Edexcel iGCSE Chemistry -1.59C Half-Equations- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.59C Half-Equations- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.59C Half-Equations- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.59C write ionic half-equations representing the reactions at the electrodes during electrolysis and understand why these reactions are classified as oxidation or reduction
1.59C Ionic Half-Equations in Electrolysis and Oxidation/Reduction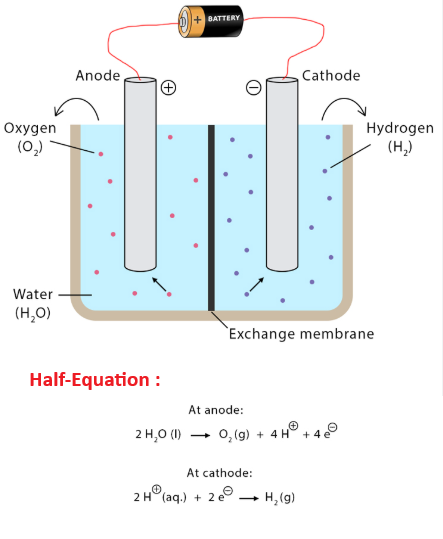
Key Idea:
A half-equation shows what happens at one electrode during electrolysis.
It must include:
- The correct ion
- Electrons (\( e^- \))
- Balanced charges and atoms
Oxidation and Reduction
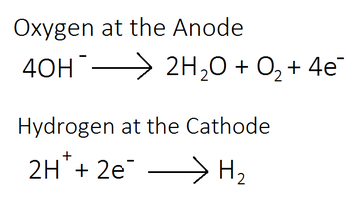
- Reduction = gain of electrons.
- Oxidation = loss of electrons.
- At the cathode (–): Reduction occurs.
- At the anode (+): Oxidation occurs.
Example 1 – Molten Lead(II) Bromide
Electrolyte: \( \mathrm{PbBr_2} \)
Cathode (Reduction):
\( \mathrm{Pb^{2+} + 2e^- \rightarrow Pb} \)
Lead ions gain electrons → reduction.
Anode (Oxidation):
\( \mathrm{2Br^- \rightarrow Br_2 + 2e^-} \)
Bromide ions lose electrons → oxidation.
Example 2 – Aqueous Copper(II) Sulfate (Inert Electrodes)
Cathode:
\( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \)
Reduction (gain of electrons).
Anode:
\( \mathrm{4OH^- \rightarrow O_2 + 2H_2O + 4e^-} \)
Oxidation (loss of electrons).
How to Write Half-Equations (Step Method)
1. Write the ion involved.
2. Write the product formed.
3. Add electrons to show gain or loss.
4. Check that charge balances on both sides.
| Electrode | Process | Electrons |
|---|---|---|
| Cathode (–) | Reduction | Electrons gained |
| Anode (+) | Oxidation | Electrons lost |
Why They Are Redox Reactions
Electrolysis always involves both:
- Reduction at one electrode
- Oxidation at the other electrode
This means electrolysis is a redox process.
Example 1 (Conceptual):
Why is \( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \) reduction?
▶️ Answer/Explanation
Electrons are gained.
Gain of electrons is reduction.
Example 2 (Application):
Write the half-equation for chlorine forming from chloride ions.
▶️ Answer/Explanation
\( \mathrm{2Cl^- \rightarrow Cl_2 + 2e^-} \)
Electrons are lost, so this is oxidation.
Example 3 (Hard):
Explain fully why electrolysis is described as a redox process using molten magnesium chloride as an example.
▶️ Answer/Explanation
Ions present: \( \mathrm{Mg^{2+}} \) and \( \mathrm{Cl^-} \).
Cathode: \( \mathrm{Mg^{2+} + 2e^- \rightarrow Mg} \) (reduction).
Anode: \( \mathrm{2Cl^- \rightarrow Cl_2 + 2e^-} \) (oxidation).
Electrons are gained at one electrode and lost at the other.
Therefore electrolysis involves both oxidation and reduction.
