Edexcel iGCSE Chemistry -1.58C Electrolysis Experiments- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.58C Electrolysis Experiments- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.58C Electrolysis Experiments- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.58C describe experiments to investigate electrolysis, using inert electrodes, of molten compounds and aqueous solutions, and to predict the products
1.58C Electrolysis with Inert Electrodes – Experiments and Products
Definition:
Electrolysis is the decomposition of an ionic compound using electricity.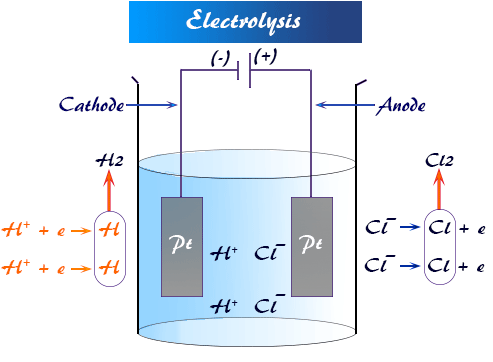
It requires:
- An electrolyte (molten or aqueous)
- Two electrodes
- A direct current power supply
In these experiments, inert electrodes (graphite or platinum) are used. They do not react.
Key Principles for Predicting Products
At the cathode (–): Reduction (gain of electrons).
At the anode (+): Oxidation (loss of electrons).
In molten compounds → only the compound’s ions are present.
In aqueous solutions → water provides \( \mathrm{H^+} \) and \( \mathrm{OH^-} \), so competition occurs.
A. Molten Lead(II) Bromide
Electrolyte: molten \( \mathrm{PbBr_2} \) 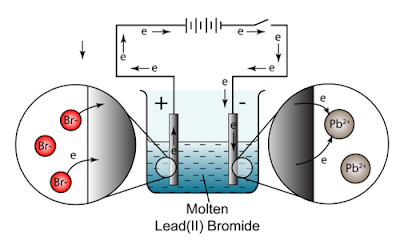
Ions present:
\( \mathrm{Pb^{2+}} \) and \( \mathrm{Br^-} \)
Cathode:
\( \mathrm{Pb^{2+} + 2e^- \rightarrow Pb} \)
Grey lead metal forms.
Anode:
\( \mathrm{2Br^- \rightarrow Br_2 + 2e^-} \)
Brown bromine gas forms.
B. Aqueous Sodium Chloride (Brine)
Ions present: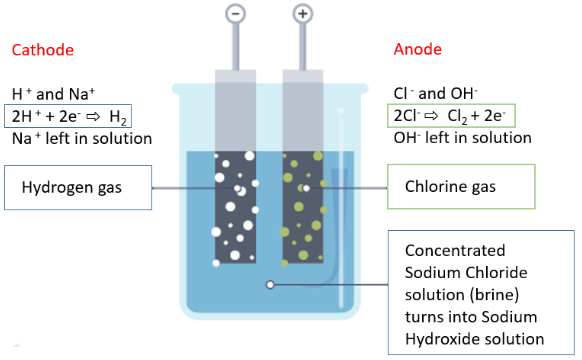
\( \mathrm{Na^+} \), \( \mathrm{Cl^-} \), \( \mathrm{H^+} \), \( \mathrm{OH^-} \)
Cathode:
Hydrogen is produced:
\( \mathrm{2H^+ + 2e^- \rightarrow H_2} \)
Anode:
Chlorine gas forms:
\( \mathrm{2Cl^- \rightarrow Cl_2 + 2e^-} \)
Remaining solution becomes sodium hydroxide.
C. Dilute Sulfuric Acid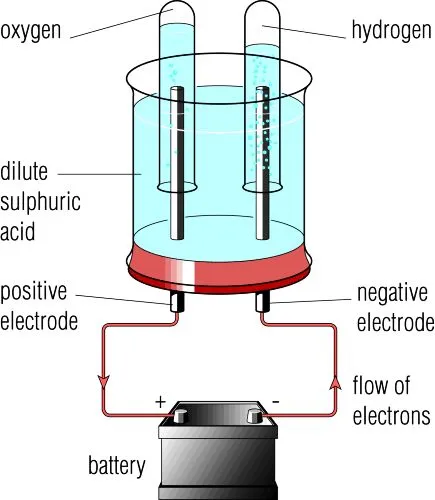
Ions present:
\( \mathrm{H^+} \), \( \mathrm{SO_4^{2-}} \), \( \mathrm{OH^-} \)
Cathode:
\( \mathrm{2H^+ + 2e^- \rightarrow H_2} \)
Anode:
\( \mathrm{4OH^- \rightarrow O_2 + 2H_2O + 4e^-} \)
Products: hydrogen and oxygen.
D. Aqueous Copper(II) Sulfate (Inert Electrodes)
Ions present: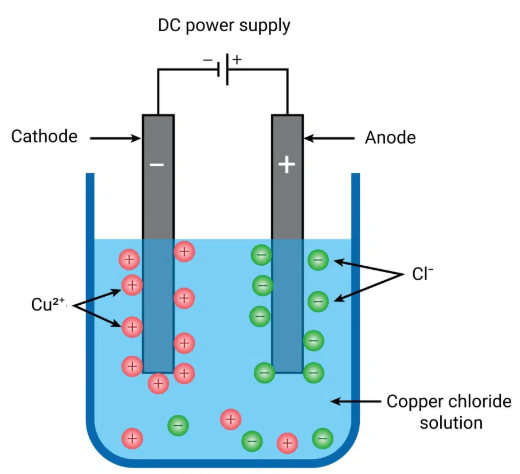
\( \mathrm{Cu^{2+}} \), \( \mathrm{SO_4^{2-}} \), \( \mathrm{H^+} \), \( \mathrm{OH^-} \)
Cathode:
\( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \)
Brown copper metal deposits.
Anode:
\( \mathrm{4OH^- \rightarrow O_2 + 2H_2O + 4e^-} \)
Oxygen gas forms.
| Electrolyte | Cathode Product | Anode Product |
|---|---|---|
| Molten \( \mathrm{PbBr_2} \) | Lead | Bromine |
| Aqueous \( \mathrm{NaCl} \) | Hydrogen | Chlorine |
| Dilute \( \mathrm{H_2SO_4} \) | Hydrogen | Oxygen |
| Aqueous \( \mathrm{CuSO_4} \) | Copper | Oxygen |
Example 1 (Conceptual):
Why must lead(II) bromide be molten before electrolysis?
▶️ Answer/Explanation
Ions in solid form are fixed in a lattice.
When molten, ions are free to move.
Mobile ions are needed to conduct electricity.
Example 2 (Prediction):
Predict the products of electrolysis of molten magnesium chloride.
▶️ Answer/Explanation
Ions present: \( \mathrm{Mg^{2+}} \), \( \mathrm{Cl^-} \)
Cathode: \( \mathrm{Mg^{2+} + 2e^- \rightarrow Mg} \)
Anode: \( \mathrm{2Cl^- \rightarrow Cl_2 + 2e^-} \)
Example 3 (Hard):
Explain fully why hydrogen is produced at the cathode during electrolysis of aqueous sodium chloride.
▶️ Answer/Explanation
In aqueous solution, \( \mathrm{Na^+} \) and \( \mathrm{H^+} \) ions are present.
Sodium is more reactive than hydrogen.
Therefore hydrogen ions are reduced instead.
\( \mathrm{2H^+ + 2e^- \rightarrow H_2} \)
Hydrogen gas is produced at the cathode.
