathrm{Br^-} \) ions become free to move.
Moving ions carry charge.
Therefore electricity flows.
Example 3 (Hard):
Explain fully why sodium chloride solution conducts electricity but solid sodium chloride does not.
▶️ Answer/Explanation
Solid sodium chloride has ions fixed in a lattice.
The ions cannot move.
When dissolved in water:
\( \mathrm{NaCl(s)} \rightarrow \mathrm{Na^+(aq)} + \mathrm{Cl^-(aq)} \)
The ions separate and are free to move.
Mobile charged particles carry current.
Therefore, the solution conducts electricity but the solid does not.
1.57C Anions and Cations 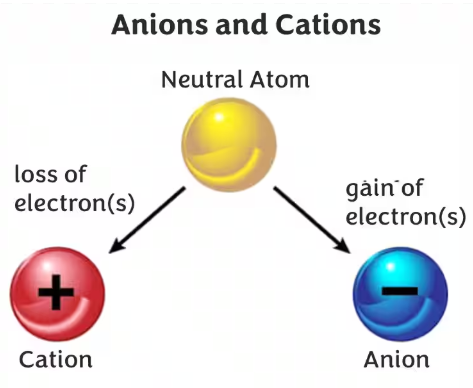
Definition:
- A cation is a positively charged ion.
- An anion is a negatively charged ion.
How Ions Form
Ions are formed when atoms gain or lose electrons.
Loss of electrons → Cation
\( \mathrm{Na} \rightarrow \mathrm{Na^+} + e^- \)
When an atom loses electrons, it has more protons than electrons, so it becomes positively charged.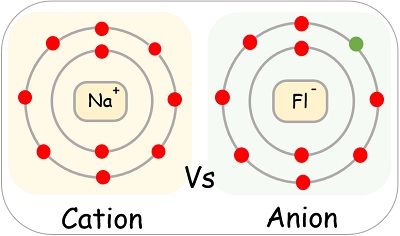
Gain of electrons → Anion
\( \mathrm{Cl} + e^- \rightarrow \mathrm{Cl^-} \)
When an atom gains electrons, it has more electrons than protons, so it becomes negatively charged.
Memory Tip
Cation → “Cats are positive” (starts with C and P in alphabetical order).
| Term | Charge | Formed By | Example |
|---|---|---|---|
| Cation | Positive (+) | Loss of electrons | \( \mathrm{Na^+} \) |
| Anion | Negative (–) | Gain of electrons | \( \mathrm{Cl^-} \) |
Why This Matters in Ionic Bonding
Ionic bonding is the strong electrostatic attraction between cations and anions.
Example 1 (Conceptual):
Why is \( \mathrm{Mg^{2+}} \) called a cation?
▶️ Answer/Explanation
Magnesium loses two electrons.
It becomes positively charged.
Positive ions are called cations.
Example 2 (Application):
Is \( \mathrm{O^{2-}} \) an anion or a cation? Explain.
▶️ Answer/Explanation
\( \mathrm{O^{2-}} \) has a negative charge.
It gained electrons.
Negative ions are called anions.
Example 3 (Hard):
Explain fully why a sodium ion is a cation and a chloride ion is an anion in sodium chloride.
▶️ Answer/Explanation
Sodium loses one electron:
\( \mathrm{Na} \rightarrow \mathrm{Na^+} + e^- \)
It has more protons than electrons.
It becomes positively charged, so it is a cation.
Chlorine gains one electron:
\( \mathrm{Cl} + e^- \rightarrow \mathrm{Cl^-} \)
It has more electrons than protons.
It becomes negatively charged, so it is an anion.
