Edexcel iGCSE Chemistry -2.1 Reactions of Alkali Metals with Water- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.1 Reactions of Alkali Metals with Water- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.1 Reactions of Alkali Metals with Water- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.1 understand how the similarities in the reactions of these elements with water provide evidence for their recognition as a family of elements
2.1 Similarities in Reactions with Water – Evidence for a Family of Elements
Some elements show very similar chemical reactions. These similarities provide evidence that they belong to the same family (group) in the Periodic Table.
A key example is Group 1 (alkali metals).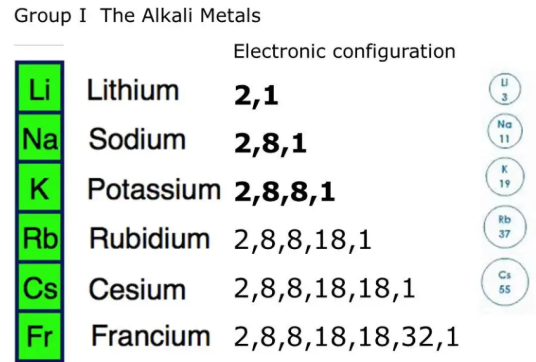
Group 1 Elements
- Lithium (Li)
- Sodium (Na)
- Potassium (K)
These metals all react with water in a similar way.
Reaction with Water
General equation:
\( \mathrm{2M(s) + 2H_2O(l) \rightarrow 2MOH(aq) + H_2(g)} \)
Where \( \mathrm{M} \) represents a Group 1 metal.
Example (Sodium):
\( \mathrm{2Na(s) + 2H_2O(l) \rightarrow 2NaOH(aq) + H_2(g)} \)
Common Observations
- Metal floats on water.
- Fizzing (hydrogen gas released).
- Metal moves around on surface.
- Forms an alkaline solution (metal hydroxide).
These similar observations show that they react in the same pattern.
Trend Down the Group
- Reactivity increases down the group.
- Lithium reacts gently.
- Sodium reacts more vigorously.
- Potassium reacts very vigorously (may ignite).
Although reactivity changes, the type of reaction remains the same.
| Element | Product Formed | Gas Produced | Relative Reactivity |
|---|---|---|---|
| Lithium | Lithium hydroxide | Hydrogen | Low |
| Sodium | Sodium hydroxide | Hydrogen | Medium |
| Potassium | Potassium hydroxide | Hydrogen | High |
Why This Is Evidence for a Family
- All form metal hydroxides.
- All produce hydrogen gas.
- All form alkaline solutions.
- The pattern of reaction is consistent.
Because they react in similar ways, they are placed in the same group (family) in the Periodic Table.
Example 1 (Conceptual):
Why are lithium, sodium and potassium placed in the same group?
▶️ Answer/Explanation
They react with water in the same way.
They form hydroxides and hydrogen gas.
They show similar chemical properties.
Example 2 (Application):
Write the balanced equation for the reaction between potassium and water.
▶️ Answer/Explanation
\( \mathrm{2K(s) + 2H_2O(l) \rightarrow 2KOH(aq) + H_2(g)} \)
Example 3 (Hard):
Explain fully how the reactions of Group 1 metals with water provide evidence that they are a family of elements.
▶️ Answer/Explanation
All Group 1 metals react with water to form a metal hydroxide and hydrogen gas.
The same type of products is formed in each case.
The reactions show similar observations such as fizzing and alkaline solutions.
The reactivity increases down the group but the pattern remains consistent.
This similarity in chemical behaviour shows they belong to the same family.
