Edexcel iGCSE Chemistry -1.60C Core Practical: Electrolysis- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.60C Core Practical: Electrolysis- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.60C Core Practical: Electrolysis- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.60C practical: investigate the electrolysis of aqueous solutions
1.60C Practical: Investigating the Electrolysis of Aqueous Solutions
Aim:
To investigate the products formed at the cathode and anode during the electrolysis of aqueous ionic solutions using inert electrodes.
Theory
Electrolysis is the decomposition of an ionic compound using electricity.
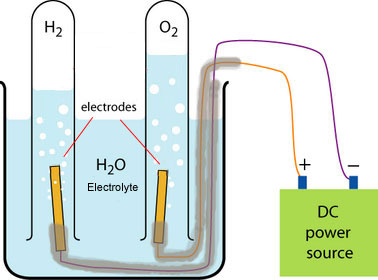
In aqueous solutions, ions from the compound and ions from water are present.
Water provides:
\( \mathrm{H^+} \) and \( \mathrm{OH^-} \)
At the cathode (−), reduction occurs (gain of electrons). At the anode (+), oxidation occurs (loss of electrons).
Apparatus
- Beaker
- Aqueous electrolyte (e.g. \( \mathrm{NaCl(aq)} \), \( \mathrm{CuSO_4(aq)} \), dilute \( \mathrm{H_2SO_4} \))
- Two inert graphite electrodes
- DC power supply
- Connecting wires and crocodile clips
- Test tubes (for gas collection)
- Wooden splints (for gas testing)
Method
- Pour the aqueous solution into a beaker.
- Place two inert graphite electrodes into the solution.
- Connect the electrodes to a DC power supply.
- Switch on the power supply.
- Observe changes at each electrode.
- Test any gases produced.
Example Investigation 1: Dilute Sulfuric Acid
Ions present:
\( \mathrm{H^+} \), \( \mathrm{SO_4^{2-}} \), \( \mathrm{OH^-} \)
Cathode (Reduction):
\( \mathrm{2H^+ + 2e^- \rightarrow H_2} \)
Observation: Colourless gas that produces a squeaky pop with a lit splint (hydrogen).
Anode (Oxidation):
\( \mathrm{4OH^- \rightarrow O_2 + 2H_2O + 4e^-} \)
Observation: Colourless gas that relights a glowing splint (oxygen).
Example Investigation 2: Aqueous Copper(II) Sulfate (Inert Electrodes)
Ions present:
\( \mathrm{Cu^{2+}} \), \( \mathrm{SO_4^{2-}} \), \( \mathrm{H^+} \), \( \mathrm{OH^-} \)
Cathode:
\( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \)
Observation: Pink-brown copper metal forms on the cathode.
Anode:
\( \mathrm{4OH^- \rightarrow O_2 + 2H_2O + 4e^-} \)
Observation: Oxygen gas bubbles form.
The blue solution gradually becomes paler as \( \mathrm{Cu^{2+}} \) ions are removed.
Example Investigation 3: Aqueous Sodium Chloride (Brine)
Ions present:
\( \mathrm{Na^+} \), \( \mathrm{Cl^-} \), \( \mathrm{H^+} \), \( \mathrm{OH^-} \)
Cathode:
\( \mathrm{2H^+ + 2e^- \rightarrow H_2} \)
Observation: Hydrogen gas produced.
Anode:
\( \mathrm{2Cl^- \rightarrow Cl_2 + 2e^-} \)
Observation: Green chlorine gas produced (bleaches damp litmus paper).
How to Predict Products (Exam Rules)
At the cathode:
- If the metal is more reactive than hydrogen → hydrogen forms.
- If the metal is less reactive than hydrogen → the metal forms.
At the anode:
- If halide ions are present → halogen forms.
- If no halide is present → oxygen forms.
Example 1 (Conceptual):
Why does copper form at the cathode during electrolysis of aqueous \( \mathrm{CuSO_4} \)?
▶️ Answer/Explanation
\( \mathrm{Cu^{2+}} \) ions gain electrons.
\( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \)
This is reduction.
Copper is less reactive than hydrogen.
Example 2 (Prediction):
Predict the products of electrolysis of aqueous calcium chloride.
▶️ Answer/Explanation
Calcium is more reactive than hydrogen.
Hydrogen forms at the cathode.
Chlorine forms at the anode.
Example 3 (Hard):
Explain fully the observations during electrolysis of aqueous sodium chloride.
▶️ Answer/Explanation
At the cathode:
\( \mathrm{2H^+ + 2e^- \rightarrow H_2} \)
Hydrogen gas forms.
At the anode:
\( \mathrm{2Cl^- \rightarrow Cl_2 + 2e^-} \)
Chlorine gas forms.
The remaining solution contains sodium hydroxide.
