Edexcel iGCSE Chemistry -2.10 Determination of Oxygen in Air- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -2.10 Determination of Oxygen in Air- Study Notes- New syllabus
Edexcel iGCSE Chemistry -2.10 Determination of Oxygen in Air- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
2.10 understand how to determine the percentage by volume of oxygen in air using experiments involving the reactions of metals (e.g. iron) and non-metals (e.g. phosphorus) with air
2.10 Determining the Percentage by Volume of Oxygen in Air
The percentage of oxygen in air can be determined experimentally by removing oxygen from a known volume of air and measuring the decrease in volume.
This can be done using reactions with:
- A metal (e.g. iron)
- A non-metal (e.g. phosphorus)
Method 1: Using Iron (Rusting Experiment) 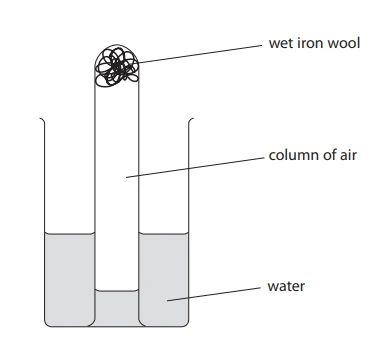
Iron wool is placed in an inverted measuring cylinder over water.
- The iron reacts slowly with oxygen in the trapped air.
- Rust forms.
- Oxygen is removed from the air.
- Water rises to replace the volume of oxygen used.
Chemical reaction (simplified):
\( \mathrm{4Fe(s) + 3O_2(g) \rightarrow 2Fe_2O_3(s)} \)
Method 2: Using Phosphorus
- Phosphorus is placed in a sealed container over water.

- It is ignited.
- Phosphorus reacts rapidly with oxygen.
- White phosphorus(V) oxide forms.
- The volume of air decreases.
- Water rises to fill the space left by oxygen.
Chemical equation:
\( \mathrm{4P(s) + 5O_2(g) \rightarrow P_4O_{10}(s)} \)
How the Percentage is Calculated
If the initial volume of air is known and the decrease in volume is measured:
\( \mathrm{Percentage\ of\ oxygen = \dfrac{Volume\ decrease}{Initial\ volume} \times 100} \)
The volume decrease represents the volume of oxygen removed.
Expected Result
- Approximately 20–21% of the air volume is removed.
- This shows oxygen makes up about one-fifth of air.
| Method | Substance Used | Observation | Why Volume Decreases |
|---|---|---|---|
| Rusting | Iron | Water level rises slowly | Oxygen reacts to form iron oxide |
| Combustion | Phosphorus | White smoke, water rises | Oxygen reacts to form phosphorus oxide |
Example 1 (Conceptual):
Why does water rise inside the measuring cylinder during the experiment?
▶️ Answer/Explanation
Oxygen reacts and is removed from the air.
This reduces the volume of gas inside.
Water rises to fill the empty space.
Example 2 (Numerical):
An experiment starts with 100 cm³ of air. After the reaction, 79 cm³ of gas remains. Calculate the percentage of oxygen in air.
▶️ Answer/Explanation
Volume decrease = \( \mathrm{100 – 79 = 21\ cm^3} \)
\( \mathrm{Percentage = \dfrac{21}{100} \times 100 = 21\%} \)
Example 3 (Hard):
Explain fully how reacting phosphorus with air allows the percentage of oxygen in air to be determined.
▶️ Answer/Explanation
Phosphorus reacts completely with oxygen in a sealed container.
The reaction forms solid phosphorus(V) oxide.
Oxygen is removed from the air inside the container.
This causes a decrease in gas volume.
Water rises to fill the space left by oxygen.
The decrease in volume represents the volume of oxygen originally present.
By dividing the volume decrease by the initial volume and multiplying by 100, the percentage of oxygen can be calculated.
The result is approximately 21%.