Edexcel iGCSE Chemistry -1.8–1.9 Classification of Substances; Pure Substances and Mixtures- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -1.8–1.9 Classification of Substances; Pure Substances and Mixtures- Study Notes- New syllabus
Edexcel iGCSE Chemistry -1.8–1.9 Classification of Substances; Pure Substances and Mixtures- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
1.8 understand how to classify a substance as an element, compound or mixture
1.9 understand that a pure substance has a fixed melting and boiling point, but that a mixture may melt or boil over a range of temperatures
1.8 Classifying Substances as Elements, Compounds or Mixtures
All substances can be classified as elements, compounds or mixtures based on their composition and how their particles are arranged.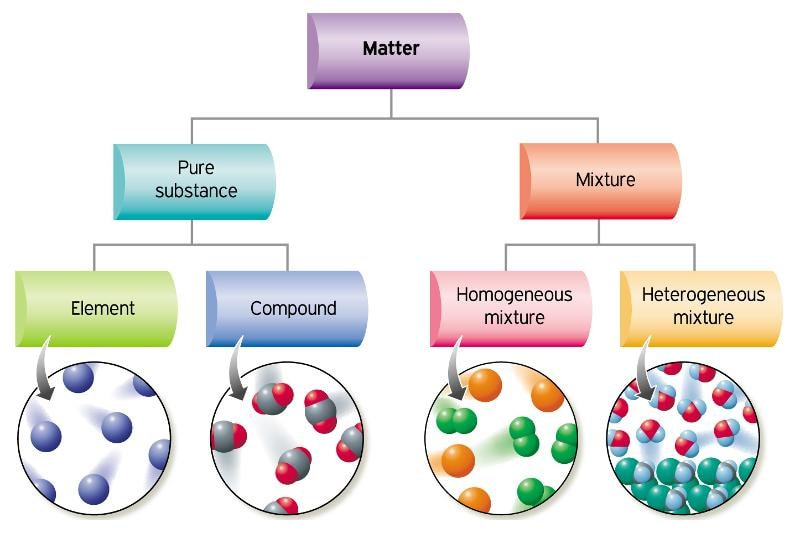
Element
An element is a substance made of only one type of atom.
Elements cannot be broken down into simpler substances by chemical means.
Examples include oxygen (\( \mathrm{O_2} \)), iron (\( \mathrm{Fe} \)) and carbon (\( \mathrm{C} \)).
Compound
A compound is a substance formed when two or more different elements are chemically bonded together.
Compounds have a fixed ratio of elements.
They can only be separated into elements by chemical reactions.
Example: Water (\( \mathrm{H_2O} \)) contains hydrogen and oxygen chemically combined.
Mixture
A mixture contains two or more substances physically combined, not chemically bonded.
The substances in a mixture can be in any proportion.
They can be separated by physical methods such as filtration or distillation.
Example: Air (a mixture of gases).
Key Differences
| Feature | Element | Compound | Mixture |
|---|---|---|---|
| Number of substances | One type of atom | Two or more elements | Two or more substances |
| Bonding | No bonding between different atoms | Chemically bonded | No chemical bonding |
| Ratio | Single element only | Fixed ratio | Any proportion |
| Separation method | Cannot be broken down chemically | Chemical reaction required | Physical methods |
Exam Tip
If the components are chemically bonded in a fixed ratio, it is a compound. If the components are not bonded and can be physically separated, it is a mixture.
Example 1 (Conceptual):
Magnesium reacts with oxygen to form magnesium oxide. Is magnesium oxide an element, compound or mixture?
▶️ Answer/Explanation
It is a compound.
Magnesium and oxygen are chemically bonded in a fixed ratio.
Example 2 (Numerical):
Water is made of hydrogen and oxygen in a mass ratio of 1:8. Does this suggest water is a mixture or compound? Explain.
▶️ Answer/Explanation
It suggests water is a compound.
The fixed mass ratio (1:8) shows the elements combine in a fixed proportion.
Example 3 (Hard):
A student mixes iron filings and sulfur. The mixture can be separated with a magnet. After heating strongly, a new substance forms that cannot be separated with a magnet. Explain fully what has happened.
▶️ Answer/Explanation
Before heating, iron and sulfur form a mixture.
They are not chemically bonded and can be separated physically using a magnet.
After heating, a chemical reaction occurs forming iron sulfide.
Iron and sulfur are now chemically bonded in a fixed ratio.
Iron sulfide is a compound and cannot be separated by physical methods.
1.9 Melting and Boiling Points of Pure Substances and Mixtures
Melting point and boiling point are physical properties used to identify substances and determine whether they are pure.
Pure Substance
A pure substance contains only one element or one compound with no other substances mixed in.
A pure substance has: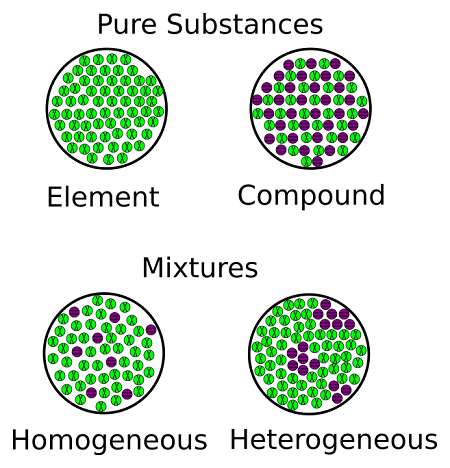
• A fixed melting point
• A fixed boiling point
This is because all particles are identical and have the same strength of attractive forces.
Example: Pure water boils at \( 100^\circ \mathrm{C} \) at standard pressure.
Mixture
A mixture contains two or more substances physically combined.
A mixture:
• Melts over a range of temperatures
• Boils over a range of temperatures
This happens because different substances in the mixture have different melting and boiling points.
Impurities usually:
• Lower the melting point
• Raise the boiling point
Comparison: Pure Substance vs Mixture
| Property | Pure Substance | Mixture |
|---|---|---|
| Composition | Single substance only | Two or more substances |
| Melting Point | Sharp / fixed temperature | Range of temperatures |
| Boiling Point | Sharp / fixed temperature | Range of temperatures |
| Effect of Impurities | Not applicable | Changes melting/boiling point |
Why This Happens (Particle Explanation)
In a pure substance, all particles have identical attractive forces, so they overcome these forces at the same temperature.
In a mixture, different particles have different attractive forces, so they require different amounts of energy to separate.
This causes melting or boiling to occur over a range rather than at one fixed point.
Example 1 (Conceptual):
A substance melts sharply at \( 80^\circ \mathrm{C} \). What does this suggest?
▶️ Answer/Explanation
It suggests the substance is pure.
A sharp melting point indicates all particles have identical bonding and overcome forces at the same temperature.
Example 2 (Numerical):
A solid begins to melt at \( 72^\circ \mathrm{C} \) and finishes melting at \( 78^\circ \mathrm{C} \).
Calculate the melting range and suggest whether it is pure.
▶️ Answer/Explanation
Melting range = \( 78 – 72 = 6^\circ \mathrm{C} \)
Since it melts over a range, it is likely a mixture or impure substance.
Example 3 (Hard):
Explain fully why adding salt to ice lowers its melting point.
▶️ Answer/Explanation
Pure ice has a fixed melting point of \( 0^\circ \mathrm{C} \).
When salt is added, it forms a mixture with the ice.
The salt particles disrupt the regular arrangement of water molecules.
This weakens the attractive forces between water molecules.
Less energy is required to overcome these forces, so melting occurs at a lower temperature.
